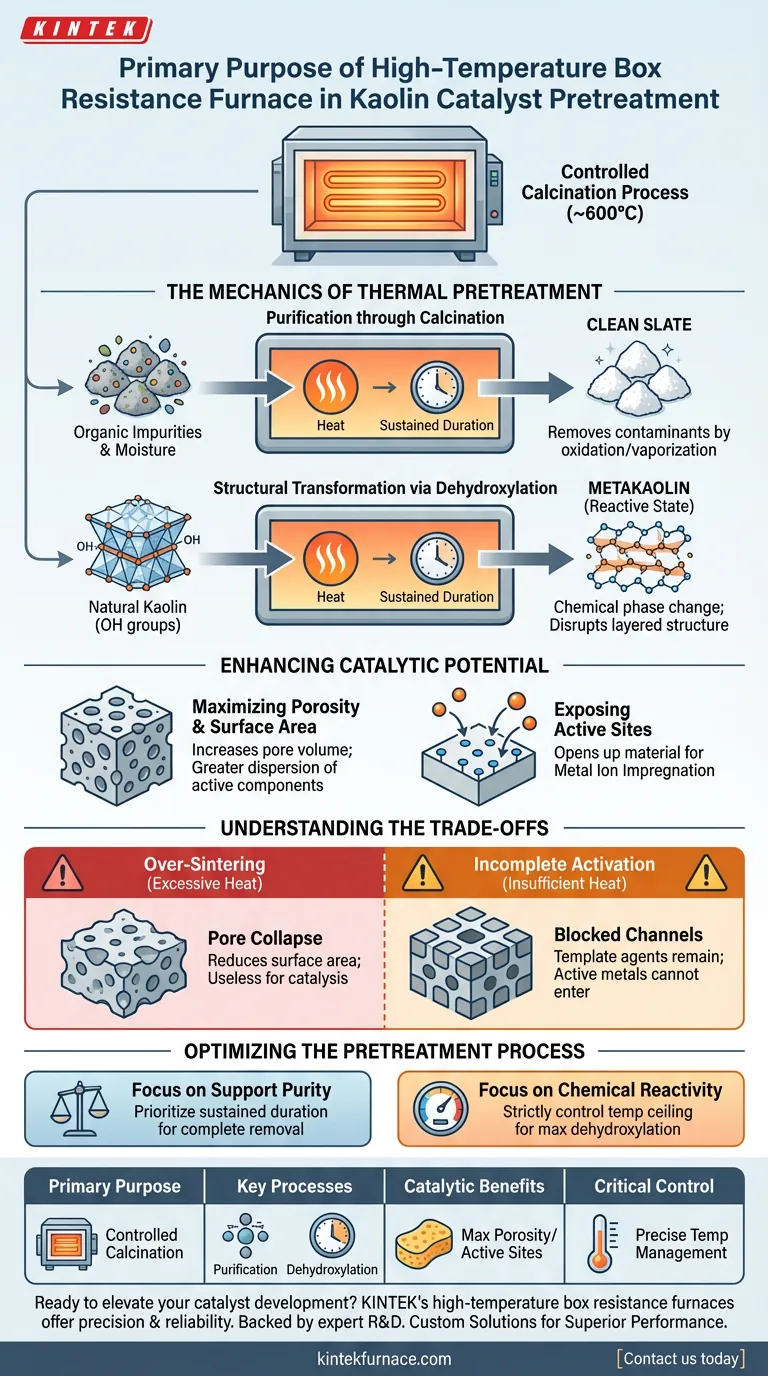
The primary purpose of using a high-temperature box resistance furnace during kaolin catalyst pretreatment is to execute a controlled calcination process, typically at 600°C. This thermal treatment is critical for purifying the raw material by eliminating organic impurities and moisture while simultaneously altering its physical structure to enhance chemical reactivity.
By triggering dehydroxylation and removing volatile components, the furnace transforms raw kaolin into a highly porous support structure. This is essential for maximizing surface area and exposing the active sites required for effective metal ion impregnation.

The Mechanics of Thermal Pretreatment
Purification through Calcination
Natural kaolin often contains organic matter, moisture, and other volatile impurities that can inhibit catalytic performance.
The high-temperature furnace ensures these contaminants are thoroughly oxidized or vaporized. By maintaining a sustained temperature (e.g., 600°C for 10 hours), the process guarantees a "clean slate" for the catalyst support.
Structural Transformation via Dehydroxylation
Beyond simple cleaning, the furnace induces a chemical phase change known as dehydroxylation.
Heat causes the removal of hydroxyl groups (-OH) from the kaolin crystal lattice. This transformation disrupts the original layered structure of the clay, converting it into a more reactive state often referred to as metakaolin.
Enhancing Catalytic Potential
Maximizing Porosity and Surface Area
The removal of internal water and organic polymers creates voids within the material.
This significantly increases the total pore volume and specific surface area of the kaolin. A larger surface area allows for a greater dispersion of active catalytic components later in the manufacturing process.
Exposing Active Sites
For a catalyst to function, it must have accessible locations where chemical reactions can occur.
The thermal treatment "opens up" the material, exposing specific active sites. This preparation is a prerequisite for metal ion impregnation, ensuring that subsequent active metals (like nickel or copper) can attach firmly and distribute uniformly.
Understanding the Trade-offs
The Risk of Over-Sintering
While high heat is necessary, excessive temperature or duration can be detrimental.
If the temperature exceeds the optimal range (e.g., pushing significantly beyond the necessary dehydroxylation point toward 1000°C+), the material may begin to sinter. Sintering causes pores to collapse and fuse, drastically reducing surface area and rendering the support useless for catalysis.
Incomplete Activation
Conversely, insufficient heating fails to fully remove template agents or organic residues.
This leaves pore channels blocked, preventing the active metal components from entering the structure. Precise temperature control is therefore the single most critical variable in the operation of the resistance furnace.
Optimizing the Pretreatment Process
To ensure the highest quality catalyst support, align your furnace parameters with your specific material goals.
- If your primary focus is Support Purity: Prioritize a sustained duration at the oxidation temperature to ensure the complete removal of all organic polymers and residues.
- If your primary focus is Chemical Reactivity: Strictly control the temperature ceiling to maximize dehydroxylation without inducing structural collapse or sintering.
The effectiveness of your final catalyst is determined not by the metal loading, but by the quality of the thermal foundation laid during this pretreatment phase.
Summary Table:
| Aspect | Description/Impact |
|---|---|
| Primary Purpose | Controlled calcination (typically ~600°C) for kaolin pretreatment. |
| Key Processes | Purification (removes organic impurities, moisture), Dehydroxylation (converts to metakaolin). |
| Catalytic Benefits | Maximizes porosity & surface area, exposes active sites for metal ion impregnation. |
| Critical Control | Precise temperature management prevents over-sintering and ensures complete activation. |
Ready to elevate your catalyst development? KINTEK’s high-temperature box resistance furnaces offer the precision and reliability needed for optimal kaolin pretreatment and beyond. Backed by expert R&D and manufacturing, KINTEK offers Muffle, Tube, Rotary, Vacuum, CVD systems, and other lab high-temp furnaces, all customizable for unique needs. Achieve superior material performance with our advanced solutions – contact us today to discuss your project!
Visual Guide
References
- Luqman Buchori, Ndaru Okvitarini. Preparation of KI/KIO3/Methoxide Kaolin Catalyst and Performance Test of Catalysis in Biodiesel Production. DOI: 10.26554/sti.2024.9.2.359-370
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 1400℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- 1700℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- 1700℃ Controlled Inert Nitrogen Atmosphere Furnace
- 1700℃ High Temperature Muffle Oven Furnace for Laboratory
- 1200℃ Muffle Oven Furnace for Laboratory
People Also Ask
- In what scenarios are laboratory high-temperature tube furnaces or muffle furnaces utilized? Study MgTiO3-CaTiO3 Ceramics
- What role do high-performance box or tube furnaces play in LATP sintering? Master Densification & Ionic Conductivity
- What is the function of a furnace in CuAlMn alloy treatment? Achieve Perfect Microstructural Homogenization
- What is a high temperature tube furnace? Achieve Precise Heat and Atmosphere Control
- How does a laboratory high-temperature tube furnace contribute to the conversion of electrospun fibers? Expert Insights



















