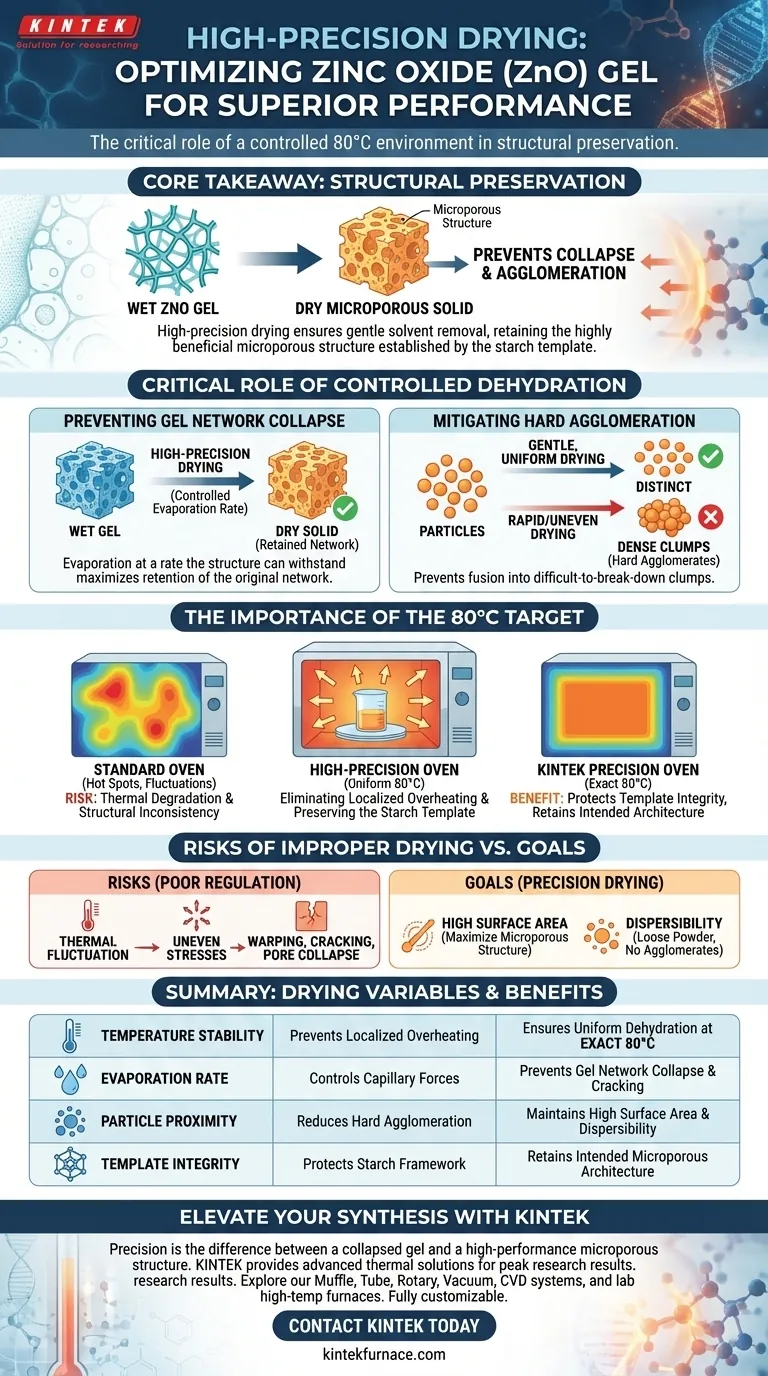
The use of a high-precision electric constant temperature drying oven is critical for ensuring the gentle and uniform removal of solvents and moisture from the Zinc Oxide (ZnO) gel. By maintaining a strictly controlled dehydration environment at 80°C, this process directly influences the physical architecture of the final material.
Core Takeaway The primary value of precision drying is structural preservation. It prevents the collapse of the gel network and the formation of hard agglomerates, ensuring the final ZnO product retains the highly beneficial microporous structure established by the starch template.

The Critical Role of Controlled Dehydration
Preventing Gel Network Collapse
The transition from a wet gel to a dry solid is a mechanically stressful phase for the material. If solvents are removed too aggressively, the internal capillary forces can crush the delicate framework.
High-precision drying ensures that evaporation occurs at a rate the structure can withstand. This maximizes the retention of the gel's original network.
Mitigating Hard Agglomeration
When drying is uneven or too rapid, particles are often forced together into dense clumps known as hard agglomerates.
These agglomerates are difficult to break down and can negatively impact the surface area of the final product. A gentle, controlled process keeps the particles distinct and prevents this fusion.
The Importance of the 80°C Target
Eliminating Localized Overheating
Standard drying equipment often suffers from "hot spots," where temperatures spike in specific areas of the chamber.
A high-precision oven eliminates this variable. It ensures that every part of the gel sample is exposed to exactly 80°C, guaranteeing that no portion of the sample is thermally degraded while others are still wet.
Preserving the Starch Template
In this specific synthesis method, a starch template is used to create a microporous structure. The effectiveness of this template relies heavily on the drying step.
By strictly controlling the temperature, the oven protects the integrity of this template. This results in a final ZnO product that successfully retains the intended microporous architecture.
Understanding the Risks of Improper Drying
The Cost of Thermal Fluctuation
It is important to recognize that "drying" is not merely about removing water; it is about how that water is removed.
If you utilize equipment with poor thermal regulation, you risk structural inconsistency. Fluctuations in temperature lead to uneven drying stresses, which causes warping, cracking, or partial collapse of the pores you worked to create.
Making the Right Choice for Your Goal
The drying step is not a passive waiting period; it is an active processing step that defines the final quality of your Zinc Oxide.
- If your primary focus is High Surface Area: Adhere strictly to the 80°C limit to prevent pore collapse and maximize the microporous structure.
- If your primary focus is Dispersibility: Use precision drying to avoid hard agglomeration, ensuring the powder remains loose and easy to process later.
Ultimately, the high-precision oven serves as a preservation tool, locking in the desirable structural properties created during the synthesis phase.
Summary Table:
| Drying Variable | Impact on ZnO Quality | Benefit of High-Precision Oven |
|---|---|---|
| Temperature Stability | Prevents localized overheating | Ensures uniform dehydration at exact 80°C |
| Evaporation Rate | Controls capillary forces | Prevents gel network collapse and cracking |
| Particle Proximity | Reduces hard agglomeration | Maintains high surface area and dispersibility |
| Template Integrity | Protects starch framework | Retains the intended microporous architecture |
Elevate Your Material Synthesis with KINTEK
Precision is the difference between a collapsed gel and a high-performance microporous structure. KINTEK provides the advanced thermal solutions necessary to ensure your research achieves peak results. Backed by expert R&D and manufacturing, we offer a comprehensive range of Muffle, Tube, Rotary, Vacuum, and CVD systems, as well as specialized lab high-temp furnaces—all fully customizable to meet your unique drying and calcination needs.
Don't let thermal fluctuations compromise your final product. Contact KINTEK today to discover how our high-precision equipment can preserve the integrity of your materials and streamline your laboratory workflow.
Visual Guide
References
- Roumaissa Djafarou, Tarek Hidouri. Starch-Assisted Eco-Friendly Synthesis of ZnO Nanoparticles: Enhanced Photocatalytic, Supercapacitive, and UV-Driven Antioxidant Properties with Low Cytotoxic Effects. DOI: 10.3390/ijms26020859
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Dental Porcelain Zirconia Sintering Ceramic Vacuum Press Furnace
- Electric Rotary Kiln Pyrolysis Furnace Plant Machine Small Rotary Kiln Calciner
- High Temperature Muffle Oven Furnace for Laboratory Debinding and Pre Sintering
- Chairside Dental Porcelain Zirconia Sintering Furnace with Transformer for Ceramic Restorations
- Electric Rotary Kiln Continuous Working Small Rotary Furnace Kiln for Pyrolysis Plant Heating
People Also Ask
- What precautions should be taken when sintering zirconia in the Fast Slow Dental Sintering Furnace? Master Key Steps for Perfect Results
- What is the importance of ramp rates in dental sintering furnaces? Ensure Zirconia Restoration Quality and Durability
- What are the consequences of inconsistent firing quality in dental sintering furnaces? Avoid Weak, Flawed Restorations
- What is the purpose of dental sintering furnaces? Transform Zirconia into Durable, High-Quality Dental Restorations
- How do conventional vs rapid sintering in dental furnaces affect zirconia? Optimize Efficiency Without Sacrificing Strength



















