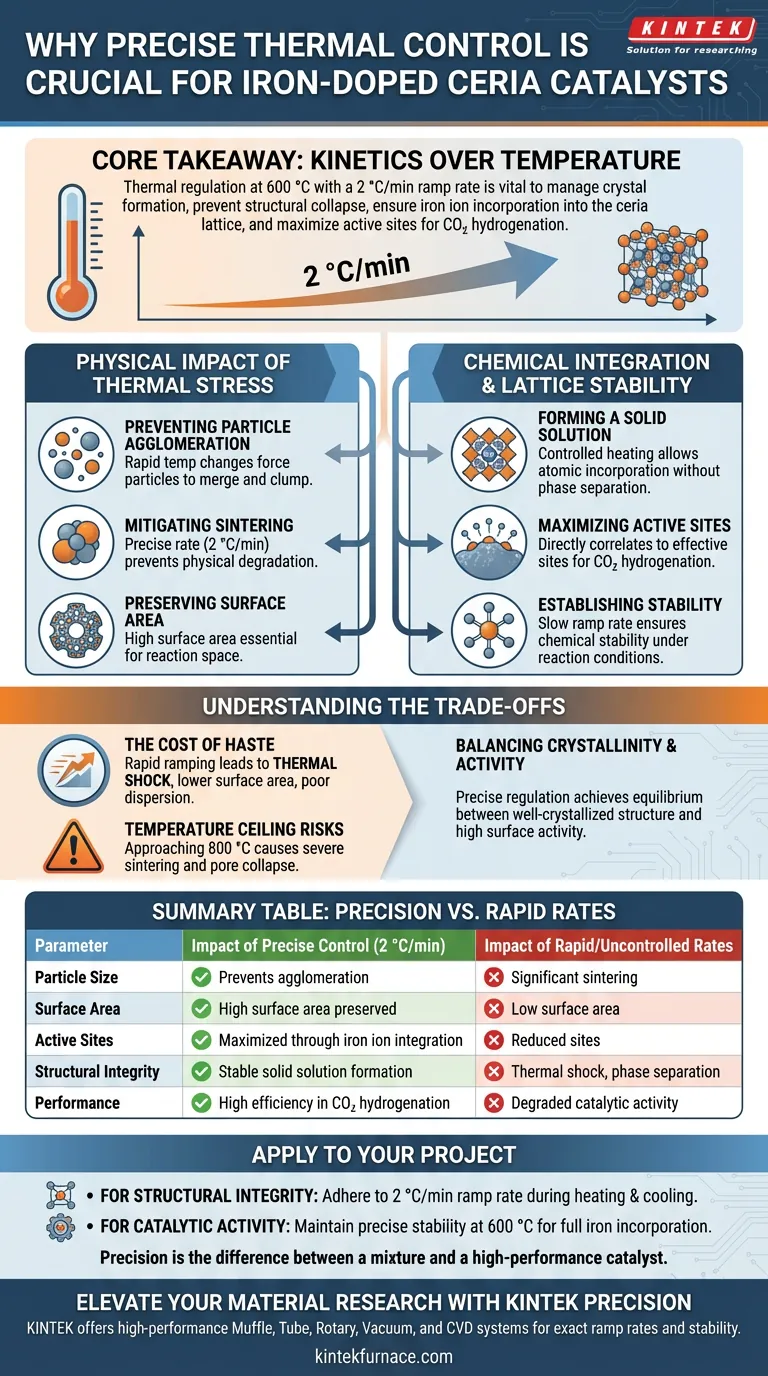
Precise control of heating and cooling rates is the defining factor in determining the structural integrity and chemical potency of iron-doped ceria catalysts. During the final calcination stage at 600 °C, utilizing a slow, governed rate—specifically 2 °C/min—is necessary to mitigate thermal stress. Without this regulation, the material is prone to excessive sintering and agglomeration, which compromises the physical architecture required for effective catalysis.
Core Takeaway Thermal regulation is not merely about reaching a target temperature; it is about managing the kinetics of crystal formation to prevent structural collapse. By strictly controlling the ramp rate, you ensure the successful incorporation of iron ions into the ceria lattice, creating a stable solid solution that maximizes active sites for carbon dioxide hydrogenation.

The Physical Impact of Thermal Stress
Preventing Particle Agglomeration
Rapid changes in temperature introduce significant thermal stress to the catalyst material. This stress forces particles to merge and clump together, a process known as agglomeration.
Mitigating Sintering
When particles agglomerate, the material undergoes sintering, effectively fusing distinct particles into larger masses. Implementing a precise heating and cooling rate, such as 2 °C/min, keeps this physical degradation in check.
Preserving Surface Area
The direct consequence of preventing sintering is the preservation of the catalyst's specific surface area. A high surface area is non-negotiable for catalytic performance, as it provides the physical space necessary for chemical reactions to occur.
Chemical Integration and Lattice Stability
Forming a Solid Solution
For iron-doped ceria to function correctly, the iron ions must be integrated into the ceria crystal structure, forming a solid solution. Controlled heating provides the necessary thermodynamic environment for this atomic incorporation to occur without inducing phase separation.
Maximizing Active Sites
The formation of this solid solution directly correlates to the number of active sites available on the catalyst. These sites are specifically tuned for carbon dioxide hydrogenation reactions, rendering the catalyst effective for its intended application.
Establishing Stability
A slow ramp rate ensures that the interaction between the iron and the ceria lattice is chemically stable. This prevents the active components from degrading or separating during subsequent use under reaction conditions.
Understanding the Trade-offs
The Cost of Haste
Accelerating the heating rate to save processing time is a common but fatal error in catalyst preparation. Rapid ramping leads to "thermal shock," which invariably results in lower surface area and poor dispersion of the active iron species.
Temperature Ceiling Risks
While the target temperature for this specific process is 600 °C, exceeding this limit can be detrimental. As seen in general catalyst preparation principles, temperatures approaching 800 °C often lead to severe sintering, pore structure collapse, and a reduction in surface oxygen vacancies.
Balancing Crystallinity and Activity
The goal of thermal treatment is to find the equilibrium between a well-crystallized structure and high surface activity. Precise temperature regulation is the only mechanism that allows you to achieve both simultaneously, rather than sacrificing one for the other.
How to Apply This to Your Project
To ensure optimal performance of your iron-doped ceria catalysts, apply the following parameters:
- If your primary focus is Structural Integrity: Strictly adhere to a ramp rate of 2 °C/min during both heating and cooling to minimize thermal stress and prevent particle agglomeration.
- If your primary focus is Catalytic Activity: maintain precise temperature stability at 600 °C to guarantee the full incorporation of iron ions into the ceria lattice for maximum hydrogenation efficiency.
Precision in thermal processing is the difference between a mixture of oxides and a high-performance catalyst.
Summary Table:
| Parameter | Impact of Precise Control (2 °C/min) | Impact of Rapid/Uncontrolled Rates |
|---|---|---|
| Particle Size | Prevents agglomeration; maintains fine particles | Significant sintering and particle clumping |
| Surface Area | High surface area preserved for reactions | Low surface area due to structural collapse |
| Active Sites | Maximized through iron ion integration | Reduced sites due to poor phase dispersion |
| Structural Integrity | Stable solid solution formation | Thermal shock and phase separation |
| Performance | High efficiency in CO2 hydrogenation | Degraded catalytic activity |
Elevate Your Material Research with KINTEK Precision
Don't let thermal stress compromise your catalyst's potential. Backed by expert R&D and manufacturing, KINTEK offers high-performance Muffle, Tube, Rotary, Vacuum, and CVD systems designed to deliver the exact ramp rates your research demands. Whether you need a standard setup or a customized lab furnace for unique doping processes, we provide the stability and control necessary to maximize your active sites.
Contact KINTEK Today for a Tailored Solution
Visual Guide
References
- Albert Gili, Reinhard Schomäcker. One-pot synthesis of iron-doped ceria catalysts for tandem carbon dioxide hydrogenation. DOI: 10.1039/d4cy00439f
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Electric Rotary Kiln Pyrolysis Furnace Plant Machine Small Rotary Kiln Calciner
- Custom Made Versatile CVD Tube Furnace Chemical Vapor Deposition CVD Equipment Machine
- Laboratory Quartz Tube Furnace RTP Heating Tubular Furnace
- Multi Heating Zones CVD Tube Furnace Machine for Chemical Vapor Deposition Equipment
- Vertical Laboratory Quartz Tube Furnace Tubular Furnace
People Also Ask
- What materials can be processed in an electric rotary kiln? Versatile Solutions for Advanced Materials
- What are the primary applications of an electric rotary kiln? Achieve High-Purity Material Processing with Precision
- What operational benefits do electric rotary kilns offer? Achieve Precision, Efficiency, and Simplified Control
- What are the benefits of smoke and ash reduction in electric rotary kilns? Achieve Cleaner, Simpler, and More Cost-Effective Processing
- What are the advantages of electric rotary kilns compared to fuel-fired ones? Boost Precision and Efficiency in Your Process







