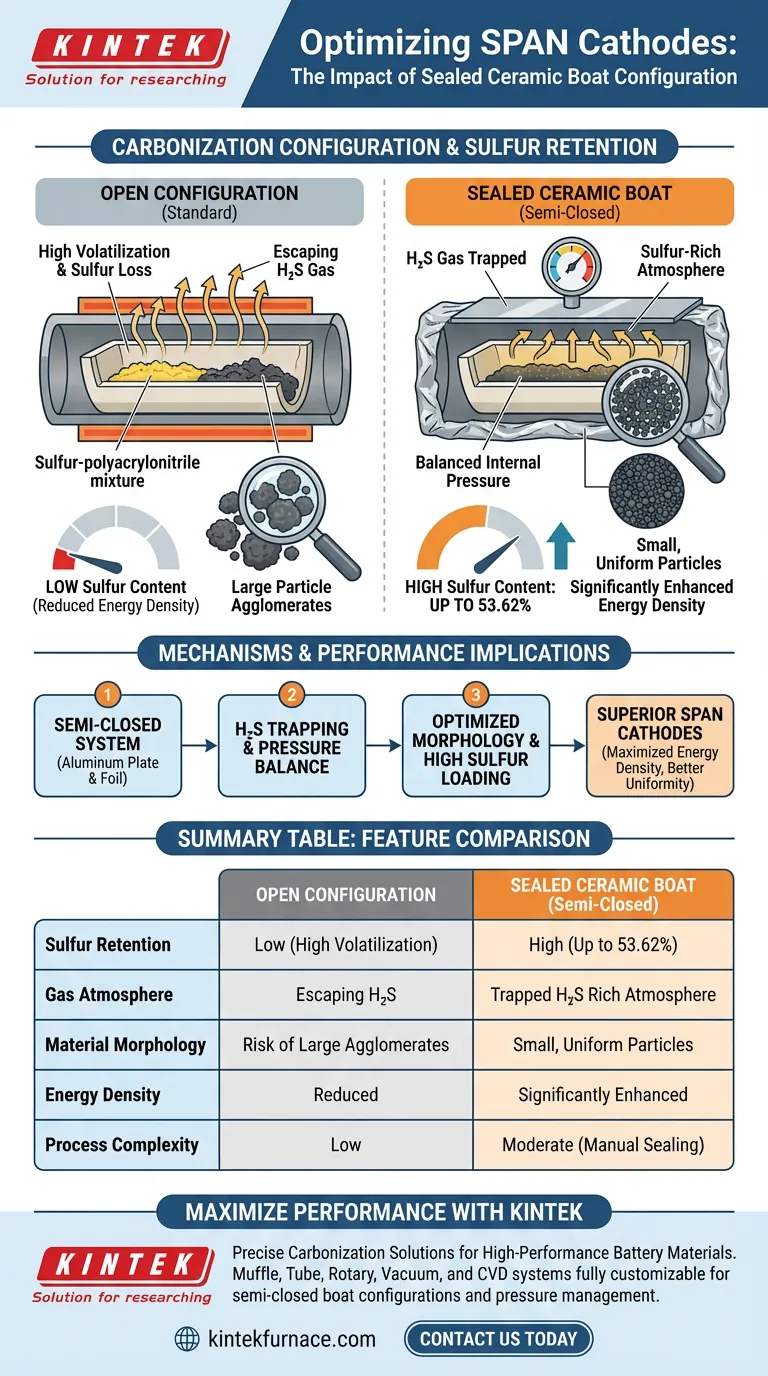
Implementing a sealed ceramic boat configuration directly increases the retained sulfur content of sulfurized polyacrylonitrile (SPAN) cathode materials during carbonization. By covering the boat with a sealed aluminum plate and wrapping it in aluminum foil, you create a semi-closed environment that significantly reduces the loss of active sulfur, enabling final sulfur contents to reach as high as 53.62%.
The semi-closed reaction environment effectively traps evolved hydrogen sulfide gas and maintains an internal pressure balance. This mechanism inhibits the formation of large particle agglomerates and maximizes sulfur retention, directly enhancing the energy density of the final battery cell.
The Mechanics of the Sealed Configuration
Creating a Semi-Closed System
The standard carbonization process often leads to the volatilization of sulfur, reducing the active material available for electrochemical reactions.
By utilizing a ceramic boat covered with an aluminum plate and wrapped in foil, you transform an open system into a semi-closed reaction environment. This physical containment is the primary driver for increased sulfur retention.
Trapping Hydrogen Sulfide ($H_2S$)
During the high-temperature reaction between sulfur and polyacrylonitrile, hydrogen sulfide gas is produced as a byproduct.
In an open system, this gas escapes immediately. In the sealed configuration, the capture of hydrogen sulfide creates a sulfur-rich atmosphere within the vessel, which suppresses further volatilization of the active sulfur from the SPAN structure.
Optimizing Internal Pressure
The sealed design does more than just trap gas; it establishes a critical internal pressure balance.
This pressure plays a physical role in shaping the material. It effectively inhibits the formation of large particle agglomerates, leading to a more favorable morphology for battery applications.
Performance Implications
Enhancing Energy Density
The direct correlation between sulfur content and battery capacity is well-established.
By reducing the loss of active sulfur, this configuration elevates the sulfur content to 53.62%. This increase translates directly to a significant enhancement in the energy density of the resulting battery.
Improving Material Uniformity
The prevention of agglomeration ensures a more uniform particle distribution.
Smaller, non-agglomerated particles generally offer better electrolyte contact and shorter ion diffusion paths, complementing the high sulfur loading to improve overall electrochemical performance.
Understanding the Trade-offs
Managing Safety and Pressure
While trapping gases improves performance, it introduces a variable of internal pressure that must be managed.
Operators must ensure the "semi-closed" nature allows for safety release if pressure becomes critical, avoiding the dangers of a fully pressurized vessel in a high-temperature furnace.
Process Complexity
This method introduces additional manual steps compared to standard open-boat carbonization.
The requirement to seal plates and wrap foil adds time to the manufacturing process, which may impact scalability or throughput in larger production environments.
Making the Right Choice for Your Goal
To apply this configuration effectively, consider your primary synthesis objectives:
- If your primary focus is Maximizing Energy Density: Adopt the sealed aluminum foil/plate method to drive sulfur content toward the 53% threshold.
- If your primary focus is Morphology Control: Utilize the internal pressure of the sealed boat to prevent large particle agglomerates without needing complex chemical additives.
Leveraging a semi-closed environment is a mechanical solution to a chemical problem, offering a high-impact pathway to superior SPAN cathodes.
Summary Table:
| Feature | Open Configuration | Sealed Ceramic Boat (Semi-Closed) |
|---|---|---|
| Sulfur Retention | Low (High Volatilization) | High (Up to 53.62%) |
| Gas Atmosphere | Escaping H2S | Trapped H2S Rich Atmosphere |
| Material Morphology | Risk of Large Agglomerates | Small, Uniform Particles |
| Energy Density | Reduced | Significantly Enhanced |
| Process Complexity | Low | Moderate (Manual Sealing) |
Maximize Your SPAN Cathode Performance with KINTEK
Precise control over the carbonization environment is critical for high-performance battery materials. At KINTEK, we understand that achieving a 53.62% sulfur retention requires more than just a furnace—it requires a system designed for precision and reliability.
Backed by expert R&D and manufacturing, KINTEK offers a wide range of Muffle, Tube, Rotary, Vacuum, and CVD systems, all of which are fully customizable to accommodate semi-closed boat configurations and internal pressure management. Whether you are scaling up production or refining laboratory synthesis, our high-temperature solutions provide the thermal stability and atmospheric control needed to prevent agglomeration and maximize energy density.
Ready to optimize your material synthesis? Contact us today to discuss your unique lab furnace needs!
Visual Guide
References
- Krishna Kumar Sarode, Vibha Kalra. Solid–liquid–solid mediated artificial SEI coated stable lithium and high-sulfur percentage SPAN for high performance Li–S batteries. DOI: 10.1039/d3ya00423f
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Vacuum Dental Porcelain Sintering Furnace for Dental Laboratories
- 1400℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- 1700℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- Stainless Steel Quick Release Vacuum Chain Three Section Clamp
- Molybdenum Disilicide MoSi2 Thermal Heating Elements for Electric Furnace
People Also Ask
- What are the risks of inadequate dental furnace ventilation? Protect Your Lab Quality & Team Safety
- What is the use of porcelain in dentistry? Achieve Lifelike, Durable Dental Restorations
- What is the main purpose of a sintering furnace in dentistry? Transform Zirconia into Strong Dental Restorations
- Why are synthesized CdS nanorods dried in a laboratory vacuum oven? Preserving Nanostructure and Chemical Integrity
- How does an efficient cooling system benefit dental furnace operations? Boost Productivity and Quality in Your Lab



















