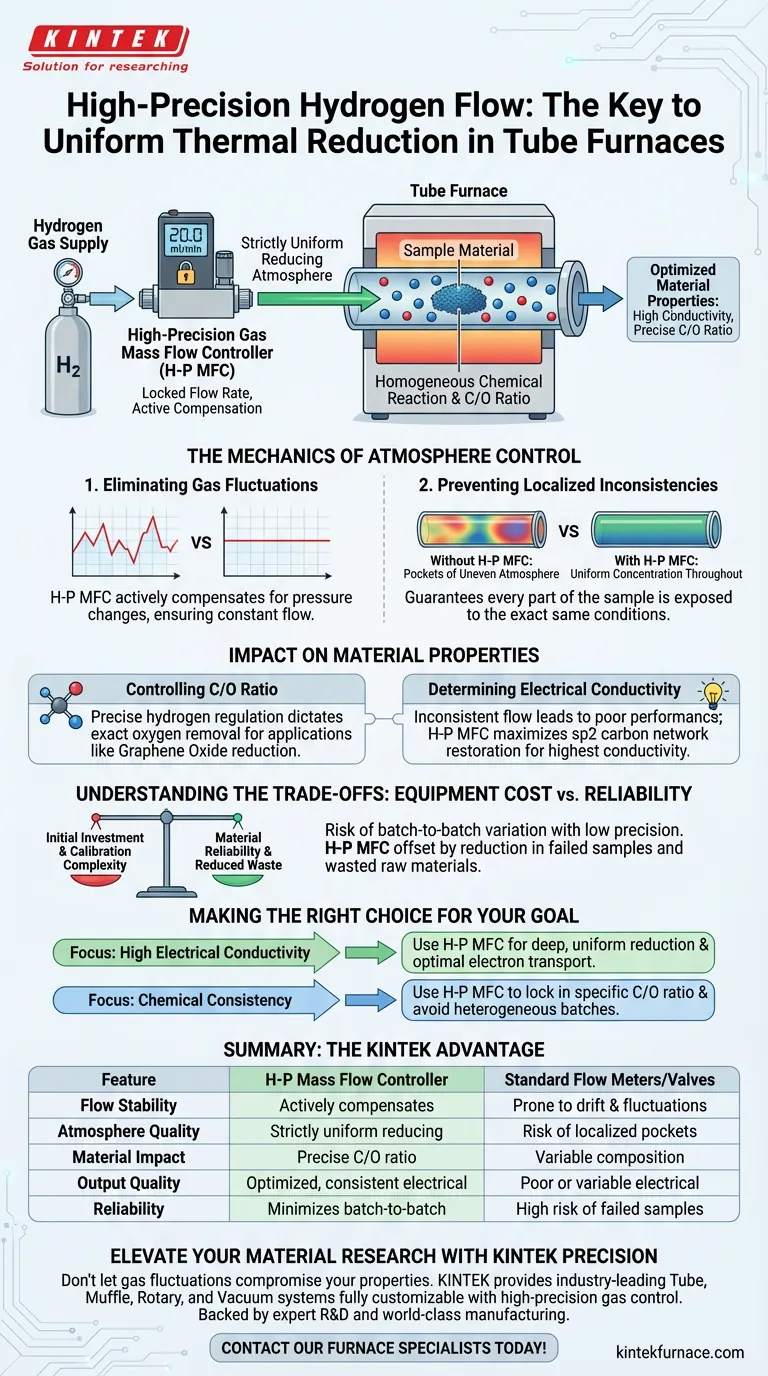
The primary significance of using a high-precision gas mass flow controller is the creation of a strictly uniform reducing atmosphere. By rigorously maintaining a specific flow rate—such as 20 ml/min—this device prevents the natural fluctuations in gas supply that occur with less precise equipment. This stability is the foundational requirement for achieving a homogeneous chemical reaction across the entire sample within the tube furnace.
In thermal reduction processes, the consistency of the gas flow is directly linked to the quality of the final material. Precise control prevents localized defects, ensuring that the critical Carbon-to-Oxygen ratio is uniform and the electrical properties of the product are optimized.

The Mechanics of Atmosphere Control
Eliminating Gas Fluctuations
Standard valves or low-precision meters often allow the flow rate of hydrogen to drift over time.
A high-precision mass flow controller actively compensates for pressure changes to maintain a locked flow rate. This ensures the furnace chamber receives a constant supply of the reducing agent (hydrogen) throughout the entire thermal cycle.
Preventing Localized Inconsistencies
When hydrogen flow fluctuates, "pockets" of uneven atmosphere can develop inside the tube.
This leads to reduced efficiency in certain areas of the sample while others are processed correctly. High-precision control guarantees that every part of the sample is exposed to the exact same concentration of reducing gas, eliminating these localized variances.
Impact on Material Properties
Controlling the Carbon-to-Oxygen (C/O) Ratio
For applications like the reduction of Graphene Oxide, the chemical composition of the final product is paramount.
The reference specifically notes that a stable atmosphere allows for precise control over the Carbon-to-Oxygen (C/O) ratio. By regulating the hydrogen supply accurately, you dictate exactly how much oxygen is removed from the graphene oxide lattice.
Determining Electrical Conductivity
The physical performance of the material is a direct result of the chemical reduction described above.
Inconsistent flow leads to inconsistent reduction, which results in poor or variable electrical conductivity. High-precision flow control ensures the highest possible conductivity by maximizing the restoration of the sp2 carbon network uniformly across the material.
Understanding the Trade-offs
Equipment Cost vs. Material Reliability
The main trade-off when selecting a high-precision mass flow controller is the initial investment and calibration complexity compared to simple rotameters.
However, relying on manual or low-precision flow control introduces a high risk of batch-to-batch variation. If your application requires specific electrical properties, the cost of the controller is offset by the reduction in failed samples and wasted raw materials.
Making the Right Choice for Your Goal
To ensure your thermal reduction process yields useful results, consider your specific targets:
- If your primary focus is High Electrical Conductivity: You must use a high-precision controller to ensure the deep, uniform reduction necessary for optimal electron transport.
- If your primary focus is Chemical Consistency: You need precise flow regulation to lock in a specific Carbon-to-Oxygen ratio and avoid chemically heterogeneous batches.
Precision in gas flow is not a luxury; it is a critical variable that dictates the functional success of your thermal reduction.
Summary Table:
| Feature | High-Precision Mass Flow Controller | Standard Flow Meters/Valves |
|---|---|---|
| Flow Stability | Actively compensates for pressure changes | Prone to drift and fluctuations |
| Atmosphere Quality | Strictly uniform reducing environment | Risk of localized pockets/inconsistencies |
| Material Impact | Precise Carbon-to-Oxygen (C/O) ratio | Variable chemical composition |
| Output Quality | Optimized, consistent electrical properties | Poor or variable electrical conductivity |
| Reliability | Minimizes batch-to-batch variation | High risk of failed samples/wasted materials |
Elevate Your Material Research with KINTEK Precision
Don't let gas fluctuations compromise your material properties. KINTEK provides industry-leading thermal solutions, including Tube, Muffle, Rotary, and Vacuum systems specifically designed for sensitive processes like thermal reduction. Our systems are fully customizable to integrate high-precision gas control, ensuring you achieve the exact chemical ratios and electrical conductivity your research demands.
Backed by expert R&D and world-class manufacturing, we help you eliminate batch-to-batch variation.
Contact our furnace specialists today to discuss your unique needs!
Visual Guide
References
- Dilek Öztekin, Sena Yaşyerli. Preparation of RGO with Enhanced Electrical Conductivity: Effects of Sequential Reductions of L-Ascorbic Acid and Thermal. DOI: 10.1007/s13369-024-09915-5
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Ultra Vacuum Electrode Feedthrough Connector Flange Power Lead for High Precision Applications
- Custom Made Versatile CVD Tube Furnace Chemical Vapor Deposition CVD Equipment Machine
- High Performance Vacuum Bellows for Efficient Connection and Stable Vacuum in Systems
- MPCVD Machine System Reactor Bell-jar Resonator for Lab and Diamond Growth
- Cylindrical Resonator MPCVD Machine System for Lab Diamond Growth
People Also Ask
- Why is a PTFE-lined stainless steel autoclave used for Ni12P5 synthesis? Key Benefits for Nanomaterial Production
- Why is it necessary to use a mechanical vacuum pump for SnSe growth? Ensure High-Purity Material Synthesis
- Why is vacuum sealing technology essential for K2In2As3 synthesis? Master High-Purity Solid-State Reactions
- What role do vacuum-sealed high-purity silica ampoules play in phase equilibrium experiments? Enhance Sample Integrity
- What is the advantage of using an integrated UHV preparation chamber? Ensure Pristine In2Se3 Surface Integrity











