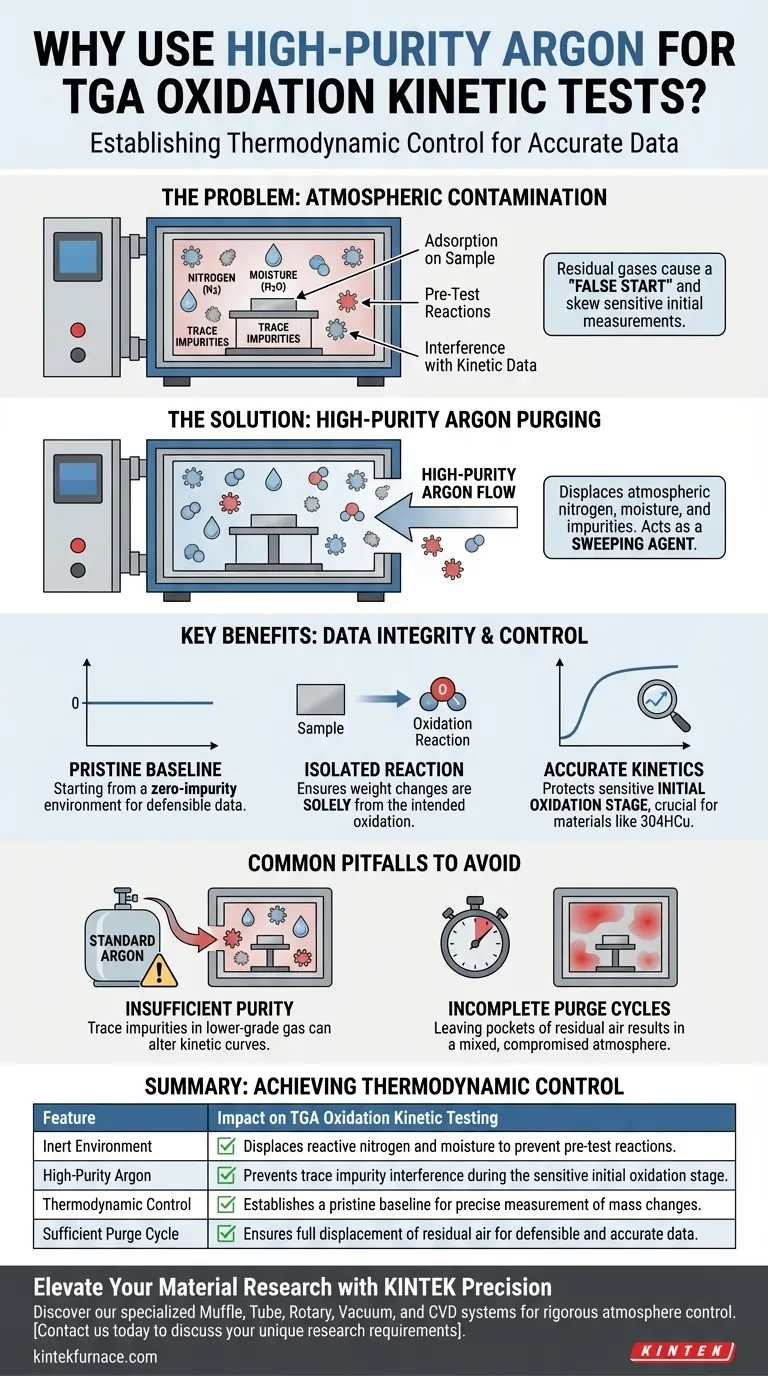
High-purity argon gas is the standard for establishing a pristine baseline environment within a thermogravimetric analyzer (TGA). By flooding the heating furnace chamber with this inert gas prior to testing, you displace atmospheric nitrogen, moisture, and trace impurities that would otherwise skew the delicate measurements of initial oxidation kinetics.
Core Takeaway: The goal of purging is not just cleaning; it is about establishing thermodynamic control. By eliminating reactive atmospheric variables, argon purging ensures that subsequent weight changes are attributable solely to the intended oxidation reaction, preserving the integrity of your kinetic data.

The Physics of Atmosphere Control
Displacing Atmospheric Contaminants
The air in a standard furnace chamber contains a mixture of gases, primarily nitrogen and oxygen, along with variable amounts of moisture.
For high-sensitivity experiments, these are unwanted variables. High-purity argon acts as a sweeping agent, physically pushing these atmospheric components out of the chamber.
Why Moisture and Nitrogen Interference Matters
Moisture is particularly problematic because it can adsorb onto the sample surface or react with the material even at lower temperatures.
Nitrogen, while generally stable, can interfere with specific reactions or act as a thermal insulator in ways that differ from the test gas. Removing these ensures the environment is strictly controlled.
Securing Kinetic Data Integrity
Isolating the Oxidation Reaction
Thermogravimetric oxidation kinetic tests measure precise changes in mass as a material reacts with oxygen over time.
If the chamber is not purged, residual gases react with the sample before the test formally begins. This creates a "false start," making it impossible to separate the intended oxidation data from background interference.
Protecting the Initial Oxidation Stage
The primary reference highlights that this step is critical for preventing interference specifically during the initial oxidation stage.
This early phase is often the most sensitive and kinetically significant. For materials like 304HCu stainless steel, accurate characterization of this initial behavior relies entirely on starting from a zero-impurity baseline.
Common Pitfalls to Avoid
The Risk of Insufficient Purity
Using standard industrial argon instead of high-purity argon can introduce the very contaminants you are trying to remove.
Trace impurities in lower-grade gas can react with the sample surface, subtly altering the kinetic curve and leading to incorrect calculations of reaction rates.
Incomplete Purging Cycles
Simply introducing argon is not enough; the volume of the chamber must be turned over multiple times.
Failing to purge for a sufficient duration leaves pockets of residual air, resulting in a mixed atmosphere that compromises the experimental controls.
Making the Right Choice for Your Goal
To ensure your thermogravimetric data is defensible and accurate, apply the following standards:
- If your primary focus is Kinetic Modeling: Ensure you use certified high-purity argon to eliminate all variables that could skew the reaction rate calculations.
- If your primary focus is Material Characterization: Verify that the purge cycle is long enough to fully displace moisture, preventing surface adsorption artifacts on sensitive alloys like 304HCu.
Control the atmosphere first, and your data will tell the true story of the material.
Summary Table:
| Feature | Impact on TGA Oxidation Kinetic Testing |
|---|---|
| Inert Environment | Displaces reactive nitrogen and moisture to prevent pre-test reactions. |
| High-Purity Argon | Prevents trace impurity interference during the sensitive initial oxidation stage. |
| Thermodynamic Control | Establishes a pristine baseline for precise measurement of mass changes. |
| Sufficient Purge Cycle | Ensures full displacement of residual air for defensible and accurate data. |
Elevate Your Material Research with KINTEK Precision
Don’t let atmospheric interference compromise your kinetic data. Backed by expert R&D and manufacturing, KINTEK offers specialized Muffle, Tube, Rotary, Vacuum, and CVD systems designed for rigorous atmosphere control. Whether you are characterizing alloys like 304HCu or performing complex TGA modeling, our customizable lab high-temp furnaces provide the stability and purity you need.
Ready to achieve superior thermal control? Contact us today to discuss your unique research requirements.
Visual Guide
References
- M.S. Archana, S. Ningshen. Initial Stage Oxidation of 304HCu Stainless Steel in Oxygen Environment. DOI: 10.1007/s12666-024-03478-7
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Split Chamber CVD Tube Furnace with Vacuum Station CVD Machine
- Vacuum Hot Press Furnace Machine Heated Vacuum Press Tube Furnace
- Vacuum Heat Treat Furnace with Ceramic Fiber Liner
- 2200 ℃ Graphite Vacuum Heat Treat Furnace
- Laboratory Quartz Tube Furnace RTP Heating Tubular Furnace
People Also Ask
- What is a CVD tube furnace and what is its primary function? Discover Precision Thin-Film Deposition
- Why are CVD tube furnace sintering systems indispensable for 2D material research and production? Unlock Atomic-Scale Precision
- What role does a Tube Furnace play in the CVD growth of carbon nanotubes? Achieve High-Purity CNT Synthesis
- What are the key application fields of CVD tube furnaces? Unlock Precision in Thin-Film Synthesis
- What are the practical applications of gate media prepared by CVD tube furnaces? Unlock Advanced Electronics and More



















