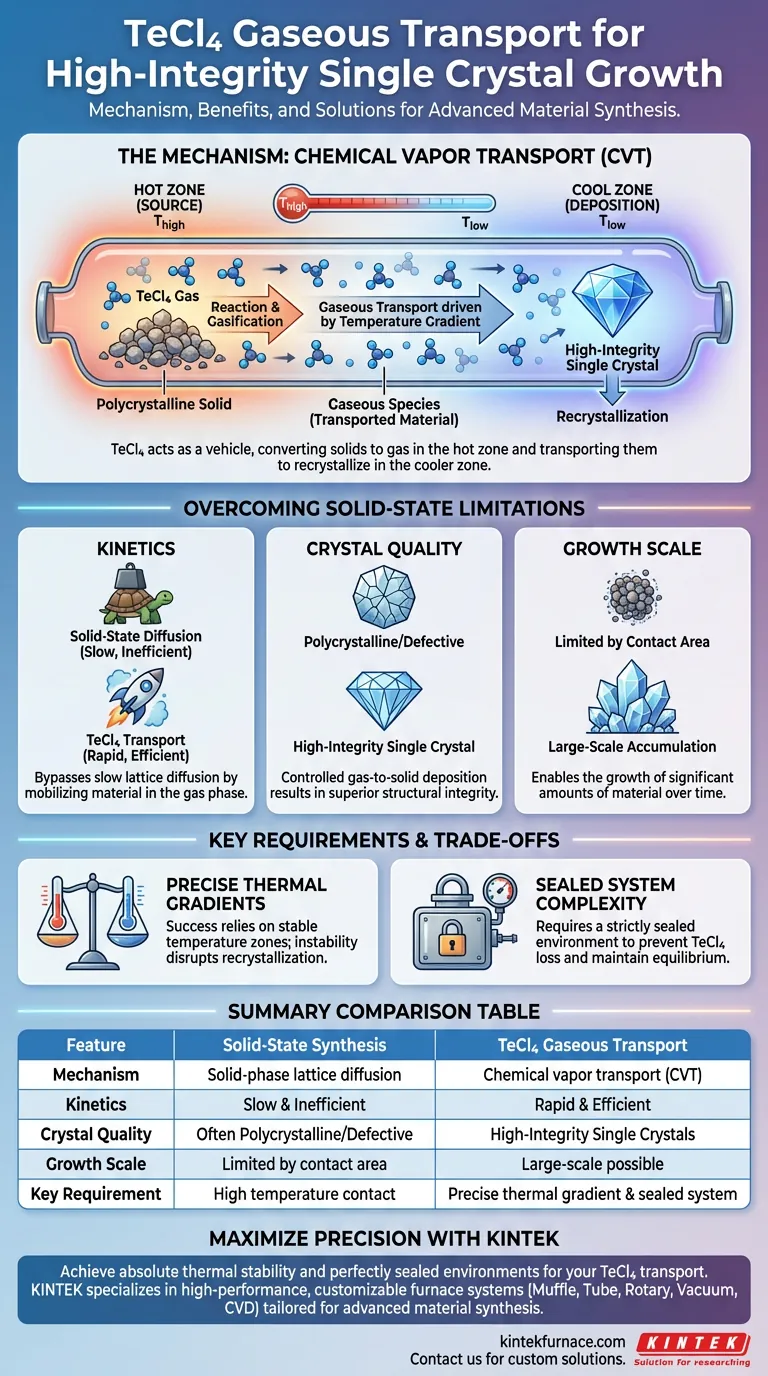
TeCl4 functions as a chemical transport medium that converts solid materials into gaseous species within a sealed furnace environment. This mechanism enables the movement of chemical components from a high-temperature source zone to a lower-temperature deposition zone. By facilitating this phase change, it effectively bypasses the inherent sluggishness of solid-state diffusion to grow high-integrity crystals.
While standard synthesis relies on slow direct contact between solids, TeCl4 acts as a vehicle to vaporize and transport material. This process is the key to producing large-scale single crystals with structural integrity that solid-state methods generally fail to achieve.

The Physics of the Transport Process
The Role of the Sealed Environment
The process begins in a sealed furnace environment. This closed system is critical to prevent the loss of the volatile transport agent and to maintain the chemical equilibrium necessary for transport.
Inside this system, TeCl4 interacts with the solid polycrystalline source material.
Reaction at High Temperatures
At the furnace's hotter zone, TeCl4 reacts with the polycrystalline solids.
This reaction converts the solid material into gaseous species. This phase conversion is the engine of the entire process, mobilizing the material for transport.
Recrystallization in the Cooler Zone
Once gasified, the chemical components travel to a lower-temperature zone within the furnace.
Driven by the temperature difference, the thermodynamic equilibrium shifts. This causes the gaseous species to release the transported material, which then recrystallizes as a single crystal.
Overcoming Solid-State Limitations
Bypassing Slow Diffusion
Standard solid-state synthesis relies on solid-phase diffusion, which is kinetically slow and often inefficient.
Atoms move with difficulty through solid lattices. By converting the material to a gas, TeCl4 removes this bottleneck, allowing for rapid and efficient movement of components.
Achieving High Integrity
The controlled nature of the gas-to-solid deposition results in superior crystal quality.
Because the material builds up gradually from the gas phase, the resulting crystals possess high integrity. This method is specifically used when standard methods fail to produce crystals of sufficient quality.
Enabling Large-Scale Growth
The efficiency of gaseous transport allows for the accumulation of significant amounts of material over time.
This makes TeCl4 transport essential for producing large-scale single crystals, a feat that is often impossible using traditional solid-state mixing techniques.
Understanding the Trade-offs
Dependence on Temperature Gradients
The success of this method relies entirely on the precise management of temperature zones.
Since the transport moves from a high-temperature zone to a lower-temperature zone, any instability in this thermal gradient can disrupt the recrystallization process or stop transport altogether.
Complexity of Sealed Systems
Unlike open-air solid-state firing, this method requires a strictly sealed environment.
This adds a layer of technical complexity to the furnace setup, as leaks or seal failures will compromise the transport medium (TeCl4) and halt crystal growth.
Making the Right Choice for Your Goal
This method is a specific tool for overcoming kinetic barriers in crystal growth.
- If your primary focus is crystal size: Utilize TeCl4 transport to bypass the volume limitations inherent in solid-state diffusion, enabling large-scale growth.
- If your primary focus is structural quality: rely on the gas-phase recrystallization process to produce high-integrity single crystals that are free from the defects common in polycrystalline solids.
By leveraging the phase-changing properties of TeCl4, you transform a slow physical struggle into an efficient chemical transport system.
Summary Table:
| Feature | Solid-State Synthesis | TeCl4 Gaseous Transport |
|---|---|---|
| Mechanism | Solid-phase lattice diffusion | Chemical vapor transport (CVT) |
| Kinetics | Slow and inefficient | Rapid and efficient |
| Crystal Quality | Often polycrystalline/defective | High-integrity single crystals |
| Growth Scale | Limited by contact area | Large-scale accumulation possible |
| Key Requirement | High temperature contact | Precise thermal gradient & sealed system |
Maximize Your Crystal Growth Precision with KINTEK
Achieving high-integrity single crystals through TeCl4 gaseous transport requires absolute thermal stability and a perfectly sealed environment. At KINTEK, we specialize in providing high-performance furnace systems tailored for advanced material synthesis.
Backed by expert R&D and manufacturing, we offer Muffle, Tube, Rotary, Vacuum, and CVD systems, all fully customizable to meet the unique temperature gradients and chemical requirements of your research.
Ready to elevate your lab's high-temperature capabilities? Contact us today to discuss your custom furnace solution with our technical experts.
Visual Guide
References
- Matthias Weil, Harishchandra Singh. CoTeO<sub>4</sub> – a wide-bandgap material adopting the dirutile structure type. DOI: 10.1039/d3ma01106b
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Custom Made Versatile CVD Tube Furnace Chemical Vapor Deposition CVD Equipment Machine
- Slide PECVD Tube Furnace with Liquid Gasifier PECVD Machine
- Multi Heating Zones CVD Tube Furnace Machine for Chemical Vapor Deposition Equipment
- Split Chamber CVD Tube Furnace with Vacuum Station CVD Machine
- Small Vacuum Heat Treat and Tungsten Wire Sintering Furnace
People Also Ask
- What is the working principle of a CVD tube furnace? Achieve Precise Thin Film Deposition for Your Lab
- How does CVD tube furnace sintering enhance graphene growth? Achieve Superior Crystallinity and High Electron Mobility
- How can integrating CVD tube furnaces with other technologies benefit device fabrication? Unlock Advanced Hybrid Processes
- Why are CVD tube furnace sintering systems indispensable for 2D material research and production? Unlock Atomic-Scale Precision
- What is a CVD tube furnace and what is its primary function? Discover Precision Thin-Film Deposition



















