Drying glassware in a 140 °C oven overnight is a mandatory purification step specifically designed to strip away microscopic layers of water molecules that cling to glass surfaces. Because Group Transfer Polymerization (GTP) is an "active" reaction, even invisible trace moisture acts as a potent contaminant that can chemically deactivate the reaction components and ruin the polymer's structure.
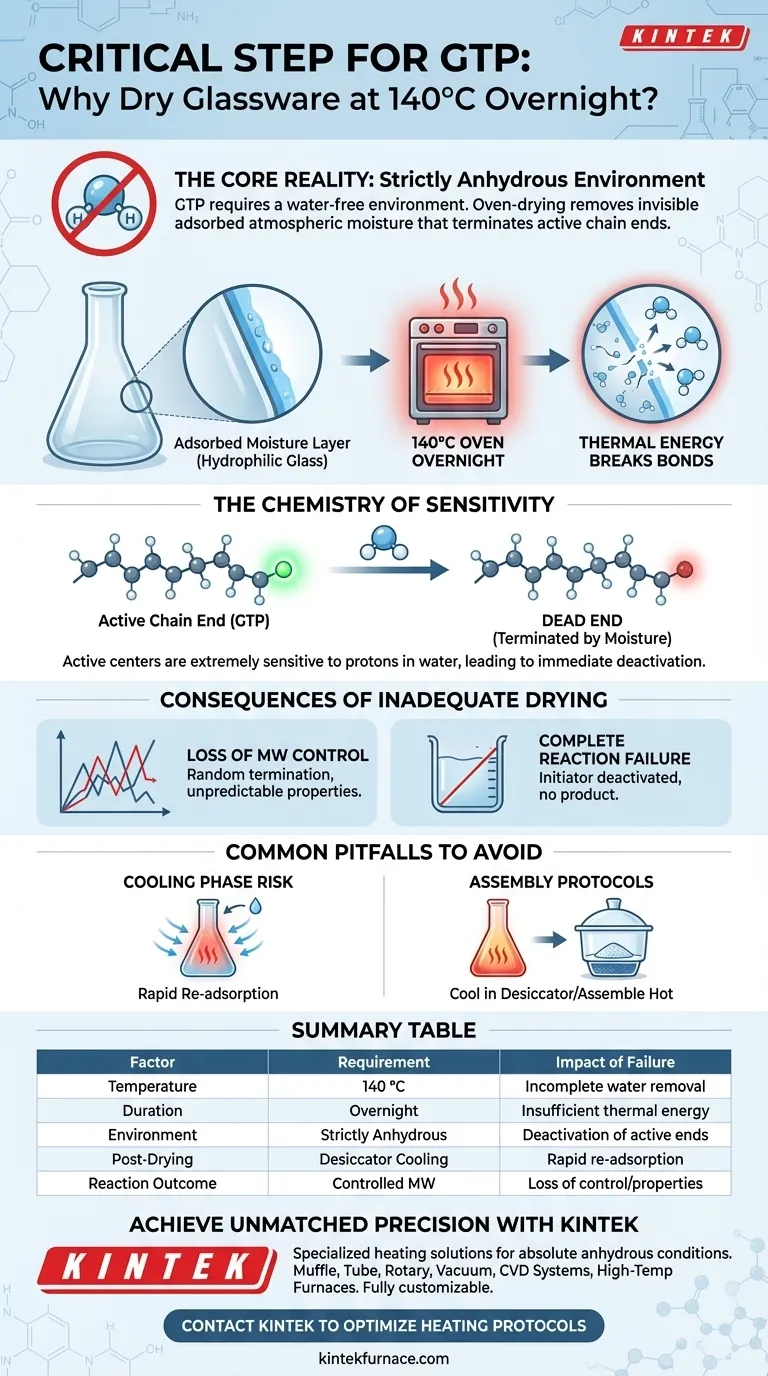
The Core Reality: GTP requires a strictly anhydrous (water-free) environment to function correctly. The oven-drying process is not about drying visible liquid, but about removing adsorbed atmospheric moisture that would otherwise terminate active chain ends and destroy molecular weight control.

The Chemistry of Sensitivity
Vulnerability of Active Centers
GTP relies on specific chemical groups—active chain ends—to continuously add monomer units to the growing polymer chain. These active centers are extremely sensitive to protons found in water.
Immediate Deactivation
When moisture is present, it reacts immediately with the initiator or the growing polymer chain. This reaction effectively "kills" the active center, transforming it into a dormant species that can no longer participate in polymerization.
Why Glass Requires Extreme Heat
The Invisible Water Layer
Glass surfaces are hydrophilic, meaning they naturally attract and hold water from the atmosphere. Even glassware that appears bone-dry to the naked eye possesses a thin film of adsorbed moisture on its surface.
Breaking the Bonds
Simply wiping the glass or air-drying it is insufficient to remove this bound layer. Sustained heating at 140 °C overnight provides the necessary thermal energy to break the physical bonds holding water molecules to the glass, driving them off completely.
Consequences of Inadequate Drying
Loss of Molecular Weight Control
One of the primary advantages of GTP is the ability to precisely control how long the polymer chain grows. If trace water randomly terminates some chains early, you lose this control, resulting in a polymer mixture with unpredictable properties.
Complete Reaction Failure
In scenarios where the moisture content is high relative to the initiator, the water may deactivate the initiator before the reaction even begins. This leads to complete failure of the polymerization, yielding no polymer product at all.
Common Pitfalls to Avoid
The Cooling Phase Risk
A common mistake is drying the glassware properly but allowing it to cool in open air. Glass will rapidly re-adsorb moisture from the humidity in the room as it cools.
Assembly Protocols
To maintain the anhydrous state achieved by the oven, glassware should typically be assembled while hot or cooled inside a desiccator filled with an active drying agent. Failing to protect the glass during the transition from oven to benchtop negates the overnight drying process.
Ensuring Success in Your Polymerization
To achieve consistent results in your GTP reactions, apply the following principles:
- If your primary focus is Precision: Ensure strictly anhydrous conditions to maintain the "active" nature of the chain ends for exact molecular weight targeting.
- If your primary focus is Yield: Eliminate all trace moisture to prevent initiator destruction, ensuring the maximum number of chains can propagate.
Treat the exclusion of water not just as a cleaning step, but as a fundamental chemical requirement for the reaction to exist.
Summary Table:
| Factor | Requirement in GTP | Impact of Failure |
|---|---|---|
| Temperature | 140 °C | Incomplete removal of adsorbed water molecules |
| Duration | Overnight | Insufficient thermal energy to break surface bonds |
| Environment | Strictly Anhydrous | Deactivation of active chain ends (reaction termination) |
| Post-Drying | Desiccator Cooling | Rapid re-adsorption of atmospheric humidity |
| Reaction Outcome | Controlled MW | Loss of molecular weight control and unpredictable properties |
Achieve Unmatched Precision in Your Polymer Research
Successful Group Transfer Polymerization (GTP) demands more than just a process; it requires high-performance equipment capable of maintaining strict thermal environments. KINTEK provides the specialized tools your lab needs to ensure absolute anhydrous conditions.
Backed by expert R&D and manufacturing, KINTEK offers a comprehensive range of Muffle, Tube, Rotary, Vacuum, and CVD systems, alongside premium lab high-temp furnaces—all fully customizable to meet your unique research requirements. Don't let trace moisture compromise your polymer's molecular weight or reaction yield.
Contact KINTEK today to optimize your lab’s heating protocols and secure the consistency your research deserves.
Visual Guide
References
- Xu Liu, Theoni K. Georgiou. Graphene inks for printing based on thermoresponsive ABC triblock terpolymer gels. DOI: 10.1039/d5lp00071h
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 1200℃ Muffle Oven Furnace for Laboratory
- 1400℃ Muffle Oven Furnace for Laboratory
- 1800℃ High Temperature Muffle Oven Furnace for Laboratory
- High Temperature Muffle Oven Furnace for Laboratory Debinding and Pre Sintering
- Laboratory Muffle Oven Furnace with Bottom Lifting
People Also Ask
- Why is a high-vacuum furnace required for CP-Ti annealing? Protect Purity and Prevent Embrittlement
- Why are synthesized CdS nanorods dried in a laboratory vacuum oven? Preserving Nanostructure and Chemical Integrity
- Why is high-purity argon protection required during the casting of alloy steel samples? Preserve Sample Integrity
- What functions does glucose perform in lithium-ion sieve synthesis? Enhance Carbothermal Reduction for LiMnO2 Purity
- What is the function of vacuum & heating in aluminum degassing? Enhancing Composite Integrity & Density



















