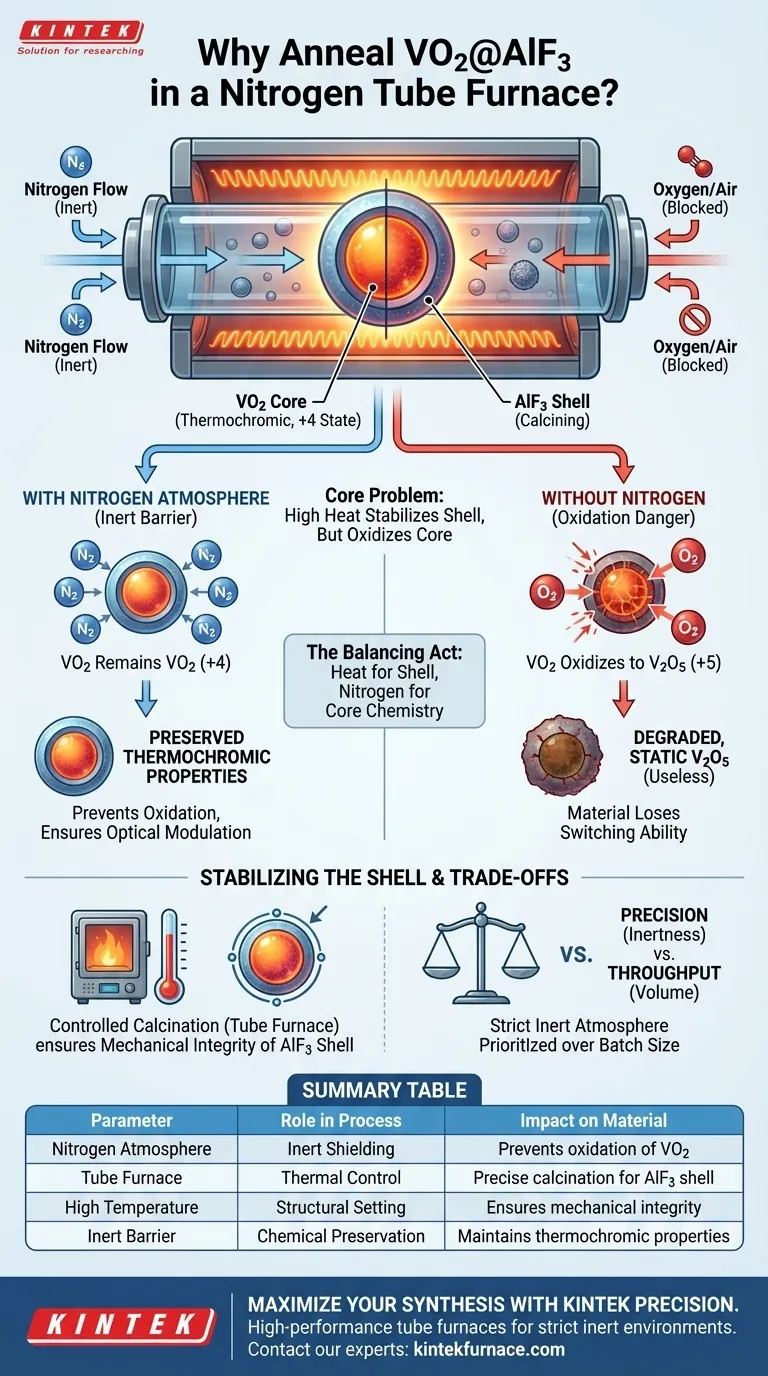
Annealing in a tube furnace with a nitrogen atmosphere is critical for stabilizing the protective coating of the material without compromising its active core. This process utilizes precise high-temperature control to calcine the aluminum fluoride ($AlF_3$) shell while the inert nitrogen environment strictly prevents the vanadium dioxide ($VO_2$) core from oxidizing into unwanted chemical states.
Core Takeaway: This specific annealing setup solves a fundamental contradiction: it applies the high heat necessary to stabilize the outer shell while simultaneously shielding the inner core from oxygen to preserve its thermochromic capabilities.

Protecting the Core Chemistry
The primary challenge in preparing $VO_2@AlF_3$ powder is that the active core is highly sensitive to oxygen at the temperatures required for synthesis.
The Danger of Oxidation
At high temperatures, vanadium is prone to changing its oxidation state. Without a protective atmosphere, the $+4$ oxidation state of vanadium in the $VO_2$ core will oxidize to the $+5$ state.
Preventing Unwanted Phase Changes
This oxidation reaction converts the desirable vanadium dioxide ($VO_2$) into vanadium pentoxide ($V_2O_5$). Nitrogen acts as an inert barrier, displacing oxygen and effectively blocking this chemical reaction.
Preserving Critical Properties
The utility of this material relies entirely on the specific properties of the $VO_2$ core. If it oxidizes to $V_2O_5$, the material loses its thermochromic phase transition properties. Nitrogen ensures the material retains its ability to modulate optical signals.
Stabilizing the Outer Shell
While the nitrogen protects the core, the tube furnace provides the thermal environment necessary to finalize the composite structure.
Controlled Calcination
The $AlF_3$ coating layer requires calcination to become stable. The tube furnace allows for a precisely controlled high-temperature environment to facilitate this process.
Ensuring Durability
By subjecting the powder to this specific heat treatment, the coating layer is properly set. This stabilization is essential for the mechanical integrity of the core-shell structure.
Understanding the Trade-offs
The process requires a strict balance between thermal requirements and chemical isolation.
The Consequence of Atmosphere Leaks
If the nitrogen atmosphere is compromised even slightly, the high heat will accelerate oxidation. This results in a material that is structurally sound (the shell is calcined) but functionally useless (the core is degraded).
Precision vs. Throughput
Using a tube furnace allows for high precision but often processes smaller batches compared to industrial kilns. This trade-off is necessary because maintaining a strictly inert atmosphere is prioritized over volume to guarantee the survival of the $VO_2$ phase.
Making the Right Choice for Your Goal
To ensure the success of your $VO_2@AlF_3$ preparation, prioritize the parameters below based on your specific objectives.
- If your primary focus is Optical Performance: Ensure the nitrogen flow is continuous and the seal is perfect to prevent the formation of static, non-switching $V_2O_5$.
- If your primary focus is Structural Stability: adhere strictly to the required calcination temperatures to fully stabilize the $AlF_3$ shell against environmental wear.
Success in this synthesis depends on using heat to build the shell while using nitrogen to freeze the chemistry of the core.
Summary Table:
| Parameter | Role in Process | Impact on Material |
|---|---|---|
| Nitrogen Atmosphere | Inert Shielding | Prevents oxidation of $VO_2$ (+4) to $V_2O_5$ (+5) |
| Tube Furnace | Thermal Control | Provides precise calcination for the $AlF_3$ shell |
| High Temperature | Structural Setting | Ensures mechanical integrity and durability of the coating |
| Inert Barrier | Chemical Preservation | Maintains thermochromic phase transition properties |
Maximize Your Material Synthesis with KINTEK Precision
Don't let oxidation compromise your core-shell research. KINTEK provides high-performance tube and vacuum furnace systems specifically designed to maintain the strict inert environments required for sensitive chemical transitions. Backed by expert R&D and manufacturing, we offer customizable high-temp solutions—including Tube, Muffle, Rotary, and CVD systems—to ensure your $VO_2$ particles retain their critical thermochromic properties.
Ready to achieve perfect annealing results? Contact our technical experts today to find the ideal furnace for your unique lab requirements!
Visual Guide
References
- Lingfeng Jiang, Li Zhao. Chemical Deposition Method for Preparing VO2@AlF3 Core–Shell-Structured Nanospheres for Smart Temperature-Control Coating. DOI: 10.3390/coatings15091045
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 1200℃ Controlled Inert Nitrogen Atmosphere Furnace
- Controlled Inert Nitrogen Hydrogen Atmosphere Furnace
- 1400℃ Controlled Inert Nitrogen Atmosphere Furnace
- Mesh Belt Controlled Atmosphere Furnace Inert Nitrogen Atmosphere Furnace
- 1700℃ Controlled Inert Nitrogen Atmosphere Furnace
People Also Ask
- How does a controlled atmosphere furnace prevent oxidation and decarburization? Master Precision Heat Treatment
- Why is a controlled atmosphere furnace required for 316L debinding? Ensure Structural Integrity & Zero Cracks
- What are the advantages of a controlled atmosphere furnace over tube furnaces? Superior Process Control for Sensitive Materials
- What types of gases are used in controlled atmosphere furnaces? Optimize Material Protection and Transformation
- What are the operational advantages of using a controlled atmosphere furnace? Boost Quality and Efficiency in Heat Treatment



















