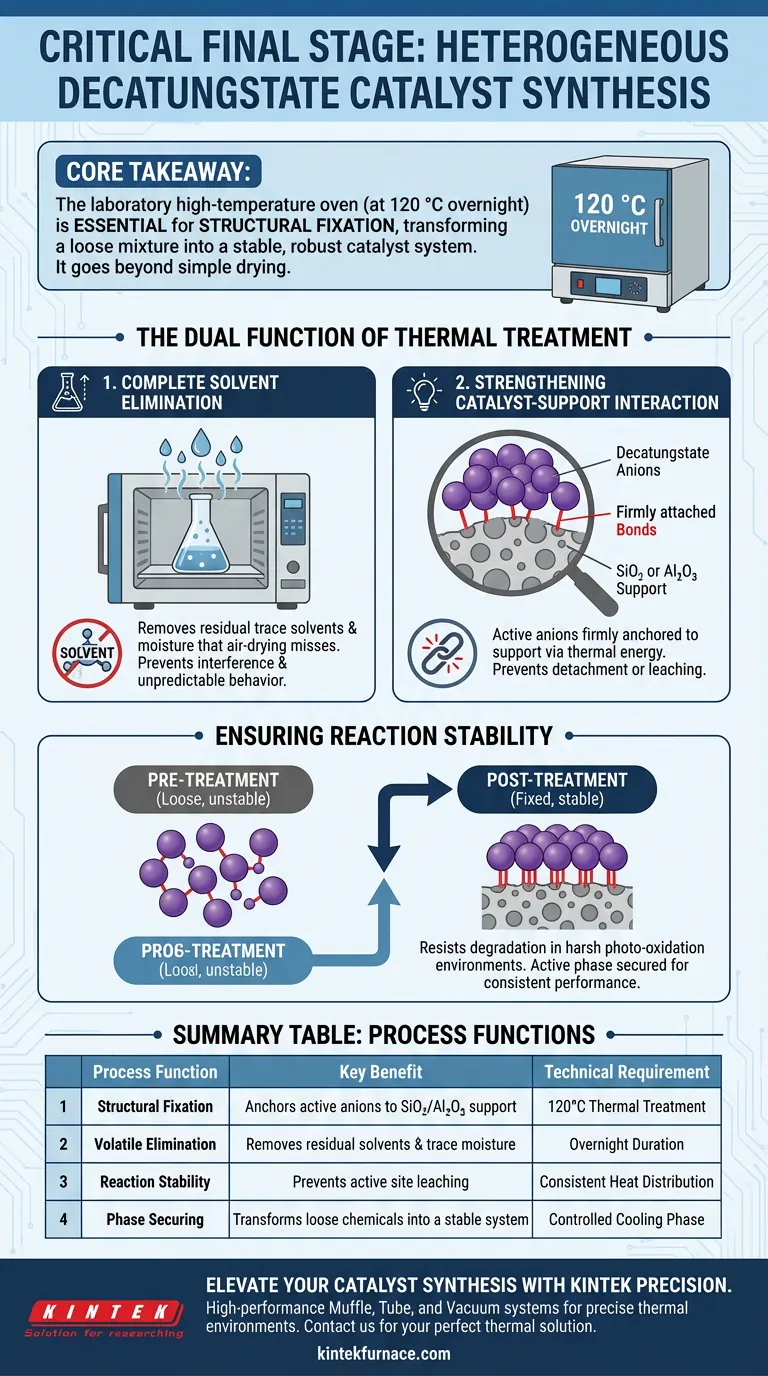
The laboratory high-temperature oven serves as the definitive stabilization step in the synthesis of heterogeneous decatungstate catalysts.
Specifically, this equipment is used to subject the supported catalyst materials to a thermal treatment at 120 °C overnight. This process is not merely for drying; it is essential for structurally anchoring the active decatungstate anions to the carrier support, ensuring the material is robust enough for practical use.
Core Takeaway While drying removes unwanted liquids, the primary function of this thermal treatment is structural fixation. By heating the material to 120 °C, you convert a loose association of chemicals into a stable, unified catalyst system capable of withstanding the rigors of photo-oxidation reactions without degrading.

The Dual Function of Thermal Treatment
To understand why this step is non-negotiable, we must look beyond simple evaporation. The high-temperature oven performs two simultaneous critical functions.
Complete Solvent Elimination
The first and most obvious function is the total removal of volatiles.
During the preparation phase, the catalyst materials are exposed to solvents and moisture.
The oven ensures the removal of residual trace solvents and moisture that simple air-drying might miss.
Eliminating these impurities is vital because retained solvent can interfere with the catalyst's active sites or cause unpredictable behavior during the reaction phase.
Strengthening Catalyst-Support Interaction
The deeper, more critical reason for this step is the strengthening of physical and chemical bonds.
The decatungstate anions (the active part of the catalyst) must be firmly attached to the carrier surface, typically SiO2 (silica) or Al2O3 (alumina).
The thermal energy provided at 120 °C drives the physical adsorption or chemical bonding processes.
Without this heat treatment, the active components might sit loosely on the surface, liable to detach or leach out during liquid-phase reactions.
Ensuring Reaction Stability
The ultimate goal of this preparation stage is to ensure the catalyst performs reliably over time.
Preventing Degradation
The stability of the catalyst during subsequent photo-oxidation reactions is directly dependent on this oven treatment.
Photo-oxidation environments can be harsh. A catalyst that has not been thermally "locked" into place may suffer from active site leaching.
Securing the Active Phase
By reinforcing the bond between the anion and the support, the oven ensures the catalyst maintains its integrity.
This distinct step transforms the precursor materials into a heterogeneous system that behaves consistently under operational conditions.
Understanding the Trade-offs
While the high-temperature oven is necessary, it is important to understand the specific parameters required for decatungstate compared to other methods.
Temperature Specificity
For decatungstate catalysts, 120 °C is the optimal set point found to balance drying speed with structural integrity.
This contrasts with other catalyst preparations (such as those mentioned in supplementary contexts) where lower-temperature vacuum drying (around 40–100 °C) might be used to prevent the migration of metal salts.
The Risk of Migration
In general catalyst preparation, rapid heating can sometimes cause active components to migrate to the surface (the "eggshell" effect).
However, for this specific decatungstate synthesis, the interaction with the silica or alumina support requires the higher thermal energy of 120 °C to effectively solidify the bond, rather than just gently evaporating the solvent.
Making the Right Choice for Your Goal
When finalizing your catalyst preparation protocol, consider these specific objectives:
- If your primary focus is Structural Integrity: Ensure the oven is set strictly to 120 °C and the duration is sufficient (overnight) to maximize the bonding between the decatungstate and the SiO2/Al2O3 support.
- If your primary focus is Reproducibility: strictly control the cooling phase after the oven treatment to prevent re-adsorption of atmospheric moisture before the catalyst is stored or used.
The high-temperature oven transforms your material from a simple mixture of components into a unified, stable tool ready for complex chemical transformations.
Summary Table:
| Process Function | Key Benefit | Technical Requirement |
|---|---|---|
| Structural Fixation | Anchors active anions to SiO2/Al2O3 support | 120°C Thermal Treatment |
| Volatile Elimination | Removes residual solvents and trace moisture | Overnight Duration |
| Reaction Stability | Prevents active site leaching during oxidation | Consistent Heat Distribution |
| Phase Securing | Transforms loose chemicals into a stable system | Controlled Cooling Phase |
Elevate Your Catalyst Synthesis with KINTEK Precision
High-performance heterogeneous catalysts require precise thermal environments to ensure long-term stability and structural integrity. Backed by expert R&D and manufacturing, KINTEK offers high-performance Muffle, Tube, and Vacuum systems, all customizable to meet your laboratory's unique high-temperature needs.
Whether you are preparing decatungstate catalysts or advanced CVD materials, our equipment provides the uniform heating necessary for critical bonding and solvent elimination. Don't compromise your research—contact us today to find the perfect thermal solution for your lab.
Visual Guide
References
- Julia Ong, J. C. Scaiano. Comparison of Composite Materials Designed to Optimize Heterogeneous Decatungstate Oxidative Photocatalysis. DOI: 10.3390/molecules30173597
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 1800℃ High Temperature Muffle Oven Furnace for Laboratory
- 1700℃ High Temperature Muffle Oven Furnace for Laboratory
- 1200℃ Muffle Oven Furnace for Laboratory
- 1400℃ Muffle Oven Furnace for Laboratory
- High Temperature Muffle Oven Furnace for Laboratory Debinding and Pre Sintering
People Also Ask
- What functions does a high-temperature muffle furnace perform during cathode precursor processing?
- What is the critical role of a laboratory high-temperature muffle furnace in TiO2/LDH? Unlock Superior Crystallization
- Why is calcination essential for NaFePO4 phase formation? Engineering High-Performance Sodium Iron Phosphate
- What is the function of a muffle furnace in 550°C NiCuCe catalyst calcination? Master Your Thermal Transformation
- How does a high-temperature laboratory muffle furnace affect material properties? Transform Anodic Oxide Films Fast














