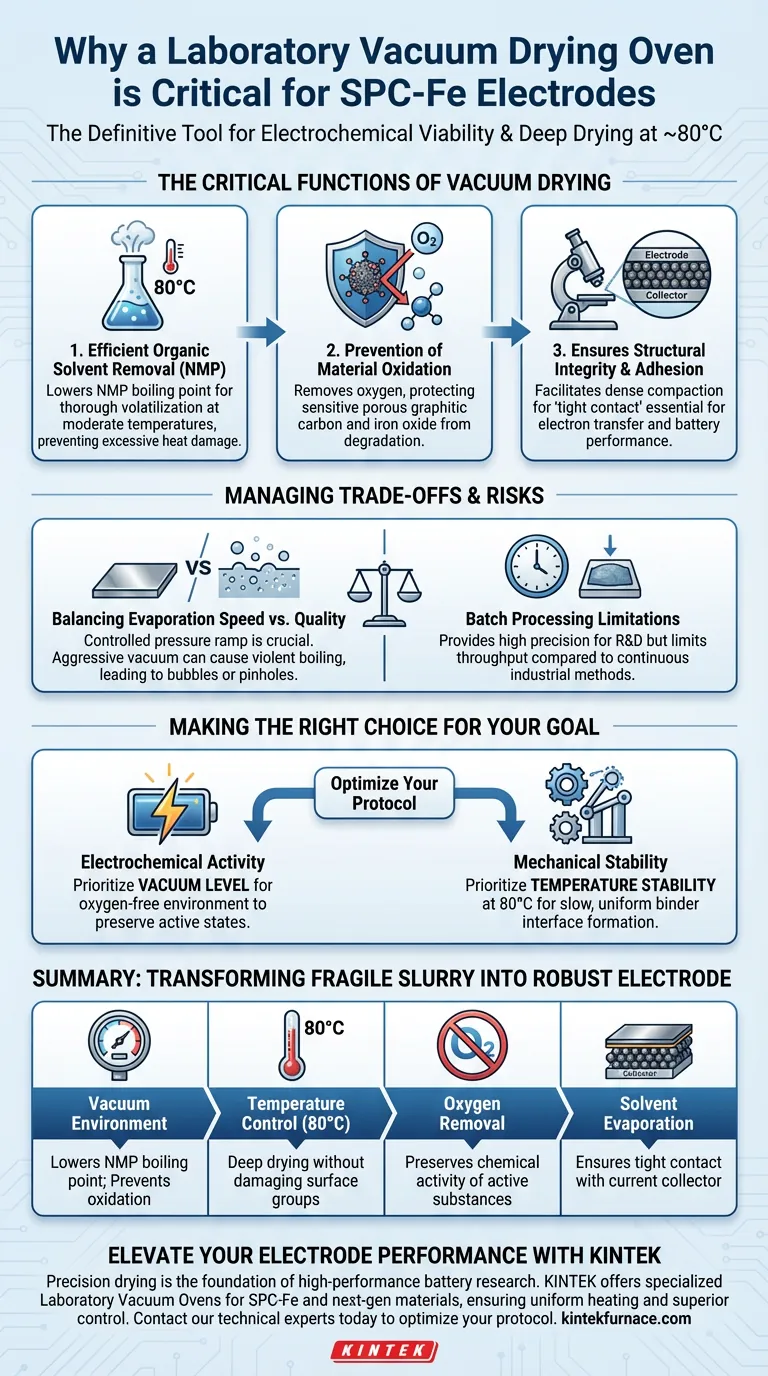
A laboratory vacuum drying oven is the definitive tool for ensuring the electrochemical viability of semi-coke based porous graphitic carbon–iron oxide (SPC-Fe) electrodes. This equipment is strictly necessary to perform deep drying of the electrode slurry at a controlled temperature of approximately 80°C. By operating under reduced pressure, it enables the complete removal of organic solvents like N-methylpyrrolidone (NMP) while simultaneously preventing the oxidation of active iron and carbon components.
Core Takeaway: The vacuum environment fundamentally alters the evaporation dynamics of the slurry, allowing for the complete removal of high-boiling-point solvents at moderate temperatures. This process preserves the chemical activity of the SPC-Fe composite and ensures the mechanical integrity of the electrode layer.
The Critical Functions of Vacuum Drying
Efficient Removal of Organic Solvents
The primary challenge in preparing SPC-Fe electrodes is removing the binder solvent, typically N-methylpyrrolidone (NMP).
NMP has a high boiling point and is difficult to evaporate fully at standard atmospheric pressure without excessive heat.
A vacuum oven lowers the boiling point of NMP, allowing it to volatilize thoroughly at a moderate 80°C.
Prevention of Material Oxidation
Both porous graphitic carbon and iron oxide are sensitive to oxidation, particularly when heated.
Exposing these materials to high temperatures in an air-filled environment can degrade their surface functional groups and alter their chemical composition.
The vacuum environment removes oxygen from the chamber, protecting the active substances from degradation during the drying phase.
Ensuring Structural Integrity and Adhesion
To function correctly, the electrode material must maintain a robust physical connection with the current collector.
The "deep drying" process facilitated by the vacuum ensures that as the solvent leaves, the remaining SPC-Fe powder and binder compact densely.
This results in tight contact between the electrode layer and the current collector, which is essential for efficient electron transfer and long-term battery performance.
Understanding the Trade-offs and Risks
Balancing Evaporation Speed and Coating Quality
While vacuum drying lowers the boiling point, applying a vacuum too aggressively can cause the solvent to boil violently.
This rapid evaporation can lead to the formation of bubbles or pinholes in the electrode surface, compromising its uniformity.
Operators must carefully control the pressure ramp to ensure solvent removal is steady rather than explosive.
Batch Processing Limitations
Unlike continuous conveyor-belt drying used in large-scale manufacturing, laboratory vacuum ovens are typically batch-process tools.
This limits throughput and requires distinct heating and cooling cycles for each batch of electrodes.
While this ensures high precision for research and development, it represents a bottleneck in terms of production speed compared to industrial methods.
Making the Right Choice for Your Goal
To maximize the performance of your SPC-Fe electrodes, align your drying protocol with your specific research objectives:
- If your primary focus is Electrochemical Activity: Prioritize the vacuum level to ensure a completely oxygen-free environment, preserving the oxidation state of the iron and the surface groups of the carbon.
- If your primary focus is Mechanical Stability: Prioritize temperature stability at 80°C, ensuring the solvent is removed slowly enough to allow the binder to form a uniform, tight interface with the current collector.
By controlling the atmosphere and temperature simultaneously, you transform a fragile slurry into a robust, high-performance electrode.
Summary Table:
| Feature | Benefit for SPC-Fe Electrodes |
|---|---|
| Vacuum Environment | Lowers NMP boiling point; prevents oxidation of carbon and iron |
| Temperature Control (80°C) | Facilitates deep drying without damaging sensitive surface groups |
| Oxygen Removal | Preserves the chemical activity and composition of active substances |
| Solvent Evaporation | Ensures tight contact between electrode layer and current collector |
Elevate Your Electrode Performance with KINTEK
Precision drying is the foundation of high-performance battery research. Backed by expert R&D and world-class manufacturing, KINTEK offers specialized Laboratory Vacuum Ovens and high-temp systems designed to meet the rigorous demands of composite material processing. Whether you are working with SPC-Fe or next-generation energy storage, our customizable solutions ensure uniform heating and superior vacuum control to protect your active materials from oxidation.
Ready to optimize your laboratory drying protocol? Contact our technical experts today to find the perfect system for your unique research needs.
Visual Guide
References
- Preparation of Semi‐Coke‐Based Porous Graphitic Carbon–Iron Oxide Composites and Their Electrochemical Performance for Rhodamine B Degradation. DOI: 10.1155/er/9943954
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Vacuum Heat Treat Sintering and Brazing Furnace
- Vacuum Heat Treat Sintering Furnace Molybdenum Wire Vacuum Sintering Furnace
- Dental Porcelain Zirconia Sintering Ceramic Vacuum Press Furnace
- Molybdenum Vacuum Heat Treat Furnace
- Vacuum Hot Press Furnace Machine Heated Vacuum Press
People Also Ask
- How does vacuum heat treating affect the grain structure of metal alloys? Achieve Precise Microstructure Control
- What are the steps involved in a typical vacuum brazing treatment? Master the Process for Strong, Clean Joints
- What is the role of high-precision furnaces in Inconel 718 heat treatment? Master Microstructural Engineering
- What is one of the most important uses of vacuum heat treating furnaces in aerospace? Achieve Superior Strength in Aircraft Alloys
- What is the function of industrial vacuum heat treatment furnaces? Elevate 3D-Printed Maraging Steel Quality












