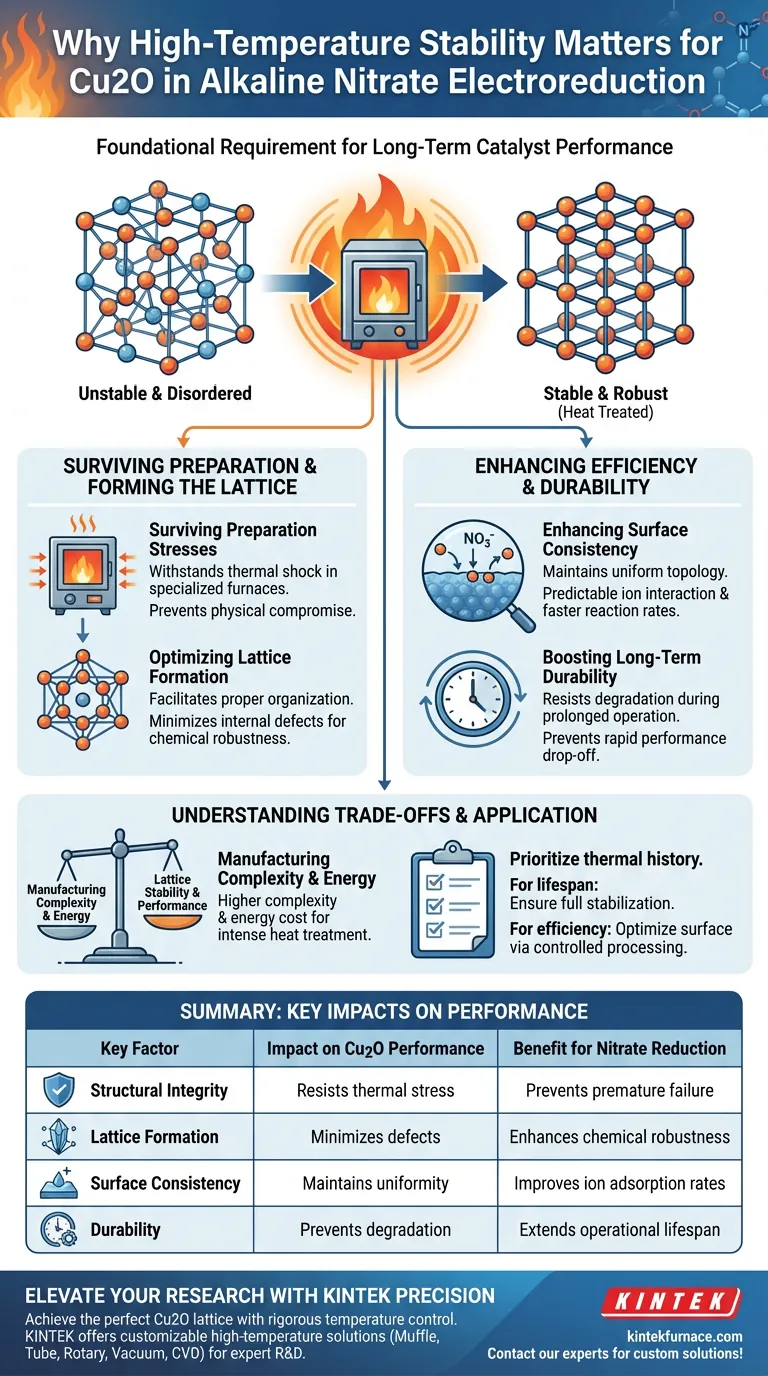
High-temperature stability is the foundational requirement for long-term catalyst performance. For cuprous oxide (Cu2O) substrates in alkaline nitrate electroreduction, this stability ensures the material survives the thermal stresses of preparation. It guarantees a well-formed crystal lattice, which is essential for maintaining structural integrity and providing a consistent surface for efficient chemical reactions.
Heat treatment determines the structural fate of the catalyst. High-temperature stability allows the Cu2O substrate to develop a robust lattice structure that resists degradation, ensuring consistent nitrate adsorption and reduction over time.
The Role of Heat in Catalyst Formation
Surviving Preparation Stresses
The manufacturing process for high-performance catalysts often requires exposure to extreme environments. The Cu2O substrate must possess innate high-temperature stability to withstand the thermal shock and stresses associated with preparation in specialized furnaces.
Without this thermal resilience, the physical structure of the substrate would likely compromise before the electrocatalytic process even begins.
Optimizing Lattice Formation
Heat treatment is not simply a durability test; it is an active formation process. High temperatures facilitate the proper organization of the Cu2O crystal lattice.
A well-formed lattice is chemically more robust. This structural organization minimizes internal defects that could otherwise act as failure points during operation.
Impact on Electroreduction Efficiency
Enhancing Surface Consistency
The efficiency of nitrate electroreduction depends heavily on how the reactants interact with the catalyst surface. High-temperature stability ensures the substrate maintains a consistent surface topology.
This uniformity is critical for the adsorption of nitrate ions. A stable, consistent surface allows for predictable and efficient ion interaction, directly influencing reaction rates.
Boosting Long-Term Durability
Electrocatalytic reactions are intense and can degrade weaker materials over time. A substrate that has been stabilized through high-temperature processing is significantly more durable.
This durability prevents the physical breakdown of the catalyst during prolonged operation, ensuring that performance does not drop off rapidly after the initial cycles.
Understanding the Trade-offs
Manufacturing Complexity
Achieving high-temperature stability requires specialized equipment, such as high-temperature furnaces. This increases the complexity of the manufacturing setup compared to room-temperature synthesis methods.
Energy Consumption
The requirement for intense heat treatment inherently raises the energy cost of production. You must balance the need for a highly stable lattice against the increased energy expenditure required to create it.
Applying This to Catalyst Development
To maximize the performance of your alkaline nitrate electroreduction system, you must prioritize the thermal history of your substrate material.
- If your primary focus is long-term operational lifespan: Ensure your synthesis process includes adequate heat treatment to fully stabilize the Cu2O lattice structure against degradation.
- If your primary focus is maximizing reaction efficiency: Verify that the substrate's surface consistency has been optimized through controlled thermal processing to aid nitrate adsorption.
A thermally stable substrate is not just a passive support; it is the active backbone of a reliable and efficient electrocatalytic system.
Summary Table:
| Key Factor | Impact on Cu2O Performance | Benefit for Nitrate Reduction |
|---|---|---|
| Structural Integrity | Resists thermal stress during preparation | Prevents premature material failure |
| Lattice Formation | Minimizes internal defects and voids | Enhances chemical robustness |
| Surface Consistency | Maintains uniform topology | Improves nitrate ion adsorption rates |
| Durability | Prevents degradation during cycling | Extends operational lifespan |
Elevate Your Catalyst Research with KINTEK Precision
High-performance electrocatalysis begins with superior thermal processing. At KINTEK, we understand that achieving the perfect Cu2O lattice requires rigorous temperature control and reliable equipment.
Backed by expert R&D and world-class manufacturing, KINTEK offers a comprehensive range of high-temperature solutions, including Muffle, Tube, Rotary, Vacuum, and CVD systems. Our furnaces are fully customizable to meet the unique thermal profiles required for your specific material synthesis, ensuring your substrates achieve maximum stability and efficiency.
Ready to optimize your high-temperature synthesis?
Contact our experts today to find your custom furnace solution!
Visual Guide
References
- Wanru Liao, Min Liu. Sustainable conversion of alkaline nitrate to ammonia at activities greater than 2 A cm−2. DOI: 10.1038/s41467-024-45534-2
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Custom Made Versatile CVD Tube Furnace Chemical Vapor Deposition CVD Equipment Machine
- Multi Heating Zones CVD Tube Furnace Machine for Chemical Vapor Deposition Equipment
- 915MHz MPCVD Diamond Machine Microwave Plasma Chemical Vapor Deposition System Reactor
- Inclined Rotary Plasma Enhanced Chemical Deposition PECVD Tube Furnace Machine
- Slide PECVD Tube Furnace with Liquid Gasifier PECVD Machine
People Also Ask
- How can integrating CVD tube furnaces with other technologies benefit device fabrication? Unlock Advanced Hybrid Processes
- How might AI and machine learning enhance CVD tube furnace processes? Boost Quality, Speed, and Safety
- What customization options are available for CVD tube furnaces? Tailor Your System for Superior Material Synthesis
- What are the practical applications of gate media prepared by CVD tube furnaces? Unlock Advanced Electronics and More
- How does CVD tube furnace sintering enhance graphene growth? Achieve Superior Crystallinity and High Electron Mobility



















