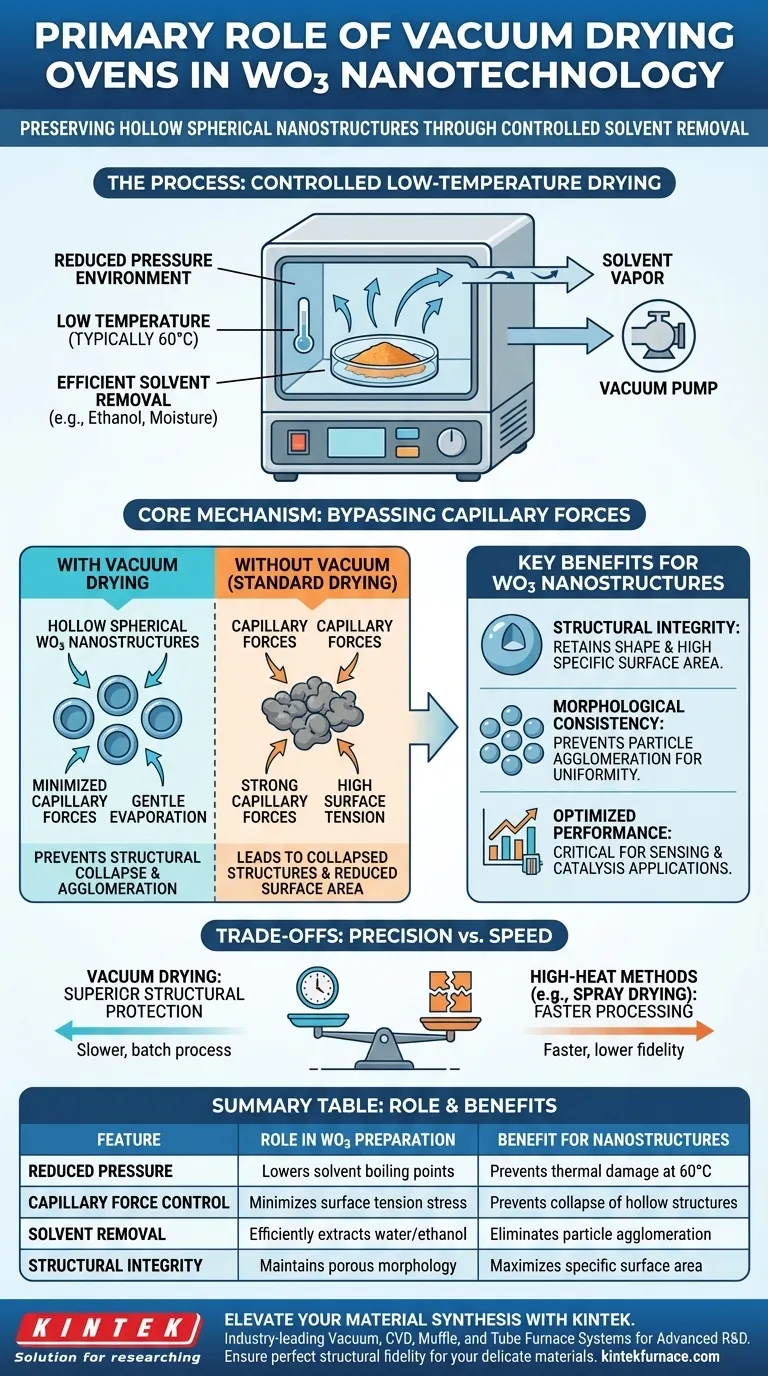
The primary role of a vacuum drying oven in this context is to remove solvents, such as moisture and ethanol, from WO3 precursors at a controlled low temperature, typically 60 degrees Celsius. By creating a reduced pressure environment, the oven enables evaporation without high heat, protecting the delicate nanostructures from thermal damage or physical stress.
Core Takeaway: The vacuum drying process is essential for bypassing the destructive effects of capillary forces during dehydration. By lowering the pressure, you ensure the hollow spherical nanostructures retain their shape and high specific surface area rather than collapsing or clumping together.

The Mechanics of Structural Preservation
Minimizing Capillary Forces
When liquid evaporates from a porous material under standard atmospheric pressure, surface tension creates strong capillary forces.
These forces pull the pore walls together, often leading to the collapse of fragile hollow structures. Vacuum drying significantly reduces these forces, allowing the solvent to exit without crushing the nanostructure.
Low-Temperature Evaporation
The vacuum environment lowers the boiling point of solvents like ethanol and water.
This allows for rapid evaporation at much lower temperatures, such as the referenced 60 degrees Celsius. This "gentle" drying is critical because high heat can alter the chemical phase or morphology of the precursor before it is stabilized.
Optimizing Material Morphology
Preventing Agglomeration
In nanostructure synthesis, particles have a natural tendency to clump together, or agglomerate.
Vacuum drying prevents the particles from adhering to one another during the drying phase. This isolation is vital for maintaining distinct, individual spheres rather than a fused mass.
Preserving Specific Surface Area
The performance of WO3 in applications (like sensing or catalysis) depends heavily on its specific surface area.
By preventing structural collapse and pore blockage, vacuum drying maximizes the available surface area. It ensures the final material maintains the porous morphology required for high performance.
Understanding the Trade-offs
Process Speed vs. Structural Fidelity
While vacuum drying provides superior structural protection, it is generally a slower, batch-oriented process compared to high-heat techniques like spray drying.
You trade processing speed for structural integrity. For high-precision nanostructures, this trade-off is usually necessary, but it may represent a bottleneck in large-scale manufacturing.
Equipment Complexity
Vacuum drying requires maintaining a sealed system and operating vacuum pumps.
This introduces more complexity and maintenance requirements than a standard convection oven. However, without this equipment, achieving the requisite hollow morphology is often impossible.
Making the Right Choice for Your Goal
To maximize the quality of your WO3 nanostructures, apply the following guidelines:
- If your primary focus is maximizing surface area: Rely on vacuum drying to prevent capillary collapse and ensure the pores remain open and accessible.
- If your primary focus is morphological consistency: Use the vacuum method to prevent particle agglomeration, ensuring uniform hollow spheres rather than irregular clumps.
The vacuum drying stage is not merely a dehydration step; it is a structural preservation technique that defines the final quality of the nanomaterial.
Summary Table:
| Feature | Role in WO3 Preparation | Benefit for Nanostructures |
|---|---|---|
| Reduced Pressure | Lowers solvent boiling points | Prevents thermal damage at 60°C |
| Capillary Force Control | Minimizes surface tension stress | Prevents collapse of hollow structures |
| Solvent Removal | Efficiently extracts water/ethanol | Eliminates particle agglomeration |
| Structural Integrity | Maintains porous morphology | Maximizes specific surface area |
Elevate Your Material Synthesis with KINTEK
Precision in nanostructure preparation requires specialized thermal equipment. KINTEK provides industry-leading Vacuum, CVD, Muffle, and Tube furnace systems designed to meet the rigorous demands of advanced R&D. Our customizable high-temperature lab solutions ensure you maintain perfect structural fidelity and surface area for your most delicate materials.
Ready to optimize your drying and sintering processes? Contact our experts today to discover how KINTEK’s manufacturing excellence can support your unique laboratory needs.
Visual Guide
References
- Peishuo Wang, Xueli Yang. Engineering Hierarchical CuO/WO3 Hollow Spheres with Flower-like Morphology for Ultra-Sensitive H2S Detection at ppb Level. DOI: 10.3390/chemosensors13070250
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Vacuum Heat Treat Sintering and Brazing Furnace
- Vacuum Heat Treat Sintering Furnace Molybdenum Wire Vacuum Sintering Furnace
- Dental Porcelain Zirconia Sintering Ceramic Vacuum Press Furnace
- Vacuum Hot Press Furnace Machine for Lamination and Heating
- Vacuum Hot Press Furnace Machine Heated Vacuum Press
People Also Ask
- What are the steps involved in a typical vacuum brazing treatment? Master the Process for Strong, Clean Joints
- What is the function of industrial vacuum heat treatment furnaces? Elevate 3D-Printed Maraging Steel Quality
- What is the role of high-precision furnaces in Inconel 718 heat treatment? Master Microstructural Engineering
- What are the benefits of using vacuum heat treating furnaces for metal alloys? Achieve Superior Metal Properties and Performance
- How does vacuum heat treating affect the grain structure of metal alloys? Achieve Precise Microstructure Control







