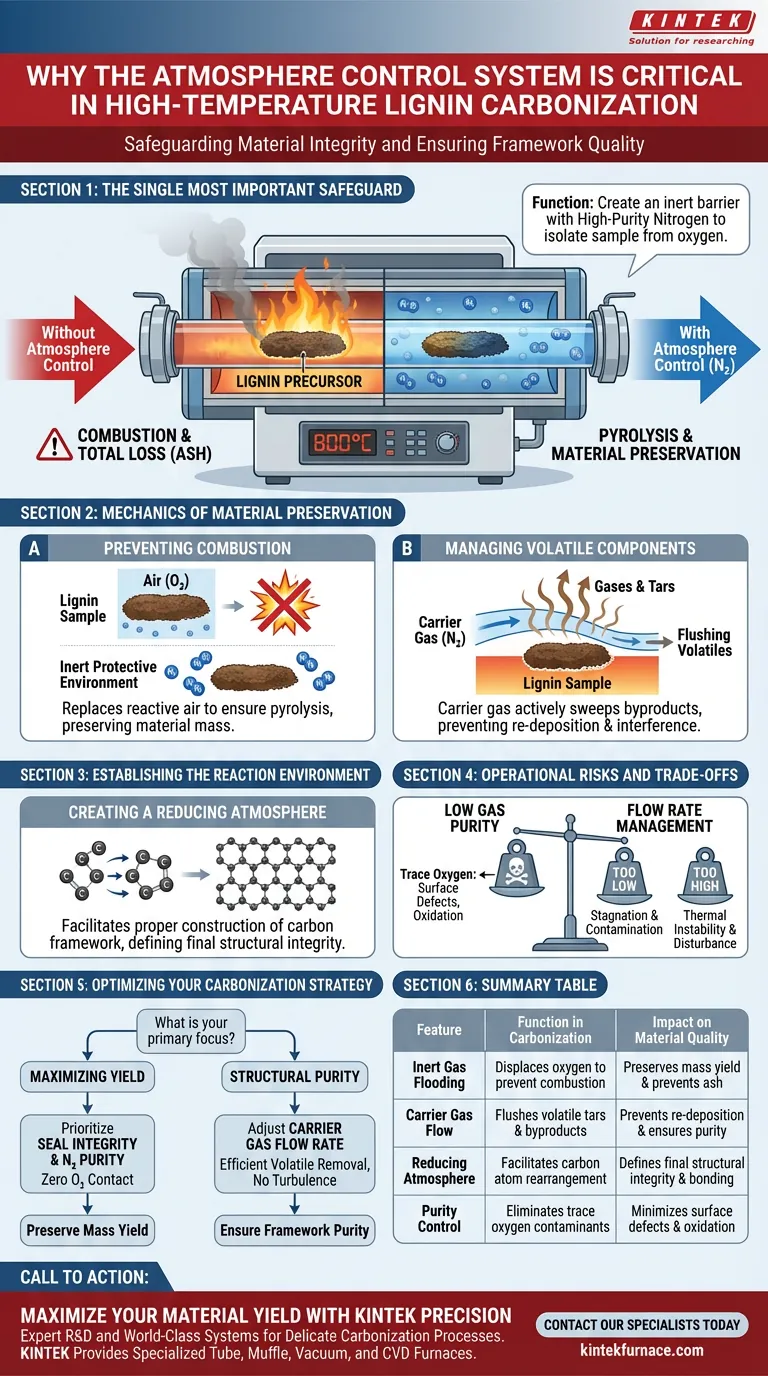
The atmosphere control system is the single most important safeguard against material destruction during high-temperature processing. Its primary function is to continuously flood the furnace chamber with high-purity nitrogen, creating an inert barrier that isolates the sample from the outside air. Without this specific control, exposing lignin-based precursors to temperatures like 800°C would result in immediate combustion and total material loss.
The atmosphere control system acts as both a shield and a transport mechanism. It prevents oxygen-induced combustion to preserve yield, while simultaneously acting as a carrier gas to flush away volatile byproducts, ensuring the carbon framework forms correctly.

The Mechanics of Material Preservation
Preventing Combustion and Loss
The fundamental risk when heating organic materials like lignin is oxidation. At high temperatures (e.g., 800°C), the presence of even trace amounts of oxygen triggers combustion.
The atmosphere control system mitigates this by replacing reactive air with an inert protective environment. This ensures that the precursor undergoes pyrolysis (chemical decomposition by heat) rather than burning, preserving the mass of the material.
Managing Volatile Components
Carbonization is a transformative process that releases gases and tars as the material degrades. The nitrogen supply serves a dual purpose by acting as a carrier gas.
It actively sweeps these generated volatile components out of the heating zone. Removing these byproducts is essential to prevent them from re-depositing on the sample or interfering with the heating uniformity.
Establishing the Reaction Environment
Creating a Reducing Atmosphere
Successful carbonization requires more than just the absence of oxygen; it requires a specific chemical state known as a reducing atmosphere.
By maintaining this environment, the system allows the carbon atoms to rearrange and bond effectively. This facilitates the proper construction of the carbon material's framework, defining the final structural integrity of the product.
Operational Risks and Trade-offs
The Impact of Gas Purity
While the system is designed to protect the sample, the quality of the output is strictly limited by the purity of the input gas. Using nitrogen with low purity introduces trace oxygen, which can cause surface defects or partial oxidation of the carbon framework.
Flow Rate Management
There is a critical balance to be maintained regarding the flow rate of the carrier gas. If the flow is too low, volatile components may stagnate and contaminate the carbon structure. However, excessively high flow rates can disrupt thermal stability or physically disturb delicate precursor structures.
Optimizing Your Carbonization Strategy
To achieve consistent, high-quality carbon materials, you must view atmosphere control as an active variable, not just a passive safety feature.
- If your primary focus is Maximizing Yield: Prioritize the absolute seal integrity and nitrogen purity to ensure zero oxygen contact at peak temperatures.
- If your primary focus is Structural Purity: Adjust the carrier gas flow rate to ensure efficient removal of volatiles without creating thermal turbulence.
Precision in atmosphere control is the difference between producing high-grade carbon and producing ash.
Summary Table:
| Feature | Function in Carbonization | Impact on Material Quality |
|---|---|---|
| Inert Gas Flooding | Displaces oxygen to prevent combustion | Preserves mass yield and prevents ash formation |
| Carrier Gas Flow | Flushes volatile tars and byproducts | Prevents re-deposition and ensures framework purity |
| Reducing Atmosphere | Facilitates carbon atom rearrangement | Defines final structural integrity and bonding |
| Purity Control | Eliminates trace oxygen contaminants | Minimizes surface defects and oxidation |
Maximize Your Material Yield with KINTEK Precision
Don't let poor atmosphere control turn your research into ash. Backed by expert R&D and world-class manufacturing, KINTEK provides specialized Tube, Muffle, Vacuum, and CVD systems designed to handle the most delicate carbonization processes. Our high-temperature furnaces offer the precise gas flow and purity controls needed to safeguard your lignin-based precursors.
Ready to elevate your thermal processing? Contact our specialists today to discuss a customizable furnace solution tailored to your unique lab requirements.
Visual Guide
References
- Yuebin Xi, Binpeng Zhang. Production of Lignin-Derived Functional Material for Efficient Electromagnetic Wave Absorption with an Ultralow Filler Ratio. DOI: 10.3390/polym16020201
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 1400℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- 1700℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- 1200℃ Split Tube Furnace Laboratory Quartz Tube Furnace with Quartz Tube
- Vertical Laboratory Quartz Tube Furnace Tubular Furnace
- 1700℃ High Temperature Muffle Oven Furnace for Laboratory
People Also Ask
- How does a laboratory high-temperature tube furnace contribute to the conversion of electrospun fibers? Expert Insights
- What factors should be considered when selecting a high temperature tube furnace? Ensure Precision and Reliability for Your Lab
- What is the function of a furnace in CuAlMn alloy treatment? Achieve Perfect Microstructural Homogenization
- What is a high temperature tube furnace? Achieve Precise Heat and Atmosphere Control
- How do high-temperature laboratory tube furnaces ensure environmental stability? Precision Thermal Reduction Tips



















