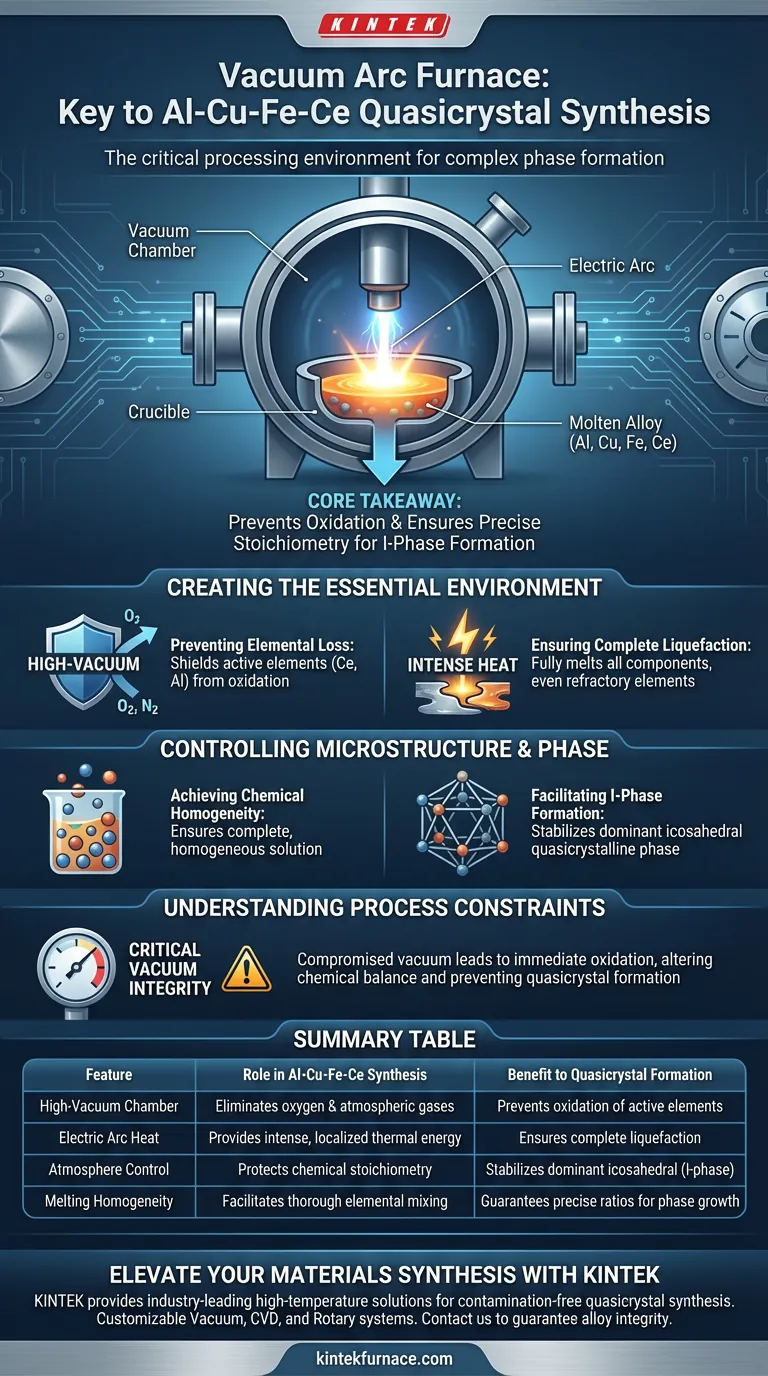
The Vacuum Arc Furnace acts as the critical processing environment for synthesizing Al-Cu-Fe-Ce quasicrystal master alloys. It utilizes intense electric arc heat within a high-vacuum chamber to melt reactive components, preventing oxidation while ensuring the precise chemical stoichiometry required for complex phase formation.
Core Takeaway The synthesis of quasicrystals is highly sensitive to impurities and compositional deviations. The Vacuum Arc Furnace solves this by eliminating oxygen during the melt, guaranteeing the stability of active elements and creating the specific conditions necessary to form the dominant icosahedral (I-phase) structure.

Creating the Essential Environment
Preventing Elemental Loss
The primary role of the furnace is to maintain a high-vacuum environment. This is non-negotiable for this specific alloy system.
Alloys containing active metallic elements (such as Cerium and Aluminum) are prone to rapid oxidation at elevated temperatures. The vacuum shield prevents these elements from reacting with air, ensuring they remain in the melt rather than becoming oxide slag.
Ensuring Complete Liquefaction
The furnace generates intense heat via an electric arc.
This thermal energy is sufficient to fully melt components with varying melting points. It ensures that even the most refractory elements in the mix are brought to a liquid state, which is the first step toward creating a unified alloy.
Controlling Microstructure and Phase
Achieving Chemical Homogeneity
Melting is not enough; the components must be thoroughly mixed.
The arc melting process ensures that all elements are fully integrated into a homogeneous solution. This precise chemical composition is vital because quasicrystals require exact stoichiometric ratios to form correctly.
Facilitating I-Phase Formation
The ultimate goal of using this furnace is to stabilize the dominant I-phase (icosahedral quasicrystalline phase).
By controlling the atmosphere and ensuring chemical uniformity, the furnace establishes the thermodynamic conditions required for this unique atomic structure to nucleate and grow. Without this controlled environment, the formation of the quasicrystal phase would likely be disrupted by impurities or segregation.
Understanding the Process Constraints
The Sensitivity of Composition
While the furnace is powerful, the process relies heavily on the maintenance of the vacuum.
If the vacuum integrity is compromised, the active elements will oxidize immediately. This alters the chemical balance of the master alloy, making it impossible to achieve the target quasicrystalline structure. The equipment does not just "melt" metal; it actively protects the recipe.
Making the Right Choice for Your Goal
To maximize the quality of your Al-Cu-Fe-Ce master alloys, consider these specific operational focuses:
- If your primary focus is Purity: Prioritize the depth of the vacuum before initiating the arc to strictly prevent the oxidation of active elements like Cerium.
- If your primary focus is Phase Stability: Ensure the melt is held under the arc long enough to guarantee complete mixing, which is the prerequisite for the dominant I-phase formation.
Success in synthesizing quasicrystals depends entirely on maintaining a contamination-free environment that preserves the precise stoichiometry of the melt.
Summary Table:
| Feature | Role in Al-Cu-Fe-Ce Synthesis | Benefit to Quasicrystal Formation |
|---|---|---|
| High-Vacuum Chamber | Eliminates oxygen and atmospheric gases | Prevents oxidation of active elements like Cerium |
| Electric Arc Heat | Provides intense, localized thermal energy | Ensures complete liquefaction of refractory components |
| Atmosphere Control | Protects chemical stoichiometry | Stabilizes the dominant icosahedral (I-phase) |
| Melting Homogeneity | Facilitates thorough elemental mixing | Guarantees the precise ratios required for phase growth |
Elevate Your Materials Synthesis with KINTEK
Precise control over stoichiometry and atmosphere is non-negotiable when synthesizing complex Al-Cu-Fe-Ce quasicrystals. KINTEK provides industry-leading high-temperature solutions—including Vacuum, CVD, and Rotary systems—engineered to eliminate contamination and ensure phase stability.
Backed by expert R&D and manufacturing, our furnaces are fully customizable to meet the unique demands of your advanced materials research. Contact KINTEK today to discover how our high-vacuum technology can guarantee the integrity of your master alloys.
Visual Guide
References
- Juan Wang, Zhong Yang. Microstructure and Properties of Al-Cu-Fe-Ce Quasicrystalline-Reinforced 6061 Aluminum Matrix Composites after Aging. DOI: 10.3390/coatings14030372
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Vacuum Hot Press Furnace Machine Heated Vacuum Press Tube Furnace
- Vacuum Sealed Continuous Working Rotary Tube Furnace Rotating Tube Furnace
- Vacuum Hot Press Furnace Machine Heated Vacuum Press
- Vacuum Heat Treat Furnace with Ceramic Fiber Liner
- Vacuum Heat Treat Sintering Furnace with Pressure for Vacuum Sintering
People Also Ask
- What is the significance of the vacuum environment for sintering stainless steel? Unlock High-Density Purity
- How does the uniaxial pressure applied by a vacuum hot press furnace affect the microstructure of ZrC-SiC materials?
- Which process parameters must be optimized for specific materials in a vacuum hot press furnace? Achieve Optimal Density and Microstructure
- What is the primary function of the vacuum environment in a vacuum hot press furnace during titanium alloy processing? Prevent Embrittlement for Superior Ductility
- Why use Vacuum Hot Press (VHP) for ZnS Ceramics? Achieve Superior IR Transparency and Mechanical Strength



















