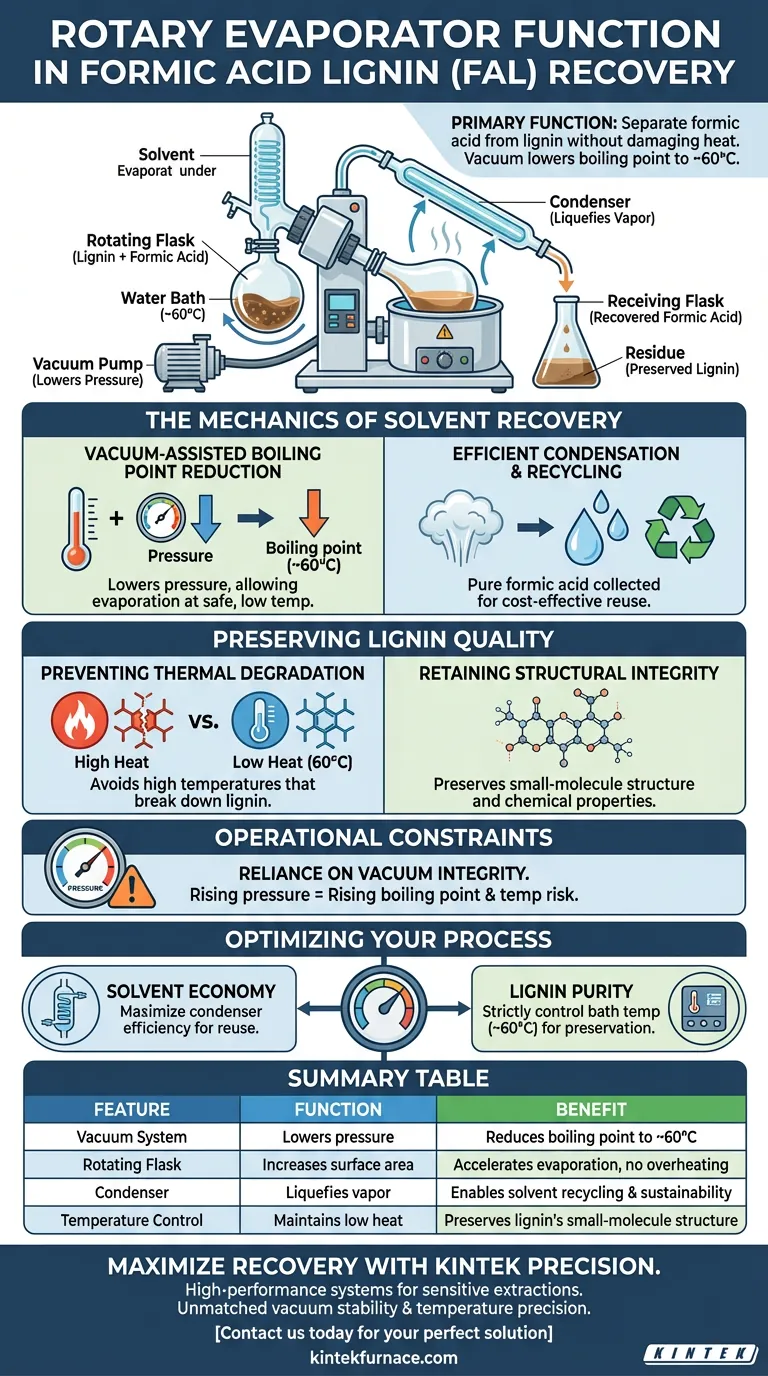
The primary function of a rotary evaporator in this context is to separate formic acid from the extracted lignin without using damaging levels of heat. By creating a vacuum within the system, the device lowers the atmospheric pressure, which significantly reduces the boiling point of the formic acid solvent. This allows the solvent to evaporate and condense for recovery at a relatively low temperature, typically around 60 °C.
In the recovery of formic acid lignin (FAL), the rotary evaporator is essential for balancing solvent recycling with product preservation. It permits the removal of high-boiling-point solvents under vacuum conditions, preventing the thermal degradation that would otherwise destroy the lignin's characteristic molecular structure.
The Mechanics of Solvent Recovery
Vacuum-Assisted Boiling Point Reduction
Formic acid is a high-boiling-point solvent, meaning it requires significant energy to vaporize under normal atmospheric pressure.
The rotary evaporator solves this by lowering the pressure inside the system.
This pressure drop allows the formic acid to boil and evaporate at much lower temperatures, specifically enabling operation at approximately 60 °C.
Efficient Condensation and Recycling
Once the solvent evaporates, it travels to a condenser where it returns to a liquid state.
This process allows for the efficient collection of pure formic acid.
Recovering the solvent in this manner makes the process more sustainable and cost-effective by enabling it to be recycled for future extractions.
Preserving Lignin Quality
Preventing Thermal Degradation
The most critical role of the rotary evaporator is protecting the lignin from heat damage.
If the solvent were removed using standard heating methods at atmospheric pressure, the high temperatures required would degrade the lignin.
By operating at a cooler 60 °C, the rotary evaporator ensures the extraction process remains gentle.
Retaining Structural Integrity
Formic acid lignin (FAL) possesses a specific, valuable characteristic: a small-molecule structure.
High-temperature exposure can alter or destroy these delicate molecular bonds.
Using vacuum evaporation preserves this small-molecule structure, ensuring the recovered lignin retains its desired chemical properties and quality.
Understanding the Operational Constraints
Reliance on Vacuum Integrity
The success of this recovery method is entirely dependent on maintaining a consistent vacuum.
If the system pressure rises, the boiling point of the formic acid will increase immediately.
This would force the operator to raise the bath temperature to maintain evaporation rates, inadvertently risking the thermal degradation of the lignin you are trying to protect.
Optimizing Your Lignin Recovery Process
To maximize the yield and quality of your formic acid lignin, align your equipment settings with your specific recovery goals.
- If your primary focus is Solvent Economy: Ensure your condenser is operating at peak efficiency to capture the maximum amount of formic acid vapor for reuse.
- If your primary focus is Lignin Purity: Strictly monitor the bath temperature to ensure it does not exceed 60 °C, prioritizing the preservation of the small-molecule structure over evaporation speed.
By controlling pressure to manage temperature, you ensure the integrity of your material is never compromised by the extraction process.
Summary Table:
| Feature | Function in FAL Recovery | Benefit to Process |
|---|---|---|
| Vacuum System | Lowers atmospheric pressure | Reduces boiling point of formic acid to ~60°C |
| Rotating Flask | Increases surface area | Accelerates solvent evaporation without overheating |
| Condenser | Liquefies formic acid vapor | Enables solvent recycling and sustainability |
| Temperature Control | Maintains low heat levels | Preserves lignin's small-molecule structure |
Maximize Your Lignin Recovery with KINTEK Precision
Don't let high temperatures compromise your material integrity. KINTEK provides high-performance rotary evaporators designed to handle sensitive extractions like formic acid lignin recovery with unmatched vacuum stability and temperature precision.
Backed by expert R&D and manufacturing, we offer customizable Rotary, Vacuum, Muffle, Tube, and CVD systems tailored to your specific lab requirements. Whether you are focused on maximizing solvent economy or ensuring the highest lignin purity, our equipment delivers the control you need.
Ready to optimize your extraction workflow? Contact us today to find your perfect solution.
Visual Guide
References
- Effects of Multiple Treatments of Formic Acid on the Chemical Properties and Structural Features of Bamboo Powder. DOI: 10.3390/molecules30020398
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Electric Rotary Kiln Small Rotary Furnace Biomass Pyrolysis Plant Rotating Furnace
- Electric Rotary Kiln Continuous Working Small Rotary Furnace Kiln for Pyrolysis Plant Heating
- Inclined Rotary Plasma Enhanced Chemical Deposition PECVD Tube Furnace Machine
- Electric Rotary Kiln Pyrolysis Furnace Plant Machine Small Rotary Kiln Calciner
- Electric Rotary Kiln Small Rotary Furnace for Activated Carbon Regeneration
People Also Ask
- What are the key components of a rotary kiln electric furnace? Unlock Efficient Thermal Processing
- What factors are critical in determining the temperature profile for a rotary kiln? Optimize Heat Control for Peak Performance
- What are the differences between direct-heat and indirect-heat rotary kilns? Choose the Right Kiln for Your Process
- How does rotational speed influence the thermal efficiency of a rotary kiln? Optimize Heat Transfer and Save Energy
- What are the key components of a rotary kiln's construction? Discover the Essential Parts for Efficient Industrial Processing









