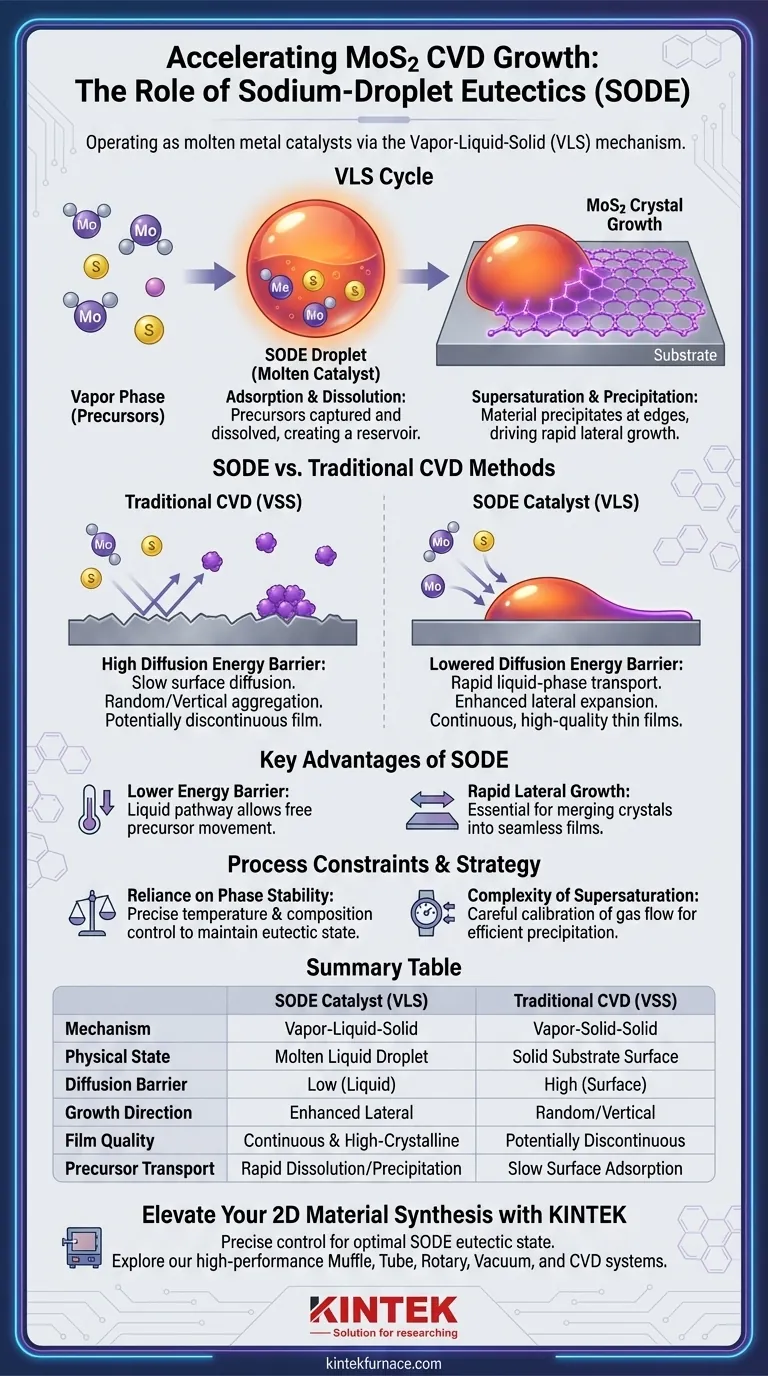
Sodium-droplet eutectics (SODE) operate as molten metal catalysts via a Vapor-Liquid-Solid (VLS) mechanism. During Chemical Vapor Deposition (CVD), these droplets capture molybdenum and sulfur precursors from the gas phase and dissolve them. Upon reaching a state of supersaturation, the droplets precipitate the material at the edges of the crystal, effectively driving the growth of the MoS2 structure.
By functioning as a liquid medium for precursor transport, SODE catalysts significantly lower the diffusion energy barrier compared to traditional methods. This mechanism is critical for achieving rapid lateral growth and ensuring the formation of continuous, high-quality MoS2 thin films.

The Mechanics of SODE Catalysis
The Vapor-Liquid-Solid (VLS) Cycle
The core of the SODE technique is the VLS mechanism.
Rather than depositing directly from a gas to a solid surface, the precursors transition through a liquid phase. The sodium-droplet eutectic acts as this intermediate liquid medium.
Adsorption and Dissolution
The process begins when the molten SODE droplets adsorb molybdenum and sulfur precursors from the surrounding gas.
These precursors do not just sit on the surface; they dissolve into the liquid droplet. This creates a "reservoir" of building materials within the catalyst itself.
Supersaturation and Precipitation
As the droplet continues to absorb precursors, it eventually reaches a supersaturated state.
At this tipping point, the droplet can no longer hold the dissolved material in liquid form. It consequently precipitates the MoS2 at the interfaces or edges of the droplet, converting the dissolved precursors into solid crystal structures.
Why SODE Outperforms Traditional Methods
Lowering the Diffusion Energy Barrier
A major challenge in standard CVD is the energy required to move atoms across a solid surface.
SODE addresses this by providing a liquid pathway. The liquid state effectively lowers the diffusion energy barrier, allowing precursors to move more freely and position themselves efficiently for crystal formation.
Driving Lateral Growth
The precipitation dynamic of SODE specifically promotes lateral growth.
Because the material precipitates at the edges of the droplet, the crystal expands outward across the substrate. This is essential for merging isolated crystals into a single, continuous thin film.
Understanding the Process Constraints
Reliance on Phase Stability
While SODE offers rapid growth, it relies heavily on maintaining the eutectic state.
The term "eutectic" implies a specific mixture that melts at a temperature lower than its individual constituents. For the catalyst to function as a molten droplet, the CVD process conditions (temperature and composition) must be precisely controlled to keep the droplet in its liquid phase without evaporating or solidifying prematurely.
The Complexity of Supersaturation
The mechanism depends on reaching supersaturation to trigger precipitation.
If the balance between precursor supply (gas phase) and dissolution (liquid phase) is off, the droplet may fail to precipitate material efficiently. This requires careful calibration of gas flow rates to ensure the "liquid engine" of the process keeps running.
Strategic Application of SODE
To leverage sodium-droplet eutectics effectively for your material synthesis, consider your specific end-goals.
- If your primary focus is growth speed: Utilize SODE to exploit the lowered diffusion energy barrier, which allows for significantly faster expansion rates than solid-phase diffusion.
- If your primary focus is film continuity: Optimize the precursor flow to maintain steady supersaturation, ensuring the lateral precipitation merges grain boundaries into a seamless film.
The SODE method transforms the CVD process by turning the diffusion bottleneck into a liquid-fueled accelerant for high-quality crystal growth.
Summary Table:
| Feature | SODE Catalyst (VLS) | Traditional CVD (VSS) |
|---|---|---|
| Mechanism | Vapor-Liquid-Solid | Vapor-Solid-Solid |
| Physical State | Molten Liquid Droplet | Solid Substrate Surface |
| Diffusion Barrier | Low (Liquid phase) | High (Surface diffusion) |
| Growth Direction | Enhanced Lateral Expansion | Random/Vertical Aggregation |
| Film Quality | Continuous & High-Crystalline | Potentially Discontinuous |
| Precursor Transport | Rapid Dissolution/Precipitation | Slow Surface Adsorption |
Elevate Your 2D Material Synthesis with KINTEK
Precise temperature and atmosphere control are critical for maintaining the SODE eutectic state and achieving optimal supersaturation. Backed by expert R&D and manufacturing, KINTEK offers high-performance Muffle, Tube, Rotary, Vacuum, and CVD systems, all customizable to meet your unique lab requirements.
Whether you are scaling MoS2 production or researching novel VLS mechanisms, our advanced high-temperature furnaces provide the stability needed for consistent, high-quality crystal growth.
Ready to optimize your research? Contact KINTEK today for a custom solution!
Visual Guide
References
- Jehyun Oh, Sang‐Yong Ju. Diffusion and Surface Effects on Sodium‐Promoted MoS <sub>2</sub> Growth Observed in <i>Operando</i>. DOI: 10.1002/smtd.202500813
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Molybdenum Disilicide MoSi2 Thermal Heating Elements for Electric Furnace
- Custom Made Versatile CVD Tube Furnace Chemical Vapor Deposition CVD Equipment Machine
- 915MHz MPCVD Diamond Machine Microwave Plasma Chemical Vapor Deposition System Reactor
- Multi Heating Zones CVD Tube Furnace Machine for Chemical Vapor Deposition Equipment
- Cylindrical Resonator MPCVD Machine System for Lab Diamond Growth
People Also Ask
- What ceramic materials are commonly used for heating elements? Discover the Best for Your High-Temp Needs
- What are the primary applications of MoSi2 heating elements in research? Achieve Reliable High-Temp Control for Material Synthesis
- What are the primary applications of Molybdenum Disilicide (MoSi2) heating elements in furnaces? Achieve High-Temp Excellence
- How can high temperature heating elements be customized for different applications? Tailor Elements for Peak Performance
- Why is a fusion process using lithium metaborate necessary for the elemental analysis of S53P4 bioactive glass?



















