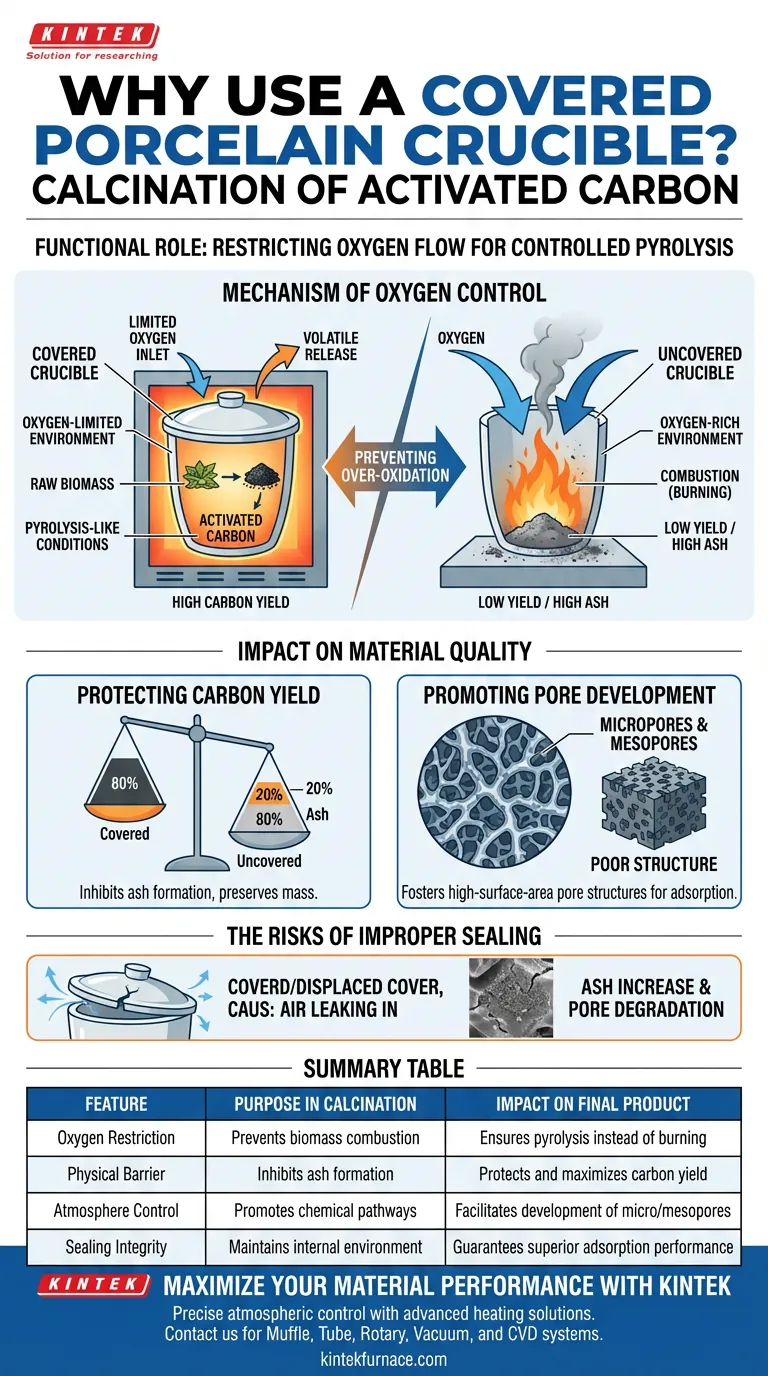
The use of a covered porcelain crucible is strictly functional: it serves as a physical barrier to restrict oxygen flow to the raw biomass during the heating process. By creating an oxygen-limited environment, the cover prevents the material from combusting completely, ensuring it undergoes a process similar to pyrolysis rather than simple burning.
By effectively limiting air intake, the cover creates a controlled environment that inhibits excessive ash production, protecting the carbon yield and facilitating the development of critical pore structures.

The Mechanism of Oxygen Control
Creating Pyrolysis-Like Conditions
The primary role of the cover is to simulate a pyrolysis environment within the crucible.
Without this barrier, the high temperatures of calcination would invite an influx of ambient air. The cover restricts this interaction, maintaining an atmosphere where thermal decomposition occurs in the absence of abundant oxygen.
Preventing Over-Oxidation
The greatest risk during calcination is over-oxidation.
If oxygen levels inside the crucible are unchecked, the biomass does not just carbonize; it burns. This reaction converts valuable organic material into useless ash, destroying the potential for activated carbon.
Impact on Material Quality
Protecting Carbon Yield
The economic and practical efficiency of the process depends on the yield of carbon.
By inhibiting the formation of ash caused by over-oxidation, the covered crucible ensures a higher percentage of the raw material is converted into usable carbon. This directly preserves the mass of the final product.
Promoting Pore Development
The physical structure of activated carbon is defined by its surface area and porosity.
The oxygen-limited environment fosters the specific chemical pathways required to form microporous and mesoporous structures. These pores are the active sites responsible for the material's adsorption capabilities.
The Risks of Improper Sealing
The Consequence of Air Leaks
While the crucible does not need to be hermetically sealed, a significant breach in the "physical barrier" defeats the purpose of the process.
If the cover is displaced or too loose, the internal environment shifts back toward combustion. This leads to a rapid increase in ash content and a degradation of the pore structure, rendering the activated carbon less effective for filtration or adsorption tasks.
Optimizing Your Calcination Strategy
To ensure you achieve the desired material properties, align your process with your specific objectives:
- If your primary focus is maximizing yield: Ensure the crucible cover is securely placed to minimize mass loss due to ash formation.
- If your primary focus is adsorption performance: strictly maintain the oxygen-limited environment to favor the development of high-surface-area micropores and mesopores.
Control the oxygen, and you control the quality of the carbon.
Summary Table:
| Feature | Purpose in Calcination | Impact on Final Product |
|---|---|---|
| Oxygen Restriction | Prevents biomass combustion | Ensures pyrolysis instead of burning |
| Physical Barrier | Inhibits ash formation | Protects and maximizes carbon yield |
| Atmosphere Control | Promotes chemical pathways | Facilitates development of micro/mesopores |
| Sealing Integrity | Maintains internal environment | Guarantees superior adsorption performance |
Maximize Your Material Performance with KINTEK
Precise atmospheric control is the difference between high-quality activated carbon and useless ash. KINTEK provides the advanced heating solutions you need to master your calcination process. Backed by expert R&D and manufacturing, we offer a comprehensive range of Muffle, Tube, Rotary, Vacuum, and CVD systems, as well as other lab high-temperature furnaces—all fully customizable to meet your unique research or production needs.
Don't let improper oxygen control compromise your yield. Contact KINTEK today to discover how our high-precision equipment can enhance your laboratory’s efficiency and material quality!
Visual Guide
References
- Dzilal Amir, Nurul Sakinah Engliman. Investigating the synthesis parameters of durian skin-based activated carbon and the effects of silver nanocatalysts on its recyclability in methylene blue removal. DOI: 10.1186/s11671-024-03974-1
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- High Temperature Muffle Oven Furnace for Laboratory Debinding and Pre Sintering
- Vacuum Dental Porcelain Sintering Furnace for Dental Laboratories
- Dental Porcelain Zirconia Sintering Ceramic Vacuum Press Furnace
- Chairside Dental Porcelain Zirconia Sintering Furnace with Transformer for Ceramic Restorations
- 1400℃ Muffle Oven Furnace for Laboratory
People Also Ask
- Why is a two-stage sintering process used for porous LATP? Master Structural Integrity and Porosity
- How does precise temperature control affect MoS2/rGO hybrids? Mastering Nanowall Morphology
- What role does a high-temperature muffle furnace play in TiO2 and PEN cross-linking? Unlock High-Performance Hybrids
- What is the function of a laboratory high-temperature muffle furnace in niobate phosphor synthesis?
- How is a muffle furnace utilized during the high-temperature annealing of forged TiAl-SiC composites?



















