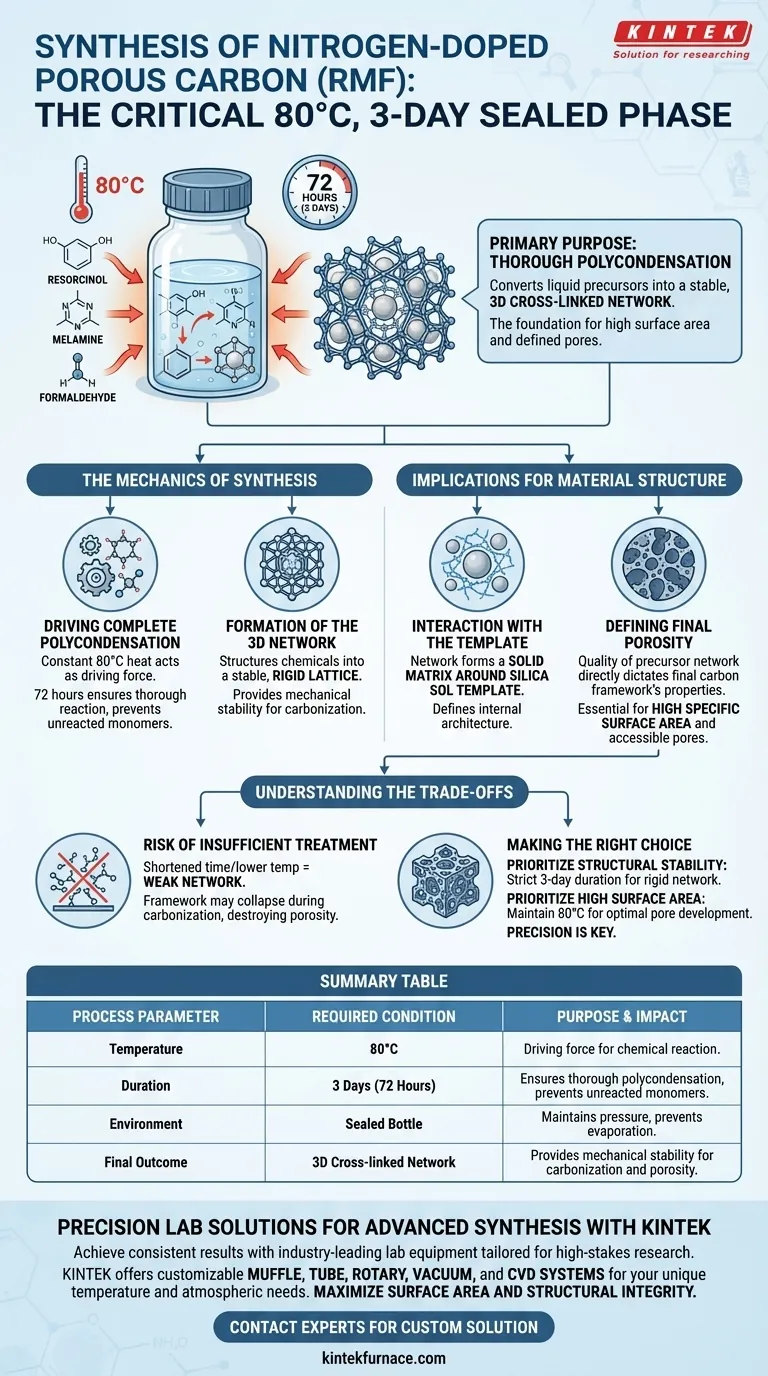
The primary purpose of sealing the reaction bottle and maintaining 80°C for three days is to drive the thorough polycondensation of the chemical precursors: resorcinol, melamine, and formaldehyde. This extended thermal treatment is the critical step that transforms these liquid components into a stable, three-dimensional cross-linked network.
By strictly controlling this environment for 72 hours, you ensure the formation of a robust structural foundation. This step is the prerequisite for achieving the material's high specific surface area and defined pore structure after the subsequent carbonization process.

The Mechanics of the Synthesis
Driving Complete Polycondensation
The application of constant heat at 80°C acts as the driving force for the chemical reaction between the precursors.
Maintaining this temperature for a full three days ensures the reaction is thorough. This prevents the presence of unreacted monomers, which could destabilize the final material.
Formation of the 3D Network
The ultimate goal of this phase is not merely to react the chemicals, but to structure them.
The process facilitates the creation of a stable three-dimensional cross-linked network. This rigid lattice provides the necessary mechanical stability for the steps that follow.
Implications for Material Structure
Interaction with the Template
It is critical to note that this network formation occurs in the presence of a silica sol template.
The polymer network creates a solid matrix around the silica. This interaction is what defines the internal architecture of the material.
Defining Final Porosity
The quality of this precursor network directly dictates the properties of the final carbon framework.
A well-developed precursor network is essential for achieving a high specific surface area. It ensures that after carbonization, the material retains a highly developed and accessible pore structure.
Understanding the Trade-offs
The Risk of Insufficient Treatment
Cutting this process short or reducing the temperature is a common pitfall that undermines structural integrity.
Incomplete polycondensation results in a weak network. Without a fully cross-linked structure, the framework may collapse during carbonization, destroying the desired porosity and reducing surface area.
Making the Right Choice for Your Goal
To maximize the quality of your Nitrogen-doped Porous Carbon, consider these priorities:
- If your primary focus is structural stability: Adhere strictly to the three-day duration to ensure the cross-linked network is rigid enough to withstand high-temperature carbonization.
- If your primary focus is high surface area: Do not alter the temperature; 80°C is the calibrated set point to form the specific precursor architecture required for optimal pore development.
Precision during this synthesis phase is the single most important factor in securing the performance of the final carbon material.
Summary Table:
| Process Parameter | Required Condition | Purpose & Impact |
|---|---|---|
| Temperature | 80°C | Acts as the driving force for chemical reaction between precursors. |
| Duration | 3 Days (72 Hours) | Ensures thorough polycondensation and prevents unreacted monomers. |
| Environment | Sealed Bottle | Maintains consistent pressure and prevents evaporation of precursors. |
| Final Outcome | 3D Cross-linked Network | Provides the mechanical stability needed for carbonization and porosity. |
Precision Lab Solutions for Advanced Synthesis
Achieving consistent results in the synthesis of Nitrogen-doped Porous Carbon requires exact thermal control. KINTEK provides industry-leading lab equipment tailored for high-stakes research. Backed by expert R&D and manufacturing, KINTEK offers Muffle, Tube, Rotary, Vacuum, and CVD systems, all fully customizable to your unique temperature and atmospheric needs.
Maximize your material's surface area and structural integrity with KINTEK's precision technology.
Contact our experts today to find your custom solution!
Visual Guide
References
- Qi Chen, Licheng Ling. Enhanced Electrochemical Performance of Dual-Ion Batteries with T-Nb2O5/Nitrogen-Doped Three-Dimensional Porous Carbon Composites. DOI: 10.3390/molecules30020227
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 915MHz MPCVD Diamond Machine Microwave Plasma Chemical Vapor Deposition System Reactor
- MPCVD Machine System Reactor Bell-jar Resonator for Lab and Diamond Growth
- Custom Made Versatile CVD Tube Furnace Chemical Vapor Deposition CVD Equipment Machine
- 1200℃ Controlled Inert Nitrogen Atmosphere Furnace
- High Temperature Muffle Oven Furnace for Laboratory Debinding and Pre Sintering
People Also Ask
- What are some key features and benefits of the microwave plasma chemical vapor deposition system? Achieve Unmatched Material Synthesis
- What are the main components of an MPCVD machine? Unlock the Secrets of Diamond Synthesis
- What is the MPCVD method and why is it considered effective for diamond deposition? Superior Purity & High Growth Rates
- What is the basic principle of operation for the microwave plasma chemical vapor deposition system? Unlock High-Purity Material Growth
- What are the essential components of an MPCVD reactor for diamond film deposition? Unlock High-Quality Diamond Growth



















