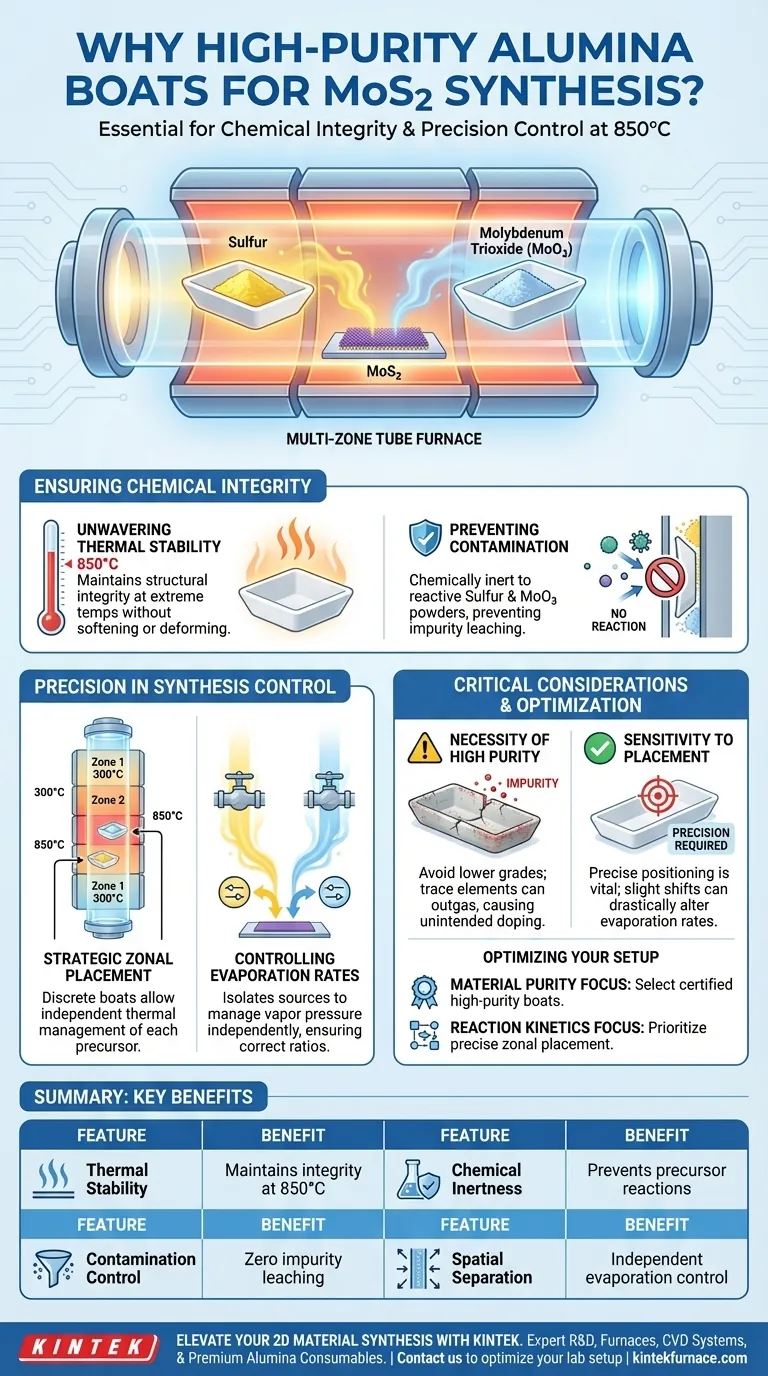
High-purity alumina boats are essential tools in MoS2 synthesis because they provide a chemically inert environment capable of withstanding extreme thermal conditions. Specifically, they facilitate the reaction by holding precursors like sulfur and molybdenum trioxide at operating temperatures of 850°C without introducing contaminants or reacting with the source materials.
The utility of high-purity alumina lies in its dual ability to resist high-temperature chemical corrosion and enable precise control over precursor evaporation rates within a multi-zone tube furnace.

Ensuring Chemical Integrity
Unwavering Thermal Stability
MoS2 synthesis requires rigorous thermal conditions, often reaching operating temperatures of 850°C.
High-purity alumina boats are selected because they maintain their structural integrity at these specific heat levels. They do not soften, deform, or degrade, ensuring the physical containment of the precursors throughout the experiment.
Preventing Precursor Contamination
The most critical function of these boats is their chemical inertness.
At elevated temperatures, reactive powders like Sulfur (S) and Molybdenum Trioxide (MoO3) can easily interact with lesser container materials. High-purity alumina does not react with these powders, preventing impurities from leaching into the synthesis environment and compromising the quality of the MoS2 layers.
Precision in Synthesis Control
Strategic Zonal Placement
The use of discrete boats allows for strategic placement within a tube furnace.
By positioning the alumina boats in specific temperature zones, researchers can manipulate the thermal environment for each precursor independently. This spatial separation is vital for managing the reaction kinetics.
Controlling Evaporation Rates
Successful synthesis depends on managing the vapor pressure of the source materials.
Because the boats isolate the Sulfur and Molybdenum sources, their evaporation rates can be controlled independently. This ensures that the correct ratio of vapors reaches the substrate, promoting the growth of high-quality films.
Critical Considerations and Pitfalls
The Necessity of "High Purity"
It is important not to compromise on the grade of alumina used. Lower-purity boats may contain trace elements that could outgas or react at 850°C, negating the benefits of the container and introducing unintended doping effects.
Sensitivity to Placement
While the boats allow for independent control, this introduces a variable that requires precision. Incorrectly placing a boat by even a small margin within the temperature gradient can drastically alter the evaporation rate, leading to non-stoichiometric growth or failed synthesis.
Optimizing Your Experimental Setup
To ensure successful MoS2 growth, align your container selection with your specific experimental needs:
- If your primary focus is Material Purity: Ensure you select certified high-purity alumina boats to eliminate any risk of reaction with Sulfur or MoO3 at 850°C.
- If your primary focus is Reaction Kinetics: Prioritize the precise placement of these boats within the furnace's temperature zones to independently tune the evaporation rates of your precursors.
By leveraging the thermal stability and inert nature of high-purity alumina, you create a controlled, contaminant-free environment essential for high-quality 2D material synthesis.
Summary Table:
| Feature | Benefit for MoS2 Synthesis |
|---|---|
| Thermal Stability | Maintains structural integrity at 850°C without deformation. |
| Chemical Inertness | Prevents reactions with Sulfur and MoO3 precursors. |
| Contamination Control | Ensures zero leaching of impurities for high-purity 2D layers. |
| Spatial Separation | Allows independent control of evaporation rates in multi-zone furnaces. |
Elevate Your 2D Material Synthesis with KINTEK
Precision in MoS2 synthesis begins with the highest quality laboratory equipment. At KINTEK, we understand that even minor impurities can compromise your research. Backed by expert R&D and manufacturing, we offer a comprehensive range of Muffle, Tube, Rotary, Vacuum, and CVD systems, along with premium high-purity alumina consumables tailored for your unique experimental needs.
Don't let subpar materials derail your results. Partner with KINTEK for customizable, high-temperature lab solutions designed for excellence.
Contact us today to optimize your lab setup
Visual Guide
References
- Feng Liao, Zewen Zuo. Optimizing the Morphology and Optical Properties of MoS2 Using Different Substrate Placement: Numerical Simulation and Experimental Verification. DOI: 10.3390/cryst15010059
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 1400℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- Magnesium Extraction and Purification Condensing Tube Furnace
- Split Chamber CVD Tube Furnace with Vacuum Station CVD Machine
- RF PECVD System Radio Frequency Plasma Enhanced Chemical Vapor Deposition
- Multi Heating Zones CVD Tube Furnace Machine for Chemical Vapor Deposition Equipment
People Also Ask
- How does a laboratory furnace address the strength-ductility trade-off in UFG titanium? Master Thermal Processing.
- Why is high-purity argon protection required during the casting of alloy steel samples? Preserve Sample Integrity
- Why is a high-vacuum furnace required for CP-Ti annealing? Protect Purity and Prevent Embrittlement
- What is the purpose of bottom-entry argon injection? Enhance Lithium-ion Battery Safety & Purge Efficiency
- What is the function of injecting water in wood thermal modification? Unlock Superior Stability and Hydrophobicity


















