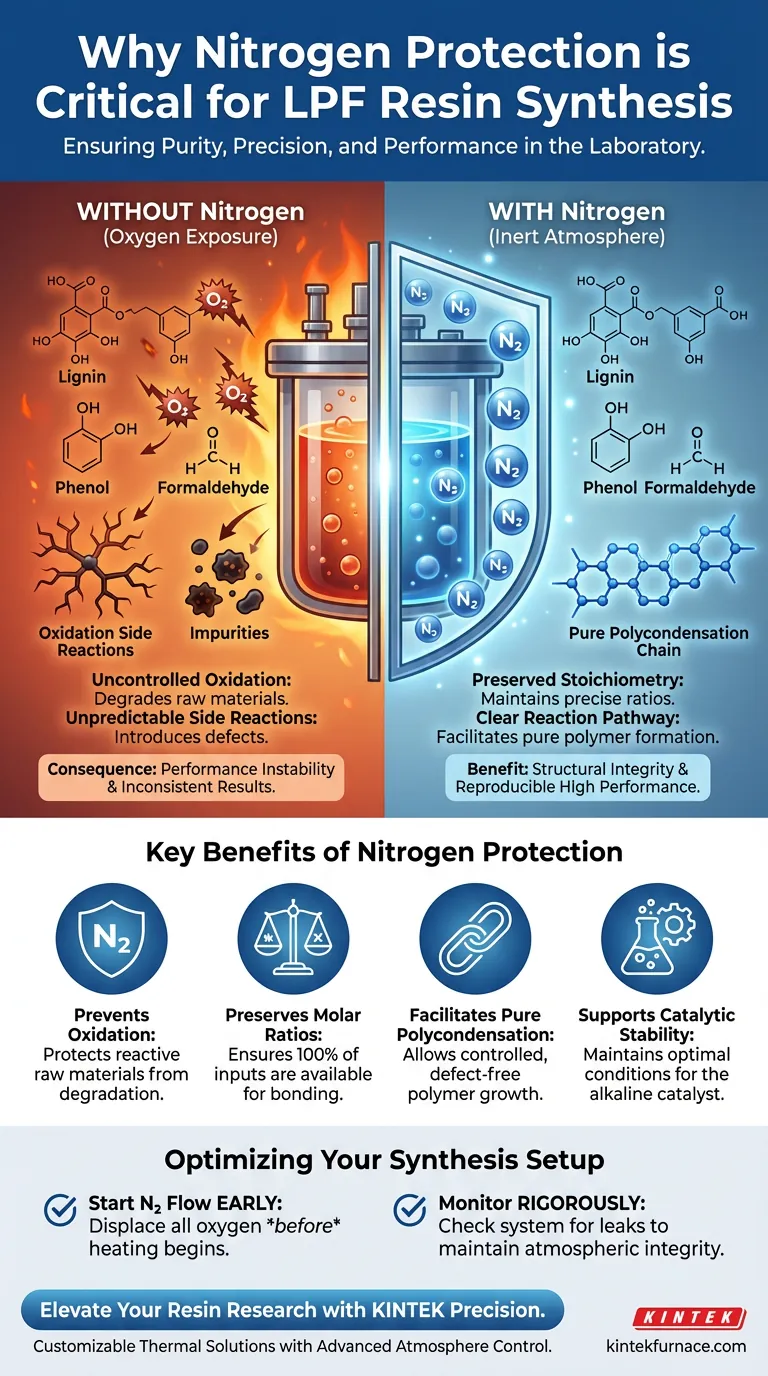
The implementation of a nitrogen protection system is critical for creating an inert environment during the synthesis of Lignin Phenol Formaldehyde (LPF) resins. By systematically excluding oxygen from the reaction vessel, nitrogen prevents the rapid, uncontrolled oxidation of raw materials that naturally occurs under the high temperatures required for synthesis.
Core Takeaway The presence of oxygen introduces chaotic variables into a precise chemical process. Nitrogen protection eliminates these variables, ensuring that oxidation does not compete with the desired polycondensation reaction, thereby preserving the structural integrity and performance of the final resin.
The Chemical Risks of Oxygen Exposure
Preventing Uncontrolled Oxidation
When synthesizing LPF resins, the reaction mixture is subjected to significant heat. Under these conditions, raw materials—specifically lignin pyrolysis products, phenol, and formaldehyde—become highly reactive with oxygen.
Without a nitrogen shield, these materials will undergo oxidation side reactions rather than the intended chemical bonding. This degrades the quality of the raw inputs before they can contribute to the polymer chain.
Preserving Stoichiometric Precision
A successful resin synthesis relies on specific molar ratios between the reactants. If a portion of the phenol or lignin oxidizes, it is effectively removed from the reaction pool.
This alters the actual molar ratio within the vessel, causing it to deviate from the calculated recipe. Nitrogen ensures that 100% of the inputs remain available for the target reaction.
Ensuring Reaction Mechanism Integrity
Facilitating Pure Polycondensation
The objective of LPF synthesis is polycondensation, where molecules link together to form complex polymer chains. This process requires a controlled pathway free from interference.
Nitrogen allows this mechanism to proceed without competition from oxidative degradation. It ensures the chemical pathways remain clear for the formation of the resin network.
Supporting Alkaline Catalytic Conditions
This synthesis typically occurs under alkaline catalytic conditions. The stability provided by an inert atmosphere is essential for the catalyst to function predictably.
By preventing the formation of oxidized byproducts, the system maintains the optimal chemical environment for the catalyst to drive the reaction forward.
The Consequences of Inadequate Protection
Performance Instability
If the reaction environment is compromised by oxygen, the resulting resin will suffer from performance instability. The final product may exhibit inconsistent curing times, strength, or thermal properties.
Unpredictable Side Reactions
The primary reference highlights that oxygen causes "unnecessary oxidation side reactions." These are not merely wasteful; they introduce impurities into the resin matrix.
These impurities can act as defects in the polymer structure, leading to a product that fails to meet laboratory or industrial specifications.
Optimizing Your Synthesis Setup
How to Apply This to Your Project
To ensure the reproducibility and quality of your LPF resins, consider the following based on your specific goals:
- If your primary focus is Chemical Consistency: Ensure the nitrogen flow is established before heating begins to displace all initial oxygen in the vessel.
- If your primary focus is Product Performance: Monitor the system for leaks rigorously, as even small amounts of oxygen can alter the effective molar ratios and degrade the final resin properties.
Control over the reaction atmosphere is just as critical as control over temperature; it is the fundamental baseline for high-quality resin synthesis.
Summary Table:
| Feature | Role in LPF Synthesis | Benefit to Research |
|---|---|---|
| Inert Atmosphere | Displaces oxygen from the reaction vessel | Prevents uncontrolled oxidation of lignin and phenol |
| Stoichiometric Control | Maintains calculated molar ratios | Ensures 100% of raw materials participate in bonding |
| Reaction Pathway | Facilitates pure polycondensation | Eliminates side reactions and structural impurities |
| Catalytic Stability | Supports alkaline catalytic conditions | Maintains a predictable chemical environment for synthesis |
Elevate Your Resin Research with KINTEK Precision
Don't let oxidative side reactions compromise your laboratory results. KINTEK provides high-performance, customizable thermal solutions—including Vacuum and Tube furnaces equipped with advanced atmosphere control systems—designed to meet the rigorous demands of LPF resin synthesis.
Backed by expert R&D and manufacturing, we offer specialized equipment to ensure your chemical pathways remain pure and your results reproducible. Contact KINTEK today to discuss your unique laboratory needs and discover how our high-temperature systems can bring superior precision to your material synthesis.
Visual Guide
References
- Johannes Karthäuser, Holger Militz. Utilizing pyrolysis cleavage products from softwood kraft lignin as a substitute for phenol in phenol-formaldehyde resins for modifying different wood species. DOI: 10.1007/s00107-024-02056-4
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- RF PECVD System Radio Frequency Plasma Enhanced Chemical Vapor Deposition
- HFCVD Machine System Equipment for Drawing Die Nano Diamond Coating
People Also Ask
- What are the advantages of PECVD in film deposition? Achieve Low-Temp, High-Quality Coatings
- How are deposition rates and film properties controlled in PECVD? Master Key Parameters for Optimal Thin Films
- What is Plasma-enhanced chemical vapor deposition (PECVD) and its applications? Unlock Low-Temperature Thin-Film Coating
- What is the role of RF power in PECVD and how does the RF-PECVD process work? Master Thin Film Deposition Control
- What are some promising applications of PECVD-prepared 2D materials? Unlock Advanced Sensing and Optoelectronics

