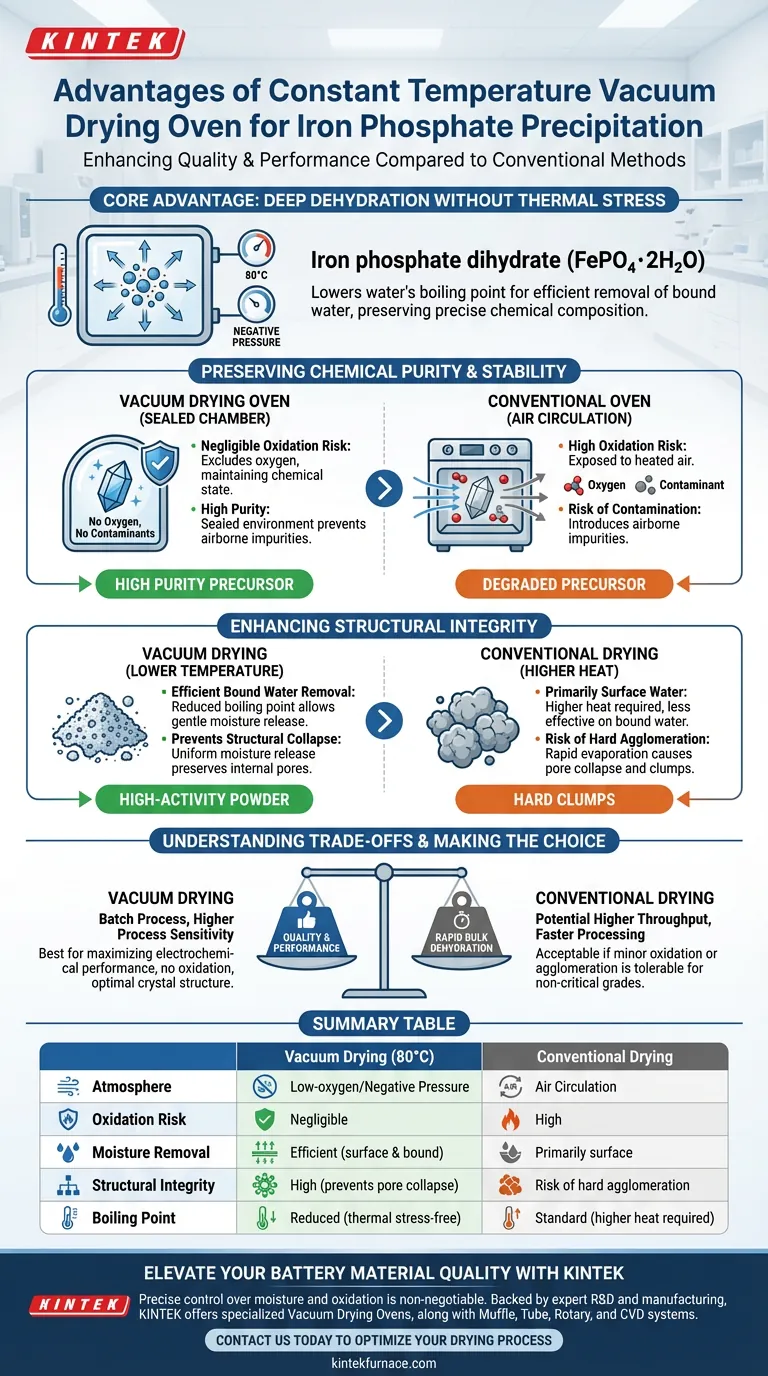
The use of a constant temperature vacuum drying oven specifically at 80 °C significantly enhances the quality of iron phosphate precipitation compared to conventional methods. This approach accelerates the removal of both adsorbed and chemically bound water while maintaining a low-oxygen environment. By eliminating the risk of oxidation and impurity introduction, this method preserves the structural stability of the iron phosphate dihydrate (FePO4·2H2O) precursor, which is critical for downstream battery performance.
The core advantage of vacuum drying lies in altering the physics of evaporation: by lowering the boiling point of water, you can achieve deep dehydration without the thermal stress that damages crystal structures. This ensures the precursor retains the precise chemical composition required for high-quality material synthesis.
Preserving Chemical Purity and Stability
The primary challenge in drying iron phosphate is removing moisture without altering the material's chemical state. Vacuum drying addresses this through precise environmental control.
Prevention of Oxidation
Conventional ovens rely on heated air circulation, which exposes the material to oxygen. For iron-based materials, this presents a significant risk of unwanted oxidation.
A vacuum oven creates a negative pressure environment that effectively excludes oxygen. This ensures the iron phosphate remains in its intended chemical state throughout the drying process.
Elimination of Contamination
Standard drying methods can introduce airborne impurities or dust. Vacuum drying occurs in a sealed chamber, preventing the introduction of external contaminants.
This isolation is vital for maintaining the high purity levels required for battery-grade precursors, where even trace impurities can degrade performance.
Enhancing Structural Integrity
Beyond chemical purity, the physical structure of the dried precipitate dictates how well it will perform in subsequent synthesis steps.
Efficient Removal of Bound Water
Iron phosphate precursors contain both surface water and "bound" water integrated into the structure.
At 80 °C under vacuum, the boiling point of water is significantly reduced. This allows for the efficient removal of stubborn bound water without resorting to extreme temperatures that could degrade the material.
Preventing Structural Collapse
Rapid evaporation at high temperatures in conventional ovens can cause surface tension issues, leading to pore collapse or hard agglomeration.
Vacuum drying promotes a more uniform release of moisture from within the material. This preserves the internal porous structure and ensures the precursor remains a loose, high-activity powder rather than forming hard clumps.
Understanding the Trade-offs
While vacuum drying offers superior material quality, it is important to recognize the operational differences compared to conventional methods.
Throughput Limitations
Vacuum drying is typically a batch process, which may offer lower throughput compared to continuous belt dryers used in conventional heating.
Process Sensitivity
The vacuum environment must be carefully controlled. Applying a vacuum too rapidly can cause "bumping," where solvents boil violently and physically displace the powder within the chamber.
Making the Right Choice for Your Goal
Selecting the correct drying method depends on the strictness of your material requirements.
- If your primary focus is maximizing electrochemical performance: Prioritize vacuum drying to ensure the iron phosphate precursor remains free of oxidation and retains its optimal crystal structure.
- If your primary focus is rapid bulk dehydration of non-critical grades: Conventional drying methods may offer faster processing times, provided that minor oxidation or agglomeration is acceptable.
By controlling the drying atmosphere and temperature, you transform drying from a simple dehydration step into a critical quality assurance process for your final material.
Summary Table:
| Feature | Vacuum Drying (80°C) | Conventional Drying |
|---|---|---|
| Atmosphere | Low-oxygen/Negative Pressure | Air Circulation |
| Oxidation Risk | Negligible | High |
| Moisture Removal | Efficient (surface & bound) | Primarily surface |
| Structural Integrity | High (prevents pore collapse) | Risk of hard agglomeration |
| Purity Level | High (sealed environment) | Risk of airborne contaminants |
| Boiling Point | Reduced (thermal stress-free) | Standard (higher heat required) |
Elevate Your Battery Material Quality with KINTEK
Precise control over moisture and oxidation is non-negotiable for high-performance iron phosphate synthesis. Backed by expert R&D and manufacturing, KINTEK offers specialized Vacuum Drying Ovens, along with Muffle, Tube, Rotary, and CVD systems—all customizable to meet your laboratory's unique high-temperature requirements.
Ensure the structural stability and chemical purity of your precursors with our advanced thermal solutions. Contact us today to optimize your drying process and see how our expertise can drive your material innovation forward.
Visual Guide
References
- Ziyang Xu, Jingkui Qu. Sustainable Utilization of Fe(Ⅲ) Isolated from Laterite Hydrochloric Acid Lixivium via Ultrasonic-Assisted Precipitation to Synthesize LiFePO4/C for Batteries. DOI: 10.3390/ma17020342
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Vacuum Heat Treat Sintering Furnace Molybdenum Wire Vacuum Sintering Furnace
- 1700℃ High Temperature Muffle Oven Furnace for Laboratory
- 2200 ℃ Tungsten Vacuum Heat Treat and Sintering Furnace
- 1200℃ Muffle Oven Furnace for Laboratory
- High Temperature Muffle Oven Furnace for Laboratory Debinding and Pre Sintering
People Also Ask
- How does vacuum sintering contribute to cost reduction in material processing? Lower Expenses with Superior Parts
- What are the main structural components of a vacuum sintering furnace? Unlock Precision in High-Temperature Processing
- What combination of pumps is typically used for vacuum sintering furnaces? Boost Efficiency with Rotary Vane & Roots Pumps
- What are the key components of a vacuum sintering furnace? Essential Parts for Precision Material Processing
- How does vacuum sintering improve dimensional tolerances? Achieve Uniform Shrinkage and Precision










