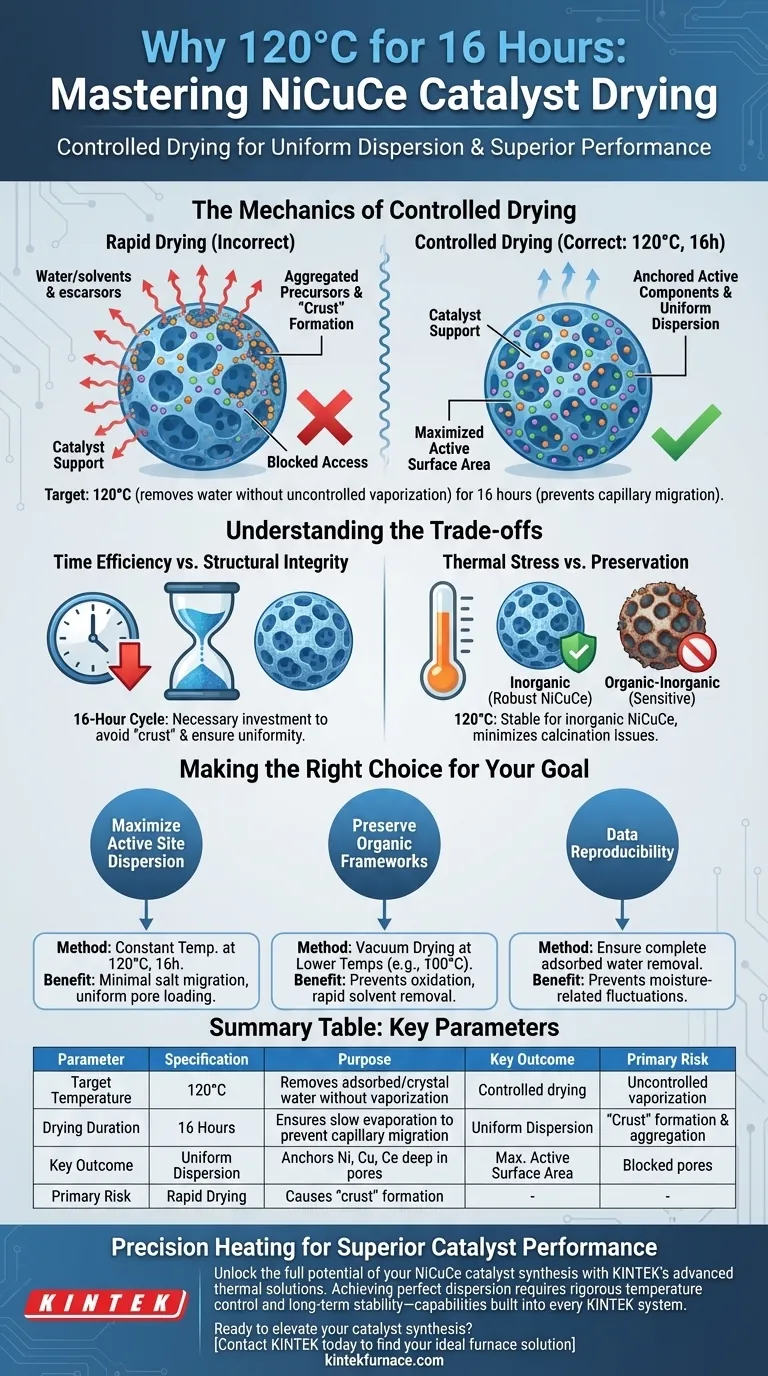
The application of a constant temperature drying oven at 120°C for 16 hours is a strategic processing step designed to achieve a controlled, gradual release of moisture from NiCuCe catalyst precursors. This specific thermal profile is chosen not merely to dry the material, but to strictly regulate the physical movement of metal salts within the support structure during the transition from liquid to solid.
By enforcing slow solvent evaporation, this method prevents the capillary migration of metal precursors, ensuring the active components remain uniformly distributed rather than aggregating at the pore openings.

The Mechanics of Controlled Drying
Removal of Specific Water Types
The target temperature of 120°C is precise. It is sufficiently high to drive off physically adsorbed water and a portion of the crystal water associated with the metal salts.
However, it is low enough to prevent the rapid, uncontrolled vaporization that occurs at higher combustion temperatures.
Managing Precursor Migration
As solvents evaporate from a porous support, there is a natural tendency for dissolved metal salts to migrate outward with the liquid.
If evaporation occurs too quickly, these salts accumulate at the pore mouths or the exterior surface.
The 16-hour duration at a constant temperature ensures the evaporation rate remains slow and steady, effectively anchoring the salts in their intended locations deep within the pores.
Ensuring Active Component Dispersion
The ultimate goal of this prolonged heating is uniformity.
By preventing uneven precipitation, the synthesis ensures that the Nickel, Copper, and Cerium components are chemically distinct and well-dispersed.
This uniform distribution is critical for maximizing the active surface area available for catalytic reactions later on.
Understanding the Trade-offs
Time Efficiency vs. Structural Integrity
The primary drawback of this method is the time investment. A 16-hour cycle represents a significant bottleneck in the production timeline compared to rapid drying techniques.
However, this time is a necessary investment to avoid the "crust" formation associated with fast drying, which renders the inner catalyst volume inaccessible.
Thermal Stress vs. Preservation
While effective for inorganic salts, this method exposes the material to sustained heat.
In scenarios involving organic-inorganic hybrids, as noted in vacuum drying comparisons, this temperature and duration could lead to oxidation or structural deterioration.
However, for robust inorganic NiCuCe systems, this thermal stability helps minimize endothermic interference during subsequent high-temperature calcination.
Making the Right Choice for Your Goal
When designing your catalyst synthesis protocol, select your drying method based on the physical properties required of the final material.
- If your primary focus is maximizing active site dispersion: Adhere to the constant temperature method at 120°C to minimize salt migration and ensure uniform pore loading.
- If your primary focus is preserving temperature-sensitive organic frameworks: Consider vacuum drying at lower temperatures (e.g., 100°C) to prevent oxidation while removing solvents rapidly.
- If your primary focus is data reproducibility: Ensure all physically adsorbed water is removed during this stage to prevent moisture-related fluctuations during high-temperature testing.
Precision in the drying phase is the invisible foundation of a high-performance catalyst.
Summary Table:
| Parameter | Specification | Purpose in Catalyst Preparation |
|---|---|---|
| Target Temperature | 120°C | Removes adsorbed and crystal water without uncontrolled vaporization. |
| Drying Duration | 16 Hours | Ensures slow evaporation to prevent capillary migration of metal salts. |
| Key Outcome | Uniform Dispersion | Anchors Ni, Cu, and Ce deep within pores for maximum active surface area. |
| Primary Risk | Rapid Drying | Causes "crust" formation and aggregation at pore openings. |
Precision Heating for Superior Catalyst Performance
Unlock the full potential of your NiCuCe catalyst synthesis with KINTEK’s advanced thermal solutions. Achieving the perfect dispersion of active components requires rigorous temperature control and long-term stability—capabilities built into every KINTEK system.
Backed by expert R&D and world-class manufacturing, KINTEK offers a comprehensive range of Muffle, Tube, Rotary, Vacuum, and CVD systems, as well as specialized high-temperature lab ovens. Our equipment is fully customizable to meet the unique needs of your research or industrial production, ensuring repeatable results and high-performance material outcomes.
Ready to elevate your catalyst synthesis? Contact KINTEK today to find your ideal furnace solution.
Visual Guide
References
- Yankun Jiang, Siqi Li. Sustainable Hydrogen from Methanol: NiCuCe Catalyst Design with CO2-Driven Regeneration for Carbon-Neutral Energy Systems. DOI: 10.3390/catal15050478
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 1700℃ High Temperature Muffle Oven Furnace for Laboratory
- 1200℃ Muffle Oven Furnace for Laboratory
- 1400℃ Muffle Oven Furnace for Laboratory
- 1800℃ High Temperature Muffle Oven Furnace for Laboratory
- High Temperature Muffle Oven Furnace for Laboratory Debinding and Pre Sintering
People Also Ask
- What role does a muffle furnace play in BCZY712 electrolyte powder production? Achieve Perfect Phase Purity
- Why is a high-temperature muffle furnace typically selected for annealing? Achieve Optimal Ceramic Performance
- What is the function of a muffle furnace in NiFe2O4/biochar prep? Optimize Your Composite Synthesis
- What role does a high-temperature muffle furnace play in the electrodeposition of high-purity iron? Achieve Precision
- What is the importance of programmable temperature control in a muffle furnace? Master g-C3N4 Synthesis Precision






