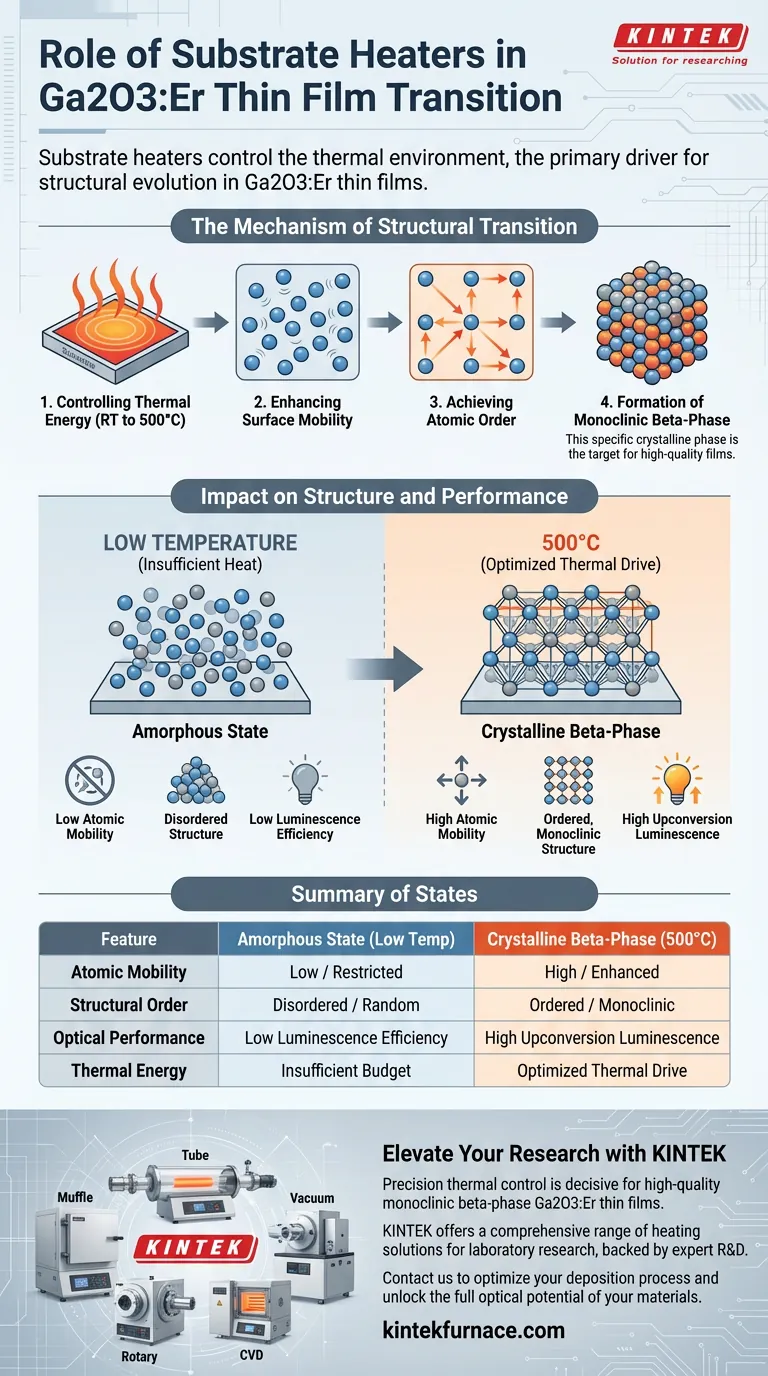
Substrate heaters act as the primary driver for structural evolution in Ga2O3:Er thin films by strictly controlling the thermal environment during deposition. By providing thermal energy up to 500 degrees Celsius, the heater increases the surface mobility of deposited atoms. This enhanced mobility allows the atoms to reorganize from a disordered, amorphous state into a highly ordered, monoclinic beta-phase crystalline structure.
The thermal energy supplied by the substrate heater is the decisive factor that enables the transition from an amorphous to a crystalline state. This structural ordering is a prerequisite for achieving significant improvements in upconversion luminescence efficiency.

The Mechanism of Structural Transition
Controlling Thermal Energy
Substrate heaters provide a tunable thermal environment for the thin film.
They are capable of maintaining conditions ranging from room temperature up to 500 degrees Celsius.
This temperature control is the fundamental variable that dictates how the film grows.
Enhancing Surface Mobility
The primary physical effect of the heater is the manipulation of atomic surface mobility.
When atoms land on a cold substrate, they have little energy to move and "freeze" in place instantly.
However, as the heater increases the temperature, it imparts kinetic energy to the atoms.
Achieving Atomic Order
With sufficient thermal energy, atoms are no longer locked in random positions.
They can migrate across the surface to find energetically favorable positions.
At 500 degrees Celsius, this mobility is high enough to prompt the atoms to arrange themselves in an orderly fashion.
Formation of the Monoclinic Beta-Phase
This orderly arrangement marks the transition from an amorphous phase to a crystalline phase.
Specifically, the material adopts a monoclinic beta-phase structure.
This specific crystalline phase is the target structure for high-quality Ga2O3:Er films.
The Impact on Performance
The Link to Luminescence
The structural state of the film is not just a morphological detail; it dictates performance.
The primary reference notes that the transition to the crystalline state is a critical condition.
It is specifically required to significantly enhance upconversion luminescence efficiency.
Critical Trade-offs in Deposition
The Cost of Insufficient Heat
Operating the substrate heater at lower temperatures limits atomic movement.
Without sufficient heat (below the 500-degree threshold), atoms remain in a disordered state.
This results in an amorphous film which fails to achieve the optical properties of the crystalline beta-phase.
Balancing Energy and Quality
High-quality crystallization requires a specific high-energy commitment.
You cannot achieve the monoclinic beta-phase—and the resulting luminescence gains—without providing the necessary thermal budget to the substrate.
Optimizing Film Deposition Strategy
To effectively manage the structural properties of your Ga2O3:Er thin films, consider the following approach based on your performance targets:
- If your primary focus is maximizing optical output: You must maintain a substrate temperature of 500 degrees Celsius to ensure the formation of the monoclinic beta-phase crystalline structure.
- If your primary focus is low-temperature processing: You must accept that the film will likely remain amorphous, resulting in reduced upconversion luminescence efficiency.
Precise thermal control is the key to unlocking the full optical potential of erbium-doped gallium oxide thin films.
Summary Table:
| Feature | Amorphous State (Low Temp) | Crystalline Beta-Phase (500°C) |
|---|---|---|
| Atomic Mobility | Low / Restricted | High / Enhanced |
| Structural Order | Disordered / Random | Ordered / Monoclinic |
| Optical Performance | Low Luminescence Efficiency | High Upconversion Luminescence |
| Thermal Energy | Insufficient Budget | Optimized Thermal Drive |
Elevate Your Thin Film Research with KINTEK
Precision thermal control is the decisive factor in achieving high-quality monoclinic beta-phase Ga2O3:Er thin films. Backed by expert R&D and world-class manufacturing, KINTEK offers a comprehensive range of Muffle, Tube, Rotary, Vacuum, and CVD systems, alongside specialized heating solutions for laboratory research.
Whether you need customizable high-temperature furnaces or precise substrate heating control, our systems are engineered to meet your unique materials science requirements. Contact us today to optimize your deposition process and unlock the full optical potential of your materials.
Visual Guide
References
- Yuanlin Liang, Yang Zhang. The Impact of the Amorphous-to-Crystalline Transition on the Upconversion Luminescence in Er3+-Doped Ga2O3 Thin Films. DOI: 10.3390/en17061397
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Mesh Belt Controlled Atmosphere Furnace Inert Nitrogen Atmosphere Furnace
- 2200 ℃ Tungsten Vacuum Heat Treat and Sintering Furnace
- Small Vacuum Heat Treat and Tungsten Wire Sintering Furnace
- Vacuum Heat Treat Sintering Furnace with Pressure for Vacuum Sintering
- Vacuum Hot Press Furnace Machine for Lamination and Heating
People Also Ask
- What industries commonly use inert atmosphere heat treating? Key Applications in Military, Automotive, and More
- What is the main purpose of heat treatment? Transform Metal Properties for Superior Performance
- How does nitrogen atmosphere heat treatment improve surface strengthening? Enhance Durability and Performance
- What is the use of nitrogen in furnace? Prevent Oxidation for Superior Heat Treatment
- How does the inert atmosphere heat treating process work? Prevent Oxidation for Superior Material Quality



















