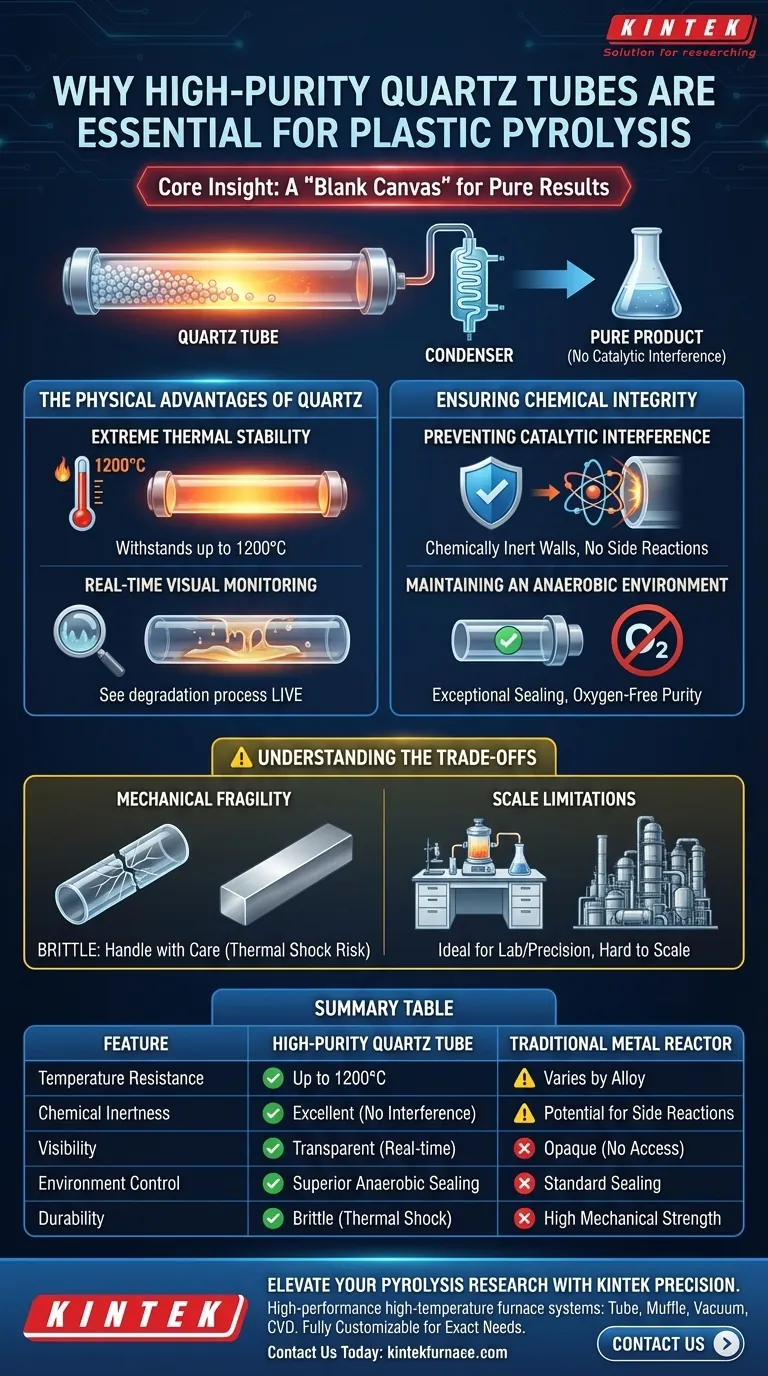
High-purity quartz tubes serve as the definitive reaction chamber for plastic pyrolysis because they provide an uncompromising barrier against chemical contamination while withstanding extreme thermal stress. By resisting temperatures up to 1200°C and offering complete transparency, these tubes allow researchers to conduct high-heat decomposition while visually monitoring the process in real-time.
Core Insight: Successful pyrolysis requires a "blank canvas" environment where the reactor itself does not influence the outcome. High-purity quartz ensures that the end products are derived strictly from the plastic feedstock, free from the catalytic side effects often caused by metal reactors.

The Physical Advantages of Quartz
Extreme Thermal Stability
Plastic pyrolysis involves breaking down polymer chains using intense heat. High-purity quartz is uniquely suited for this, capable of withstanding temperatures up to 1200°C. This thermal headroom ensures the chamber maintains its structural integrity even during aggressive heating cycles.
Real-Time Visual Monitoring
Unlike metal reactors, quartz provides optical transparency. This allows operators to engage in real-time observation of the material changes inside the chamber. Being able to see when physical degradation occurs provides critical data points that temperature sensors alone cannot capture.
Ensuring Chemical Integrity
Preventing Catalytic Interference
One of the most critical challenges in pyrolysis is preventing "side reactions." Metal furnace walls can sometimes act as inadvertent catalysts, altering the chemical pathway of the decomposing plastic. Quartz possesses superior chemical inertness, ensuring that the reactor walls do not react with the feedstock.
Maintaining an Anaerobic Environment
Pyrolysis must occur in the absence of oxygen to prevent combustion. Quartz tubes offer exceptional sealing capabilities. This allows the system to maintain a strict anaerobic environment, which is vital for ensuring the purity and consistency of the resulting fuel or chemical end products.
Understanding the Trade-offs
Mechanical Fragility vs. Chemical Purity
While quartz offers superior chemical resistance compared to metal, it lacks ductility. A quartz reaction chamber is significantly more brittle than steel. It requires careful handling to avoid catastrophic failure from mechanical shock or rapid, uneven cooling (thermal shock).
Scale Limitations
The properties that make quartz ideal for high-precision or laboratory-scale pyrolysis—such as transparency and purity—often become difficult to scale. Constructing massive industrial-scale reactors entirely out of high-purity quartz is generally cost-prohibitive and structurally complex compared to using metal alloys lined with refractory materials.
Making the Right Choice for Your Goal
If you are selecting a reactor material for plastic pyrolysis, consider your specific objectives:
- If your primary focus is fundamental research or chemical analysis: Choose high-purity quartz to eliminate variables and ensure the chemical composition of your output is not skewed by wall effects.
- If your primary focus is process optimization and monitoring: Rely on quartz to visually correlate physical material changes (like melting or charring) with specific temperature thresholds.
High-purity quartz is the superior choice when data integrity and observational access are more critical than industrial durability.
Summary Table:
| Feature | High-Purity Quartz Tube | Traditional Metal Reactor |
|---|---|---|
| Temperature Resistance | Up to 1200°C | Varies by Alloy |
| Chemical Inertness | Excellent (No catalytic interference) | Potential for catalytic side reactions |
| Visibility | Transparent (Real-time monitoring) | Opaque (No visual access) |
| Environment Control | Superior anaerobic sealing | standard sealing |
| Durability | Brittle (High thermal shock risk) | High mechanical strength |
Elevate Your Pyrolysis Research with KINTEK Precision
Don't let reactor contamination compromise your data integrity. KINTEK provides high-performance high-temperature furnace systems—including Tube, Muffle, Vacuum, and CVD systems—specifically designed to meet the rigorous demands of plastic pyrolysis and material science.
Backed by expert R&D and world-class manufacturing, our systems are fully customizable to provide the exact thermal and chemical environment your project requires. Whether you need a transparent quartz interface for real-time monitoring or a robust industrial solution, KINTEK delivers the quality you need to succeed.
Ready to optimize your lab's performance? Contact us today to discuss your unique needs!
Visual Guide
References
- Hitesh Panchal. Fuel Extraction from Plastic Waste. DOI: 10.22214/ijraset.2025.66489
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 1400℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- Laboratory Quartz Tube Furnace RTP Heating Tubular Furnace
- High Pressure Laboratory Vacuum Tube Furnace Quartz Tubular Furnace
- 1700℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- Vertical Laboratory Quartz Tube Furnace Tubular Furnace
People Also Ask
- What is the purpose of bottom-entry argon injection? Enhance Lithium-ion Battery Safety & Purge Efficiency
- How does the choice of ceramic molds affect the results when preparing steel sample ingots? Ensure Maximum Sample Purity
- Why is a high-vacuum furnace required for CP-Ti annealing? Protect Purity and Prevent Embrittlement
- What is the function of injecting water in wood thermal modification? Unlock Superior Stability and Hydrophobicity
- What is the function of vacuum & heating in aluminum degassing? Enhancing Composite Integrity & Density



















