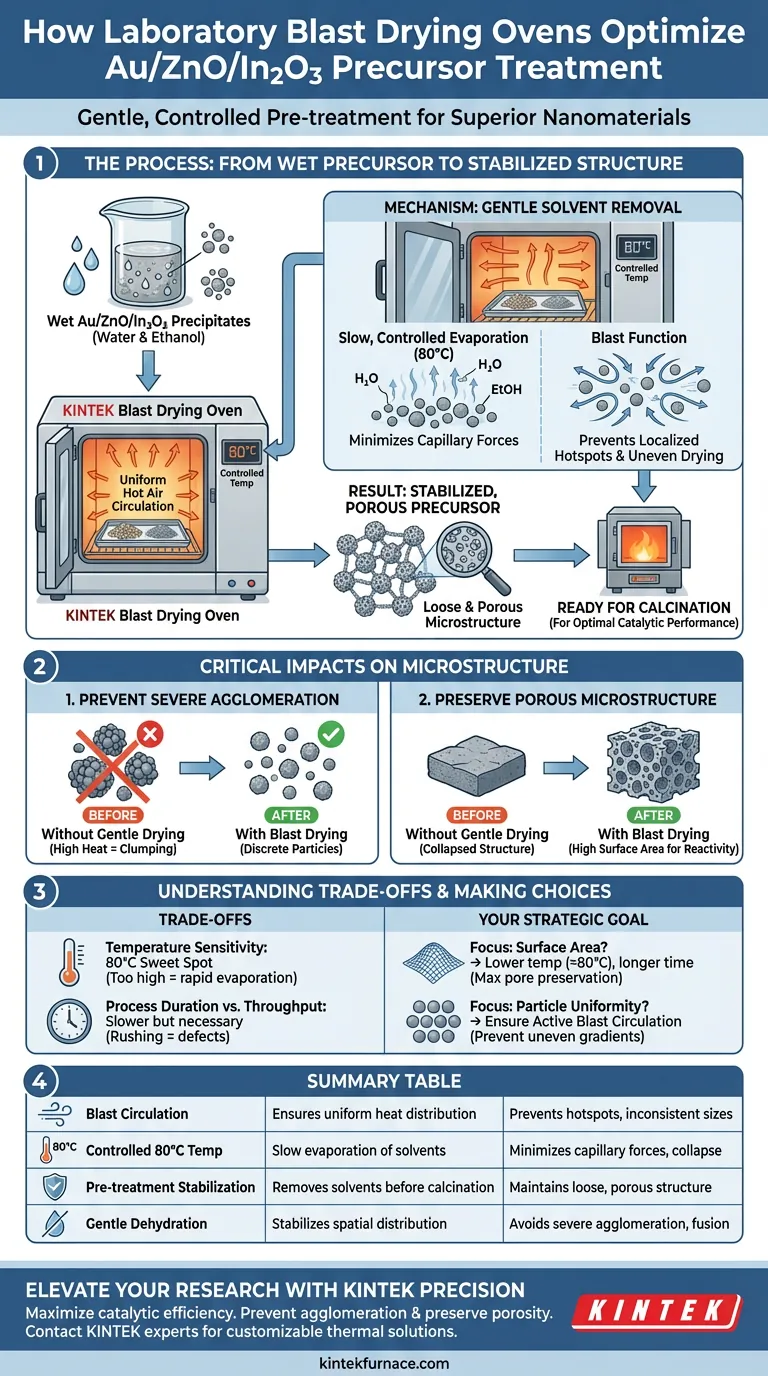
The laboratory blast drying oven facilitates the treatment of Au/ZnO/In2O3 precursor precipitates by utilizing uniform hot air circulation to gently remove adsorbed moisture and ethanol at controlled, moderate temperatures, typically around 80 °C. This specific thermal environment serves as a vital pre-treatment stage, stabilizing the delicate nano-precursors before they are subjected to high-temperature calcination.
The core function of this process is to prevent the severe particle agglomeration that occurs during rapid liquid evaporation. By removing solvents slowly, the oven ensures the material maintains the loose, porous microstructure necessary for optimal catalytic performance.

The Mechanics of Pre-Treatment Stabilization
Uniform Solvent Removal
The primary role of the blast drying oven is to eliminate residual solvents—specifically water and ethanol—remaining from the synthesis and washing phases.
Unlike static drying, the "blast" function ensures hot air circulates continuously around the sample. This prevents localized hotspots and ensures that moisture is removed evenly from the entire batch of precipitates.
Preventing Capillary Collapse
When solvents evaporate too quickly, significant capillary forces are generated between nanoparticles.
If the material is subjected immediately to high heat, these forces pull particles together tightly. The blast drying oven mitigates this by allowing for slow, controlled evaporation, effectively minimizing the physical stress placed on the precursor structure.
Impact on Material Microstructure
Avoiding Severe Agglomeration
The most critical risk during the preparation of Au/ZnO/In2O3 is the clumping of nanoparticles.
Primary references indicate that without this gentle drying step at 80 °C, the subsequent high-temperature calcination would cause "severe agglomeration." The drying oven ensures that the particles remain discrete rather than fusing into large, dense masses.
Preserving Porosity
The ultimate goal of this treatment is to maintain a "loose and porous microstructure."
By stabilizing the spatial distribution of the metal precursors early, the oven prevents component migration. This ensures that when the material is eventually calcined, the resulting structure retains the high surface area required for effective chemical reactivity.
Understanding the Trade-offs
Temperature Sensitivity
While the blast drying oven is effective, selecting the correct temperature is paramount.
Setting the temperature too high (e.g., approaching calcination temperatures prematurely) can trigger rapid evaporation, defeating the purpose of the step. For Au/ZnO/In2O3 precursors, 80 °C is the established sweet spot for balancing drying speed with structural protection.
Process Duration vs. Throughput
This gentle drying process is inherently slower than high-heat methods.
It requires an extended period to fully remove solvents from the pores of the support. Rushing this stage by increasing heat or shortening time can lead to trapped moisture, which may cause structural defects or "pop-outs" during the final calcination.
Making the Right Choice for Your Goal
To maximize the effectiveness of your precursor treatment, consider the following based on your specific objectives:
- If your primary focus is surface area: Prioritize a lower temperature setting (approx. 80 °C) and longer duration to maximize pore preservation and minimize agglomeration.
- If your primary focus is particle uniformity: Ensure the blast circulation feature is active to prevent uneven drying gradients that lead to inconsistent particle sizes.
This drying step is not merely about removing water; it is the architectural foundation that defines the final porosity and efficiency of your nanomaterial.
Summary Table:
| Feature | Function in Au/ZnO/In2O3 Treatment | Impact on Final Material |
|---|---|---|
| Blast Circulation | Ensures uniform heat distribution | Prevents localized hotspots and inconsistent particle sizes |
| Controlled 80°C Temp | Slow evaporation of water/ethanol | Minimizes capillary forces and prevents structural collapse |
| Pre-treatment Stabilization | Removes solvents before calcination | Maintains a loose, porous microstructure and high surface area |
| Gentle Dehydration | Stabilizes spatial distribution | Avoids severe agglomeration and fusion of nanoparticles |
Elevate Your Material Research with KINTEK Precision
Maximize the catalytic efficiency of your nanomaterials by ensuring perfect pre-treatment conditions. Backed by expert R&D and manufacturing, KINTEK offers high-performance laboratory blast drying ovens, Muffle, Tube, Rotary, and Vacuum systems—all fully customizable to meet the exacting temperature and uniformity requirements of your unique research.
Ready to prevent nanoparticle agglomeration and preserve your material's porosity?
Contact KINTEK experts today to find your ideal thermal solution.
Visual Guide
References
- Yuhong Zhang, Hang Liu. Au/ZnO/In<sub>2</sub>O<sub>3</sub> nanoparticles for enhanced isopropanol gas sensing performance. DOI: 10.1039/d3ra07507a
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Laboratory Muffle Oven Furnace with Bottom Lifting
- 1800℃ High Temperature Muffle Oven Furnace for Laboratory
- 1200℃ Muffle Oven Furnace for Laboratory
- High Temperature Muffle Oven Furnace for Laboratory Debinding and Pre Sintering
- 1700℃ High Temperature Muffle Oven Furnace for Laboratory
People Also Ask
- How is a laboratory muffle furnace utilized for metal phosphomolybdate catalysts? Achieve Precise Thermal Stabilization
- What role does a muffle furnace play in 600°C carbonization of palm shells? Unlock High-Performance Activated Carbon
- What is the function of a laboratory muffle furnace in the carbonization process? Transform Waste into Nanosheets
- What is the core role of a laboratory muffle furnace in rice husk-based biochar? Master Your Pyrolysis Process
- What role does a laboratory muffle furnace play in the ash analysis of plant samples? Achieve Clean Mineral Isolation






