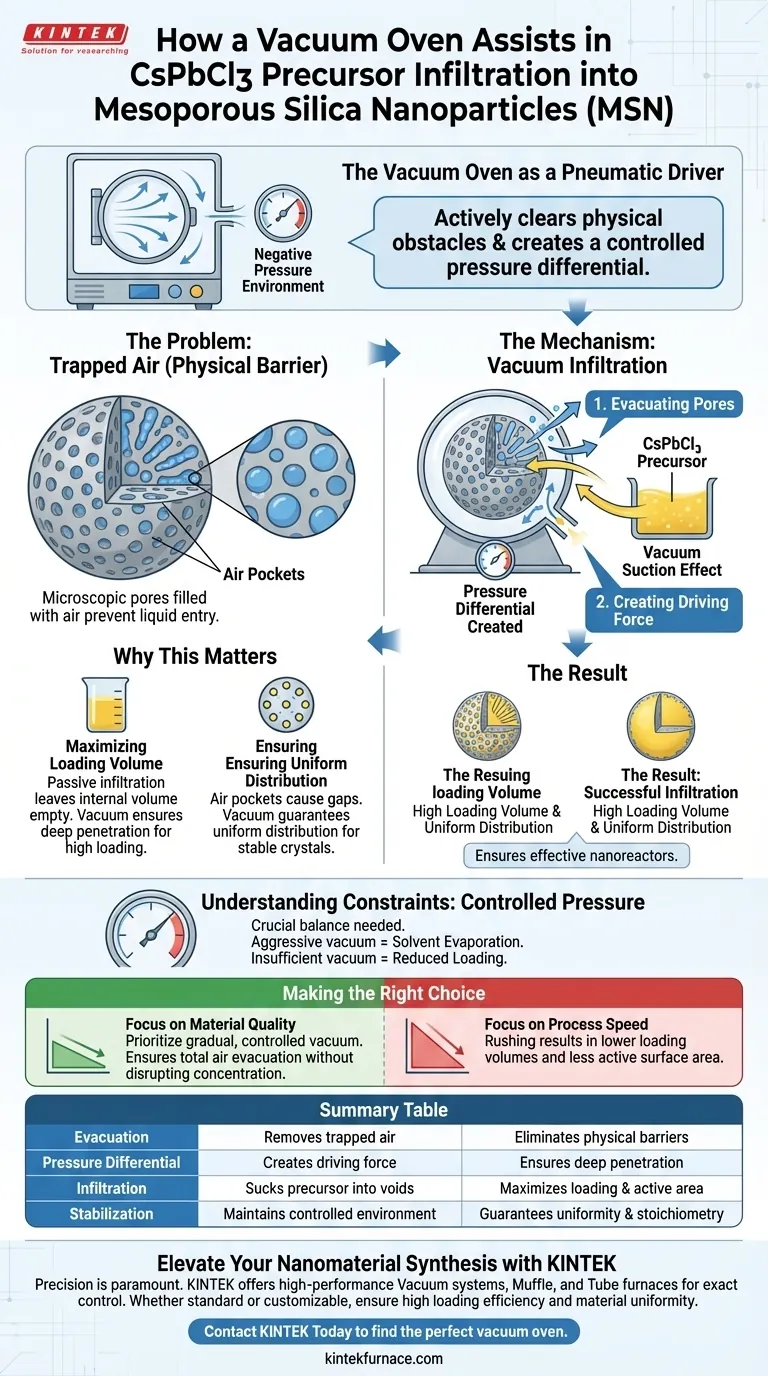
The vacuum oven functions as a pneumatic driver, actively clearing the physical obstacles that prevent successful chemical infiltration. It creates a controlled negative pressure environment that evacuates air trapped inside the internal pores of Mesoporous Silica Nanoparticles (MSN), allowing the CsPbCl3 precursor solution to occupy the space previously held by the gas.
By removing air pockets and creating a pressure differential, the vacuum oven forces the liquid precursor deep into the silica structure, ensuring the high loading volume and uniformity required for effective nanoreactors.

The Mechanics of Vacuum Infiltration
Evacuating the Pores
Under normal atmospheric conditions, the microscopic pores of Mesoporous Silica Nanoparticles are filled with air.
This trapped air acts as a physical barrier, preventing liquids from entering the deep internal structure of the particle.
The vacuum oven removes this barrier by generating a negative pressure environment, effectively sucking the air out of the nanopores.
Creating the Driving Force
Once the air is evacuated, a significant pressure differential is established between the interior of the pore and the external environment.
This differential creates a vacuum suction effect.
This force drives the CsPbCl3 perovskite precursor solution into the pores with much greater energy and efficiency than passive soaking could achieve.
Why This Matters for CsPbCl3 Synthesis
Maximizing Loading Volume
For MSNs to function effectively as nanoreactors, they must contain a sufficient amount of the active chemical material.
Passive infiltration often results in surface-level coating, leaving the internal volume empty.
The vacuum method ensures the precursor penetrates deeply, resulting in a high loading volume of CsPbCl3 within the silica host.
Ensuring Uniform Distribution
Perovskite precursors like CsPbCl3 require precise stoichiometry and placement to form stable crystals.
Air pockets can cause gaps or uneven clustering of the material.
By forcefully occupying the entire pore structure, the vacuum process guarantees a uniform distribution of the precursor throughout the nanoparticle.
Understanding the Constraints
The Importance of Controlled Pressure
The primary reference highlights the need for a controlled negative pressure environment.
If the vacuum is too aggressive, it may cause solvent evaporation rather than infiltration, altering the concentration of the precursor.
Conversely, insufficient vacuum will fail to fully evacuate the smallest pores, reducing the loading efficiency.
Making the Right Choice for Your Goal
- If your primary focus is material quality: Prioritize a gradual, controlled vacuum phase to ensure total air evacuation without disrupting the precursor concentration.
- If your primary focus is process speed: Understand that rushing this step will likely result in lower loading volumes and less active surface area in your final product.
The vacuum oven transforms the infiltration process from a passive soak into an active, high-efficiency injection mechanism.
Summary Table:
| Mechanism Phase | Function of Vacuum Oven | Benefit for CsPbCl3 Synthesis |
|---|---|---|
| Evacuation | Removes trapped air from MSN nanopores | Eliminates physical barriers to entry |
| Pressure Differential | Creates a negative pressure driving force | Ensures deep penetration into the silica structure |
| Infiltration | Sucks precursor solution into internal voids | Maximizes loading volume and active surface area |
| Stabilization | Maintains controlled environment | Guarantees uniform distribution and stoichiometry |
Elevate Your Nanomaterial Synthesis with KINTEK
Precision is paramount when working with sensitive CsPbCl3 precursors and complex silica structures. Backed by expert R&D and manufacturing, KINTEK offers high-performance Vacuum systems, Muffle, and Tube furnaces designed to deliver the exact pressure control and thermal stability your research demands. Whether you require a standard solution or a system fully customizable for unique nanoreactor needs, our equipment ensures high loading efficiency and material uniformity.
Ready to optimize your lab’s infiltration process? Contact KINTEK today to find the perfect vacuum oven for your application.
Visual Guide
References
- Jiaze Wu, Kai Huang. Generative Synthesis of Highly Stable Perovskite Nanocrystals via Mesoporous Silica for Full‐Spectrum White LED. DOI: 10.1002/smll.202507240
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 2200 ℃ Tungsten Vacuum Heat Treat and Sintering Furnace
- Vacuum Heat Treat Furnace with Ceramic Fiber Liner
- Small Vacuum Heat Treat and Tungsten Wire Sintering Furnace
- Vacuum Heat Treat Sintering and Brazing Furnace
- Vacuum Heat Treat Sintering Furnace Molybdenum Wire Vacuum Sintering Furnace
People Also Ask
- What functions does glucose perform in lithium-ion sieve synthesis? Enhance Carbothermal Reduction for LiMnO2 Purity
- What is the function of vacuum & heating in aluminum degassing? Enhancing Composite Integrity & Density
- Why is multiple remelting necessary for Bi-Sb alloys? Achieve Perfect Compositional Uniformity Today
- Why is strict control of vacuum pressure essential during EB-PBF of Ti–6Al–4V? Ensuring Purity and Beam Precision
- Why is electromagnetic stirring necessary for NbC-Cr7C3@graphene in W18Cr4V steel? Boost Alloy Performance



















