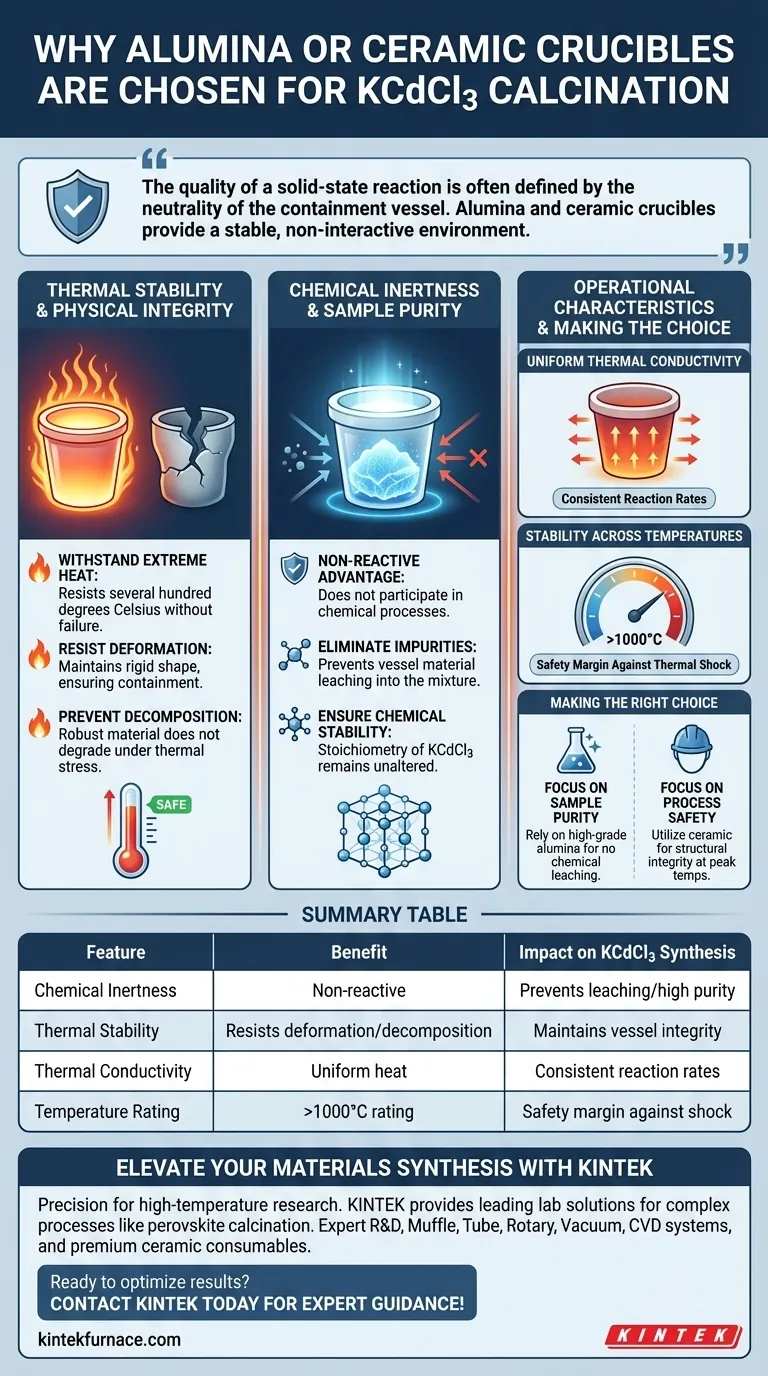
Alumina or ceramic crucibles are chosen primarily for their exceptional chemical inertness and thermal stability. When synthesizing KCdCl3 perovskite, these vessels withstand high calcination temperatures without deforming or decomposing. Crucially, they remain non-reactive, preventing the vessel material from leaching into the compound and ensuring the final polycrystalline solid remains pure.
The quality of a solid-state reaction is often defined by the neutrality of the containment vessel. Alumina and ceramic crucibles provide a stable, non-interactive environment that protects the structural integrity and chemical purity of KCdCl3 during rigorous heat treatments.
Thermal Stability and Physical Integrity
Withstanding Extreme Heat
The calcination of KCdCl3 requires sustained exposure to high temperatures. Alumina and ceramic crucibles are capable of withstanding these conditions—often reaching several hundred degrees Celsius—without failure.
Resistance to Deformation
At these elevated temperatures, lesser materials might soften or warp. Ceramic crucibles maintain their rigid shape, ensuring the reaction remains safely contained throughout the heating cycle.
Preventing Decomposition
Unlike some reaction vessels that may break down under thermal stress, alumina remains physically robust. It does not undergo chemical decomposition, which ensures the vessel itself does not degrade during the process.
Chemical Inertness and Sample Purity
The Non-Reactive Advantage
The primary danger in high-temperature synthesis is unintended reactions between the vessel and the sample. Alumina crucibles possess a non-reactive nature, meaning they do not participate in the chemical process.
Eliminating Impurities
Because the crucible resists chemical attack, it prevents foreign elements from leaching into the KCdCl3 mixture. This is vital for synthesizing a high-quality polycrystalline solid free of external contaminants.
Ensuring Chemical Stability
The inertness of the crucible guarantees that the stoichiometry of the KCdCl3 remains unaltered. The final product reflects only the intended reagents, not byproducts of the container.
Understanding Operational Characteristics
Uniform Thermal Conductivity
Beyond simple resistance, ceramic materials offer beneficial thermal properties. They facilitate the uniform heating of the powder inside, which is critical for consistent reaction rates throughout the sample.
Stability Across Temperature Ranges
While primarily selected for KCdCl3 calcination (typically several hundred degrees), these crucibles are often rated for temperatures exceeding 1000°C. This provides a significant safety margin against thermal shock or accidental overheating.
Making the Right Choice for Your Goal
When setting up your calcination process for KCdCl3, consider these factors:
- If your primary focus is Sample Purity: Rely on high-grade alumina to ensure absolutely no chemical leaching introduces impurities into your polycrystalline solid.
- If your primary focus is Process Safety: Utilize ceramic crucibles to ensure the vessel maintains its shape and structural integrity without deformation at peak temperatures.
By selecting the proper crucible, you transform the reaction vessel from a potential variable into a reliable constant.
Summary Table:
| Feature | Alumina/Ceramic Crucible Benefit | Impact on KCdCl3 Synthesis |
|---|---|---|
| Chemical Inertness | Non-reactive with reagents | Prevents leaching and ensures high sample purity |
| Thermal Stability | Resists deformation and decomposition | Maintains vessel integrity at high calcination temps |
| Thermal Conductivity | Facilitates uniform heat distribution | Ensures consistent reaction rates throughout the solid |
| Temperature Rating | Often rated >1000°C | Provides safety margin against thermal shock or overheating |
Elevate Your Materials Synthesis with KINTEK
Precision in high-temperature research begins with the right environment. KINTEK provides industry-leading lab solutions tailored for complex processes like perovskite calcination. Backed by expert R&D and manufacturing, we offer a comprehensive range of Muffle, Tube, Rotary, Vacuum, and CVD systems, alongside premium ceramic consumables. Whether you need high-purity crucibles or fully customizable high-temperature furnaces, our team is dedicated to ensuring your polycrystalline solids meet the highest standards of chemical integrity.
Ready to optimize your heat treatment results? Contact KINTEK today for expert guidance and custom solutions!
Visual Guide
References
- Md. Sunjid Sorker, Md. Abdur Razzak Sarker. First-principles and experimental study to investigate structural, elastic, electronic, thermal, and optical properties of KCdCl3 metal halide perovskite crystals. DOI: 10.1063/5.0206191
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 1400℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- 1700℃ High Temperature Laboratory Tube Furnace with Alumina Tube
People Also Ask
- How do high-temperature laboratory tube furnaces ensure environmental stability? Precision Thermal Reduction Tips
- How does a high-temperature tube furnace facilitate sulfur melt-diffusion? Precision Heating for PCFC/S Cathodes
- Why is a high-temperature tube furnace required for NiWO4 calcination? Achieving High-Performance Cathode Materials
- What is the mechanism of a high-temperature furnace in Bi-2223 sintering? Achieve Precision Phase Transformation
- What is a high temperature tube furnace? Achieve Precise Heat and Atmosphere Control

