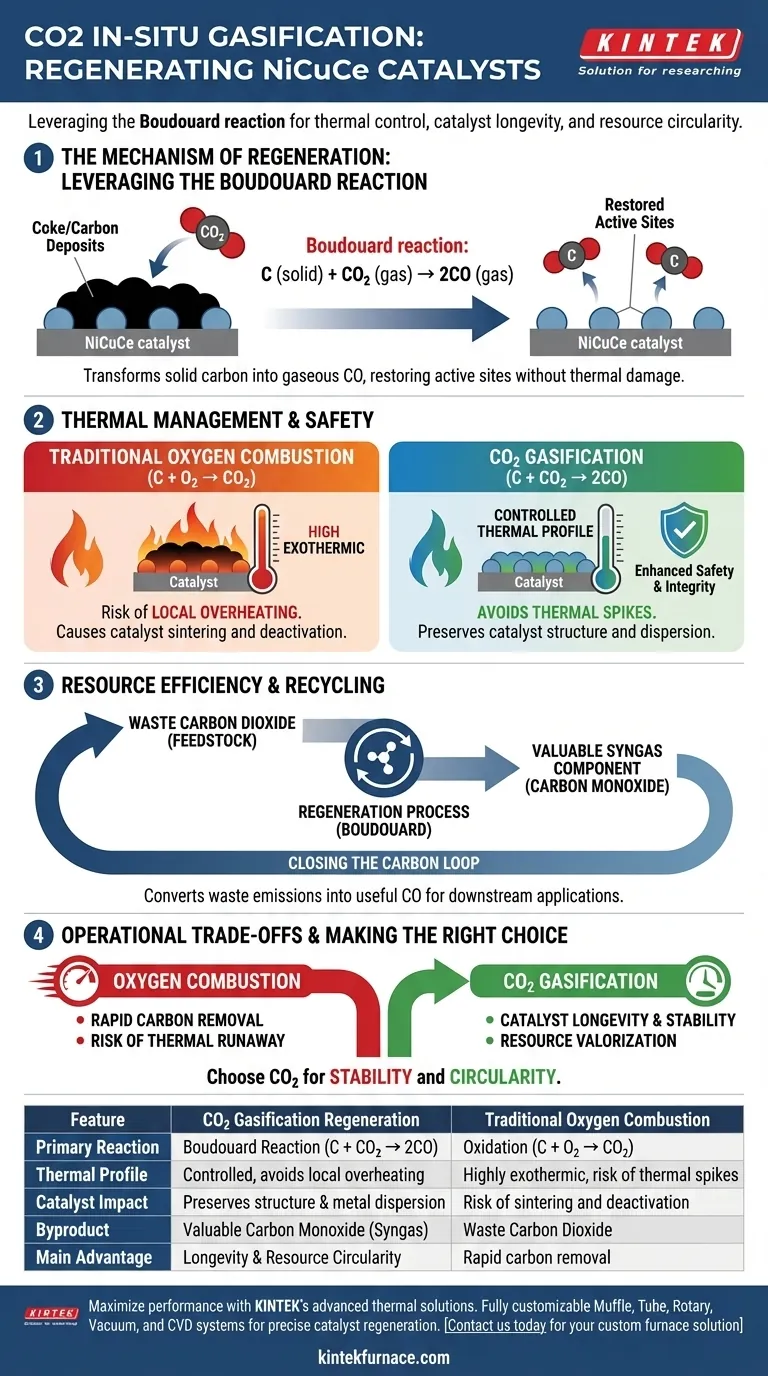
Carbon dioxide is utilized for in-situ gasification regeneration primarily to remove solid carbon deposits, known as coke, from the surface of NiCuCe catalysts without compromising the material's structural integrity. By leveraging the Boudouard reaction, this method converts solid carbon accumulation into gaseous carbon monoxide, effectively restoring the catalyst's active sites for continued use.
By substituting oxygen with carbon dioxide, this approach avoids the destructive thermal spikes of traditional combustion while simultaneously converting waste emissions into valuable syngas components.

The Mechanism of Regeneration
Leveraging the Boudouard Reaction
The core of this regeneration process is chemical rather than purely thermal.
Carbon dioxide reacts with the solid carbon deposited on the catalyst according to the Boudouard reaction ($C + CO_2 \rightarrow 2CO$).
This reaction chemically transforms the solid blocking the catalyst's active sites into a gas phase.
Restoring Active Sites
The accumulation of carbon on NiCuCe catalysts physically blocks the active sites necessary for reactions to occur.
As the carbon dioxide interacts with these deposits, it strips the carbon away, freeing the metal surface.
This returns the catalyst to an active state, allowing it to resume its intended catalytic function.
Thermal Management and Safety
Avoiding Local Overheating
A critical advantage of using carbon dioxide over traditional methods is thermal control.
Traditional regeneration often uses oxygen-based combustion to burn off carbon.
However, oxygen combustion is highly exothermic, frequently causing local overheating.
Preserving Catalyst Integrity
Excessive heat generation during regeneration can damage the catalyst structure, often leading to sintering (clumping) of the metal particles.
By utilizing the Boudouard reaction with carbon dioxide, the process avoids these intense temperature spikes.
This preserves the dispersion of the NiCuCe components and extends the overall lifespan of the catalyst.
Resource Efficiency and Recycling
Converting Waste to Fuel
Beyond cleaning the catalyst, this method serves a secondary purpose in resource management.
It utilizes waste carbon dioxide as a feedstock for the cleaning process.
Generating Syngas Components
The byproduct of the reaction is carbon monoxide (CO).
Rather than producing a waste gas, this generates a key component of syngas.
This achieves a cycle of resource recycling, turning a regeneration step into a production step.
Understanding the Operational Trade-offs
The Combustion vs. Gasification Context
It is important to understand why this method is distinct from standard air regeneration.
Oxygen-based regeneration is fast and aggressive, effectively burning off carbon through oxidation.
However, the aggression of oxygen carries the risk of thermal runaway, which can permanently deactivate the catalyst.
The Trade-off for Stability
Carbon dioxide gasification is generally a more controlled process compared to combustion.
While it eliminates the risk of thermal shock, it relies on the specific kinetics of the Boudouard reaction.
This choice prioritizes catalyst longevity and stability over the rapid, high-heat removal typical of oxidative methods.
Making the Right Choice for Your Process
When designing regeneration protocols for NiCuCe catalysts, the choice of gasification agent dictates the operational outcome.
- If your primary focus is catalyst longevity: Utilize carbon dioxide gasification to prevent local overheating and protect the structural integrity of the active sites.
- If your primary focus is resource circularity: Choose this method to valorize waste carbon dioxide by converting it into useful carbon monoxide for downstream syngas applications.
Carbon dioxide regeneration represents a shift from simple cleaning to an integrated, sustainable process that protects the catalyst while closing the carbon loop.
Summary Table:
| Feature | CO2 Gasification Regeneration | Traditional Oxygen Combustion |
|---|---|---|
| Primary Reaction | Boudouard Reaction ($C + CO_2 \rightarrow 2CO$) | Oxidation ($C + O_2 \rightarrow CO_2$) |
| Thermal Profile | Controlled, avoids local overheating | Highly exothermic, risk of thermal spikes |
| Catalyst Impact | Preserves structure and metal dispersion | Risk of sintering and deactivation |
| Byproduct | Valuable Carbon Monoxide (Syngas) | Waste Carbon Dioxide |
| Main Advantage | Longevity and Resource Circularity | Rapid carbon removal |
Maximize your catalyst performance with KINTEK’s advanced thermal solutions. Backed by expert R&D and manufacturing, KINTEK offers Muffle, Tube, Rotary, Vacuum, and CVD systems—all fully customizable to meet the precise temperature controls required for NiCuCe catalyst regeneration and gasification. Whether you are scaling lab research or optimizing industrial production, our high-temp furnaces ensure uniform heating and structural integrity for your most sensitive materials. Contact us today to find your custom furnace solution!
Visual Guide
References
- Yankun Jiang, Siqi Li. Sustainable Hydrogen from Methanol: NiCuCe Catalyst Design with CO2-Driven Regeneration for Carbon-Neutral Energy Systems. DOI: 10.3390/catal15050478
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Multi Heating Zones CVD Tube Furnace Machine for Chemical Vapor Deposition Equipment
- Electric Rotary Kiln Small Rotary Furnace for Activated Carbon Regeneration
- Slide PECVD Tube Furnace with Liquid Gasifier PECVD Machine
- Inclined Rotary Plasma Enhanced Chemical Deposition PECVD Tube Furnace Machine
- High Temperature Muffle Oven Furnace for Laboratory Debinding and Pre Sintering
People Also Ask
- What environmental protection applications utilize multi zone tube furnaces? Unlock Precision in Waste Treatment and Green Tech
- Why are multi zone tube furnaces particularly useful for nanomaterial research? Unlock Precise Thermal Control for Advanced Synthesis
- What role do multi zone tube furnaces play in new energy research? Unlock Precise Thermal Control for Innovation
- What role does a Tube Furnace play in the CVD growth of carbon nanotubes? Achieve High-Purity CNT Synthesis
- What temperature-related capabilities make multi zone tube furnaces valuable for research? Unlock Precision Thermal Control



















