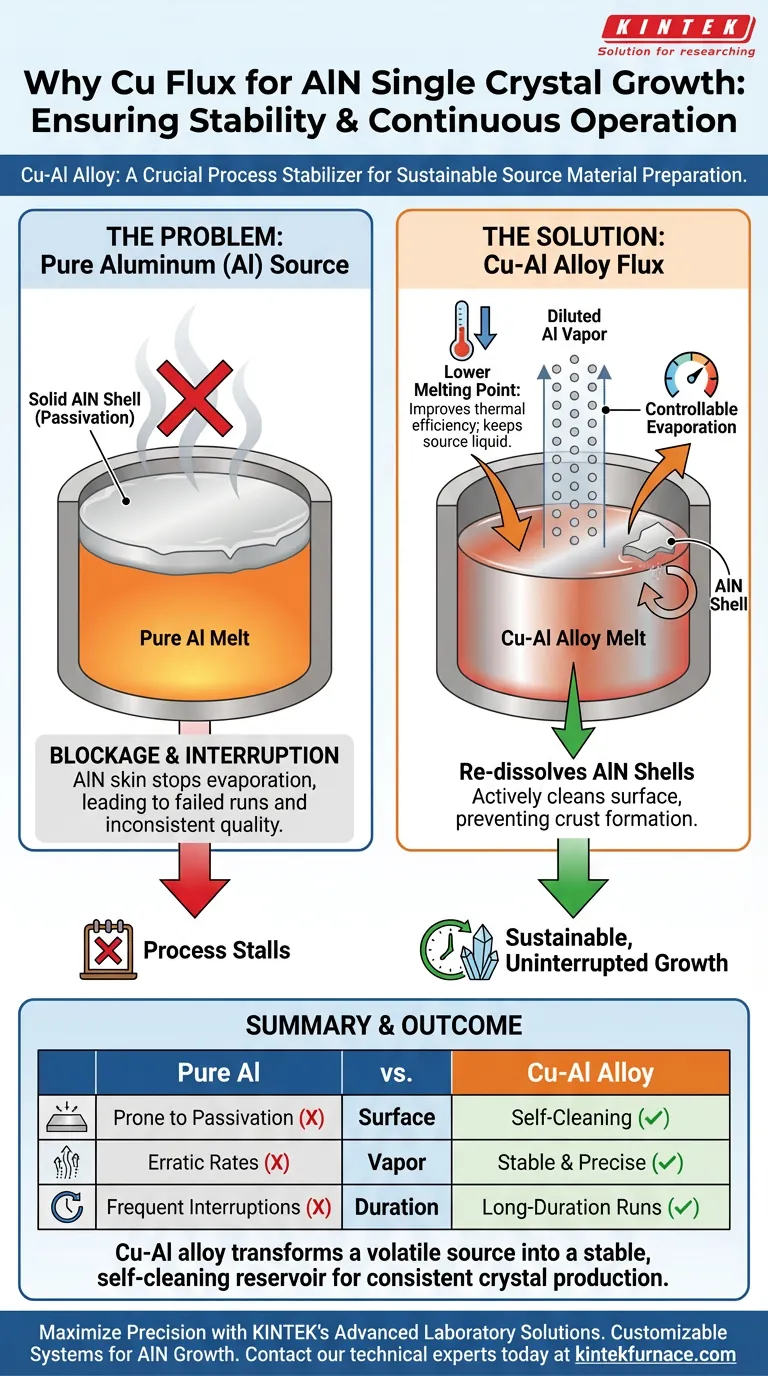
The introduction of Copper serves as a crucial process stabilizer. By alloying Copper (Cu) with the Aluminum (Al) source material, you fundamentally alter the thermodynamics of the melt to ensure continuous operation. This modification prevents physical blockages and allows for precise control over the evaporation required for crystal growth.
The use of a Cu-Al binary alloy solves the critical issue of source passivation. By lowering the melting point and actively dissolving solid surface crusts, Copper flux ensures the sustainable, unobstructed release of aluminum vapor.
Optimizing the Source Melt
To grow high-quality Aluminum Nitride (AlN) crystals, the delivery of aluminum vapor must be consistent. Pure aluminum sources present specific thermal challenges that the addition of Copper effectively mitigates.
Lowering the Melting Point
The formation of a Cu-Al binary alloy significantly changes the thermal properties of the source material. Specifically, the presence of Copper lowers the melting point of the alloy compared to pure aluminum systems.
This reduction allows the source material to remain in a liquid state at more manageable temperatures. It improves the overall thermal efficiency of the preparation phase.
Controlling Vapor Concentration
Precision is required when supplying species for crystal growth. The Cu-Al alloy acts to dilute the aluminum vapor concentration generated from the melt.
This dilution prevents excessive or erratic release of source material. It enables a highly controllable evaporation rate, which is essential for maintaining the stoichiometry of the growing crystal.
Preventing Process Interruption
The most significant technical advantage of introducing Copper is its role in maintaining the physical integrity of the evaporation surface.
Re-dissolution of AlN Shells
During the growth process, nitrogen in the system can react with the source melt to form a solid "skin" or shell of Aluminum Nitride (AlN) on the surface. If left unchecked, this solid layer blocks the liquid aluminum from evaporating.
Copper promotes the re-dissolution of these AlN shell layers back into the melt.
Ensuring Sustainable Growth
By preventing the formation of a dense, blocking AlN crust, the flux ensures the evaporation path remains open. This capability is vital for a sustainable, long-duration growth process without the need for interruption to clean or reset the source material.
Understanding the Operational Risks
While the Cu-Al alloy offers significant advantages, it is important to understand the specific operational pitfall it is designed to avoid. The primary risk in this process is source passivation.
The Consequence of Blockage
Without the dissolving action of the Copper flux, the formation of an AlN shell creates a physical barrier on top of the melt.
This barrier halts the supply of aluminum vapor to the crystal interface. Once the source is blocked, the growth process effectively stalls, leading to failed runs or inconsistent crystal quality.
Making the Right Choice for Your Goal
When preparing source materials for AlN growth, the inclusion of Copper is a strategic decision based on your specific stability requirements.
- If your primary focus is Process Continuity: Incorporate Copper to prevent surface crust formation and ensure long-duration, uninterrupted growth runs.
- If your primary focus is Rate Control: Utilize the Cu-Al alloy to dilute the vapor concentration, allowing for precise fine-tuning of the evaporation rate.
Ultimately, the Cu-Al alloy transforms a volatile, prone-to-clogging source into a stable, self-cleaning reservoir for consistent crystal production.
Summary Table:
| Feature | Pure Aluminum Source | Cu-Al Alloy Source |
|---|---|---|
| Surface State | Prone to solid AlN shell (passivation) | Self-cleaning; re-dissolves AlN crust |
| Vapor Control | High, erratic evaporation rates | Diluted, stable, and precise concentration |
| Melting Point | Higher | Lowered for better thermal efficiency |
| Process Duration | Frequent interruptions due to blockage | Sustainable for long-duration growth runs |
Maximize Your Crystal Growth Precision with KINTEK
Ensure uninterrupted production and superior material stoichiometry with KINTEK’s advanced laboratory solutions. Backed by expert R&D and manufacturing, KINTEK offers high-performance Muffle, Tube, Rotary, Vacuum, and CVD systems—all fully customizable to meet the rigorous thermal demands of AlN single crystal growth and source material preparation.
Don't let source passivation stall your research. Contact our technical experts today to discover how our high-temperature furnaces can provide the stability and control your laboratory needs.
Visual Guide
References
- Xiaochun Tao, Zhanggui Hu. Growth of Spontaneous Nucleation AlN Crystals by Al-Base Alloy Evaporation in Nitrogen Atmosphere. DOI: 10.3390/cryst14040331
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 1400℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- 1700℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- 1700℃ Controlled Inert Nitrogen Atmosphere Furnace
- 1400℃ Controlled Inert Nitrogen Atmosphere Furnace
- 1200℃ Controlled Inert Nitrogen Atmosphere Furnace
People Also Ask
- How does a laboratory high-temperature tube furnace contribute to the conversion of electrospun fibers? Expert Insights
- What is the function of a furnace in CuAlMn alloy treatment? Achieve Perfect Microstructural Homogenization
- How does a high-temperature tube furnace facilitate sulfur melt-diffusion? Precision Heating for PCFC/S Cathodes
- In what scenarios are laboratory high-temperature tube furnaces or muffle furnaces utilized? Study MgTiO3-CaTiO3 Ceramics
- What is the mechanism of a high-temperature furnace in Bi-2223 sintering? Achieve Precision Phase Transformation

















