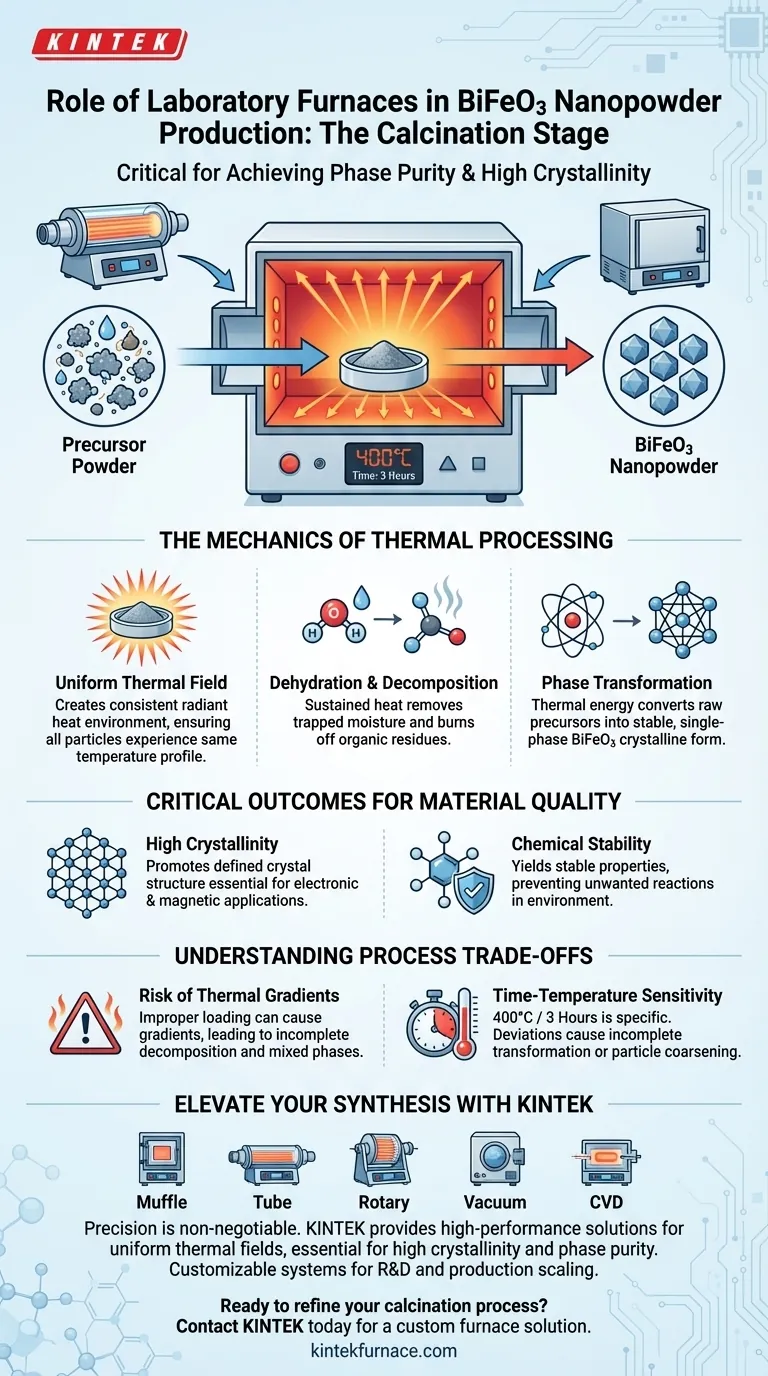
In the final production stages of bismuth ferrite (BiFeO3) nanopowder, the laboratory tube furnace or box resistance furnace serves as the critical vessel for calcination.
This equipment subjects precursor powders—typically created via hydrothermal methods—to a strictly controlled high-temperature environment to drive essential chemical and physical transformations. By maintaining a constant temperature, such as 400°C for three hours, the furnace ensures the material transitions from a raw precursor to a functional nanomaterial.
The furnace provides a uniform thermal field that acts as the catalyst for phase purity. It does not merely dry the powder; it supplies the energy required to dehydrate the structure, decompose organic residuals, and crystallize the material into a stable, single-phase form.

The Mechanics of Thermal Processing
Generating a Uniform Thermal Field
The primary function of the furnace is to create a consistent, uniform thermal field around the sample.
Unlike open-air heating, a resistance or tube furnace envelops the sample in radiant heat, ensuring that every particle of the precursor powder experiences the exact same temperature profile.
Driving Dehydration and Decomposition
The precursor powder entering the furnace often contains residual water and organic components left over from the synthesis process.
The sustained heat drives dehydration, removing moisture trapped within the molecular structure. Simultaneously, it triggers decomposition, effectively burning off organic residues that would otherwise act as contaminants in the final product.
Inducing Phase Transformation
The most critical function of the furnace is enabling phase transformation.
The thermal energy rearranges the atomic structure of the precursor. This rearrangement converts the raw ingredients into the specific crystalline arrangement required for bismuth ferrite, eliminating intermediate or impurity phases.
Critical Outcomes for Material Quality
Achieving High Crystallinity
For nanopowders to function correctly in electronic or magnetic applications, they must have a defined crystal structure.
The calcination process promotes high crystallinity, ensuring the atoms are ordered in a precise, repeating pattern. This structural integrity is directly linked to the material's final performance metrics.
Ensuring Chemical Stability
A properly calcined powder yields a material with stable chemical properties.
By completing the reaction in the furnace, the final BiFeO3 nanocrystals are rendered chemically inert relative to their environment, preventing further unwanted reactions during subsequent handling or application.
Understanding the Process Trade-offs
The Risk of Thermal Gradients
While these furnaces are designed for uniformity, improper loading or calibration can create thermal gradients.
If the thermal field is not uniform, parts of the powder may fail to reach the decomposition threshold. This results in a final product contaminated with residual organics or mixed phases, compromising the purity of the batch.
Time-Temperature Sensitivity
The parameters cited—400°C for three hours—are specific to achieving the desired phase.
Deviating from this precise window presents a trade-off. Insufficient time or temperature leads to incomplete transformation, while excessive heat can lead to particle coarsening, causing the nanopowder to lose its valuable high-surface-area characteristics.
Making the Right Choice for Your Goal
To maximize the quality of your bismuth ferrite nanopowders, you must match your furnace parameters to your specific material requirements.
- If your primary focus is Phase Purity: Prioritize a furnace capability that guarantees a uniform thermal field to ensure complete elimination of impurity phases and organic residues.
- If your primary focus is Process Consistency: Adhere strictly to verified calcination protocols (e.g., constant holding at 400°C) to ensure reproducible high crystallinity across different batches.
- If your primary focus is R&D Flexibility: Utilize tube furnaces, which are standard for exploring new synthesis parameters and small-scale process development in materials science.
By precisely controlling the thermal history of your precursor, you transform a raw chemical mixture into a high-performance, single-phase nanomaterial.
Summary Table:
| Process Function | Mechanism | Impact on BiFeO3 Nanopowder |
|---|---|---|
| Dehydration | Sustained heat removal of moisture | Removes structural water and moisture content |
| Decomposition | Thermal breakdown of organics | Eliminates contaminants and residual synthesis organic matter |
| Phase Transformation | Atomic rearrangement at 400°C | Converts precursors into stable, single-phase crystalline BiFeO3 |
| Thermal Uniformity | Radiant heat envelopment | Ensures consistent material properties and prevents impurity phases |
Elevate Your Nanomaterial Synthesis with KINTEK
Precision is non-negotiable in the final stages of bismuth ferrite production. KINTEK provides high-performance laboratory solutions—including Muffle, Tube, Rotary, Vacuum, and CVD systems—specifically designed to maintain the uniform thermal fields required for high crystallinity and phase purity.
Backed by expert R&D and world-class manufacturing, our lab high-temp furnaces are fully customizable to meet your unique materials science needs. Whether you are optimizing R&D protocols or scaling up production, KINTEK ensures your nanopowders achieve superior chemical stability and performance.
Ready to refine your calcination process? Contact KINTEK today for a custom furnace solution.
Visual Guide
References
- Farzaneh Abbasi, Marjan Tanzifi. RSM optimization and morphological investigation of bismuth ferrite for photodegradation of organic pollutant under visible light. DOI: 10.1038/s41598-025-16568-3
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 1400℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- Laboratory Quartz Tube Furnace RTP Heating Tubular Furnace
- Laboratory Vacuum Tilt Rotary Tube Furnace Rotating Tube Furnace
- 1700℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- 1200℃ Split Tube Furnace Laboratory Quartz Tube Furnace with Quartz Tube
People Also Ask
- In what scenarios are laboratory high-temperature tube furnaces or muffle furnaces utilized? Study MgTiO3-CaTiO3 Ceramics
- What is the function of a furnace in CuAlMn alloy treatment? Achieve Perfect Microstructural Homogenization
- Why is a high-temperature tube furnace required for NiWO4 calcination? Achieving High-Performance Cathode Materials
- What is the mechanism of a high-temperature furnace in Bi-2223 sintering? Achieve Precision Phase Transformation
- What role do high-performance box or tube furnaces play in LATP sintering? Master Densification & Ionic Conductivity



















