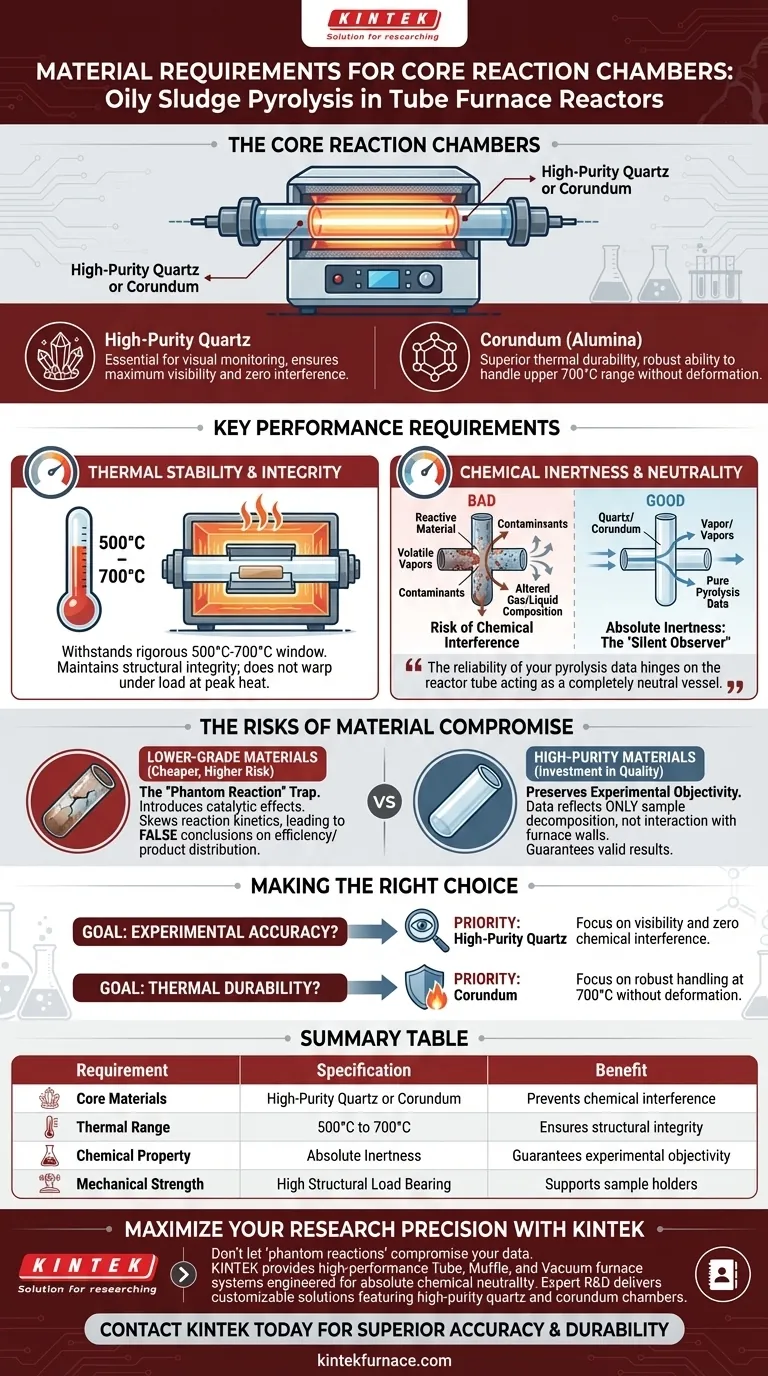
The core reaction chambers in Tube Furnace Reactors for oily sludge pyrolysis must be constructed from high-purity quartz or corundum. These materials are essential because they possess the specific thermal stability required to operate between 500°C and 700°C while maintaining the structural integrity needed to support the sample. Most importantly, they act as chemically neutral vessels, ensuring the reactor tube does not interfere with the pyrolysis process.
The reliability of your pyrolysis data hinges on the reactor tube acting as a completely neutral vessel; it must withstand intense heat without chemically interacting with the complex volatiles released by the sludge.

The Critical Role of Material Inertness
Preventing Chemical Interference
Oily sludge pyrolysis releases a complex mixture of volatile components. It is imperative that the reaction chamber material, specifically quartz or corundum, exhibits absolute chemical inertness.
If the tube material is reactive, it may interact with the pyrolysis vapors. This interaction alters the composition of the produced gases and liquids.
Maintaining Experimental Objectivity
The primary goal of using high-purity materials is to preserve the "objectivity" of your experimental results.
You need to measure the behavior of the sludge, not the behavior of the sludge reacting with the furnace wall. High-purity quartz and corundum ensure that the data collected reflects only the sample's decomposition.
Thermal Requirements and Stability
Withstanding High-Temperature Ranges
The pyrolysis of oily sludge typically occurs in a rigorous temperature window of 500°C to 700°C.
The core reaction chamber must possess exceptional thermal stability to endure these temperatures for extended periods.
Structural Integrity Under Load
Beyond just surviving the heat, the tube must physically support the sample holder and the sludge itself.
Materials like corundum and quartz retain their mechanical strength at these high temperatures, ensuring the chamber does not warp or collapse under the sample's weight.
Understanding the Risks of Material Compromise
The "Phantom Reaction" Trap
The most significant trade-off in selecting reactor materials is often between cost and purity. Lower-grade ceramics or metals might be cheaper, but they introduce the risk of catalytic effects.
If a material acts as an unintentional catalyst, it can skew reaction kinetics. This leads to false conclusions about the pyrolysis efficiency or product distribution.
Durability vs. Data Quality
While some materials may offer higher physical durability against impact, they often lack the chemical purity required for analytical pyrolysis.
Prioritizing physical toughness over chemical inertness can render an entire dataset invalid due to cross-contamination from the tube walls.
Making the Right Choice for Your Goal
Selecting the correct chamber material is about safeguarding the integrity of your chemical analysis.
- If your primary focus is experimental accuracy: Prioritize high-purity quartz to ensure maximum visibility and zero chemical interference with volatile components.
- If your primary focus is thermal durability: Choose corundum for its robust ability to handle the upper limits of the 700°C range without deformation.
Your equipment choices dictate your data quality; ensure your reactor chamber remains a silent observer, not an active participant in the chemical reaction.
Summary Table:
| Requirement | Specification | Benefit |
|---|---|---|
| Core Materials | High-purity Quartz or Corundum | Prevents chemical interference and catalytic effects |
| Thermal Range | 500°C to 700°C | Ensures structural integrity during sludge decomposition |
| Chemical Property | Absolute Inertness | Guarantees experimental data reflects only sample behavior |
| Mechanical Strength | High Structural Load Bearing | Supports sample holders without warping at peak heat |
Maximize Your Research Precision with KINTEK
Don’t let "phantom reactions" compromise your oily sludge pyrolysis data. KINTEK provides high-performance Tube, Muffle, and Vacuum furnace systems engineered for absolute chemical neutrality. Our expert R&D team delivers customizable solutions featuring high-purity quartz and corundum reaction chambers tailored to your specific temperature and purity needs.
Ready to upgrade your lab's thermal processing? Contact KINTEK today to discover how our advanced manufacturing can bring superior accuracy and durability to your research.
Visual Guide
Related Products
- Electric Rotary Kiln Small Rotary Furnace Biomass Pyrolysis Plant Rotating Furnace
- Electric Rotary Kiln Continuous Working Small Rotary Furnace Kiln for Pyrolysis Plant Heating
- Slide PECVD Tube Furnace with Liquid Gasifier PECVD Machine
- Custom Made Versatile CVD Tube Furnace Chemical Vapor Deposition CVD Equipment Machine
- Laboratory Quartz Tube Furnace RTP Heating Tubular Furnace
People Also Ask
- What are some common industrial applications of rotary kilns? Explore High-Temperature Processing Solutions
- What role does the rotary kiln serve in coal-based DRI production? Unlock Cost-Effective Ironmaking Efficiency
- What are the main applications of a rotary kiln electric furnace? Achieve Uniform Heat Treatment for Powders
- What are the key components of a rotary kiln electric furnace? Unlock Efficient Thermal Processing
- What are the differences between direct-heat and indirect-heat rotary kilns? Choose the Right Kiln for Your Process



















