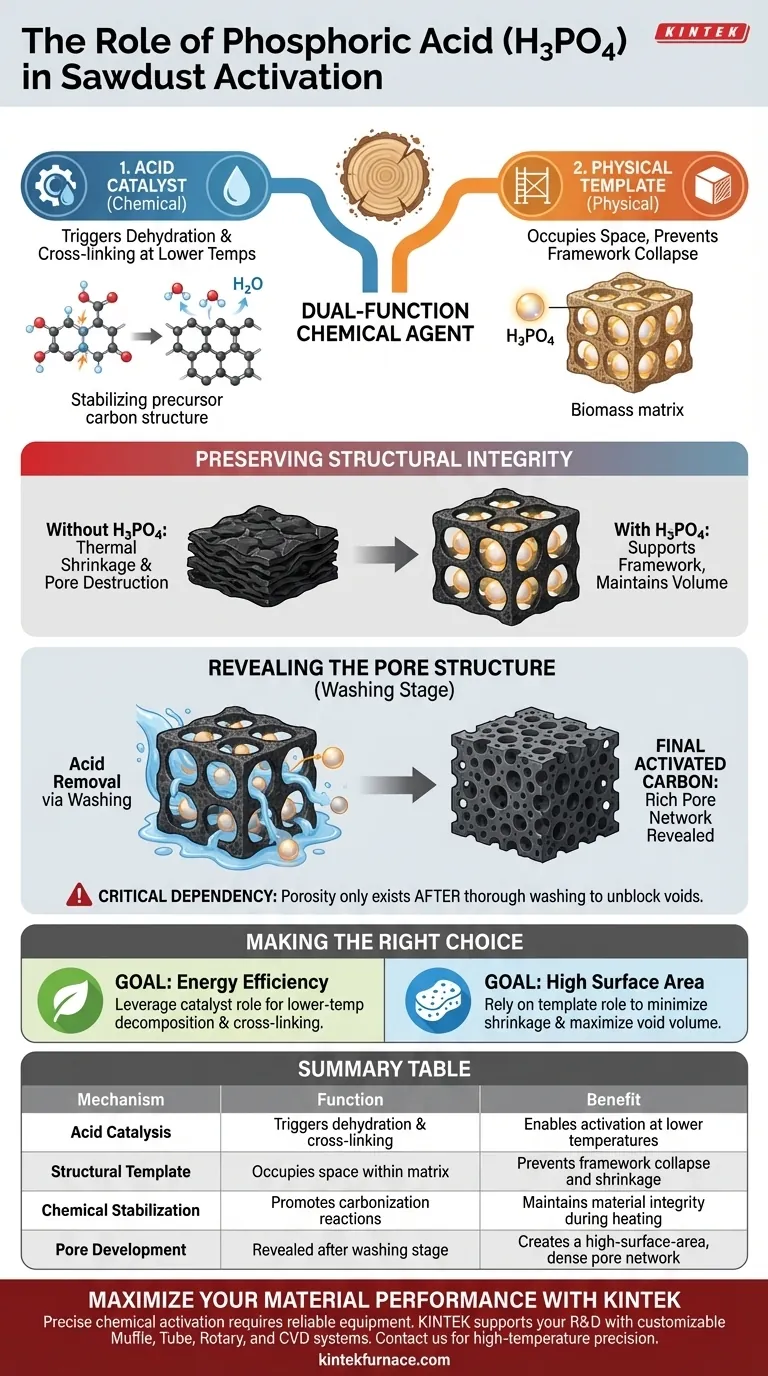
Phosphoric acid serves as a dual-function chemical agent during the activation of sawdust. It acts as an acid catalyst to trigger dehydration and cross-linking in the biomass at lower temperatures, while simultaneously serving as a physical template. This prevents the carbon framework from shrinking or collapsing during heat treatment, ensuring a porous final product.
By occupying space within the biomass during heating and preventing shrinkage, phosphoric acid essentially "props open" the carbon structure. Its removal via washing reveals a highly developed pore network that would otherwise be lost to thermal collapse.

Mechanisms of Action
To understand how phosphoric acid (H3PO4) transforms sawdust into activated carbon, one must look at both its chemical and physical interventions.
Acid Catalysis at Low Temperatures
H3PO4 functions primarily as an acid catalyst. Unlike thermal activation which relies solely on high heat, this chemical agent drives reactions at lower temperatures.
It specifically induces dehydration within the biomass components. Simultaneously, it promotes cross-linking reactions, which help stabilize the organic material as it begins to carbonize.
The Templating Effect
Beyond its chemical reactivity, phosphoric acid acts as a physical template.
It penetrates the sawdust and occupies volume within the material's matrix. During the heat treatment phase, the acid remains embedded in the structure, holding the space that will eventually become pores.
Preserving Structural Integrity
The most critical role of H3PO4 is protecting the carbon framework during the harsh conditions of activation.
Preventing Excessive Shrinkage
As biomass is heated, it naturally tends to shrink and densify, which destroys potential porosity.
Phosphoric acid counteracts this by physically supporting the carbon framework. It restricts the mechanical collapse of the structure, ensuring the material retains its volume.
Revealing the Pore Structure
The activation process concludes with a washing step.
Because the acid acted as a template, washing it away leaves behind empty spaces. These voids constitute the rich pore structure of the final activated carbon.
Critical Process Dependencies
While phosphoric acid is effective, its role creates specific dependencies in the manufacturing workflow.
The Requirement of Post-Activation Washing
The creation of porosity is not complete immediately after heating.
The pore network is only revealed after the washing process. The acid must be thoroughly extracted to unblock the internal voids; otherwise, the "template" remains in place, rendering the surface area inaccessible.
Making the Right Choice for Your Goal
When utilizing phosphoric acid for sawdust activation, your objectives should dictate how you view its role.
- If your primary focus is energy efficiency: Leverage the acid's ability to act as a catalyst to initiate necessary decomposition and cross-linking reactions at lower temperatures.
- If your primary focus is high surface area: Rely on the acid's capacity as a structural template to minimize shrinkage and maximize the void volume available after washing.
Phosphoric acid effectively bridges the gap between raw biomass and high-performance carbon by chemically stabilizing and physically shaping the material.
Summary Table:
| Mechanism | Function | Benefit |
|---|---|---|
| Acid Catalysis | Triggers dehydration & cross-linking | Enables activation at lower temperatures |
| Structural Template | Occupies space within the biomass matrix | Prevents framework collapse and shrinkage |
| Chemical Stabilization | Promotes carbonization reactions | Maintains material integrity during heating |
| Pore Development | Revealed after the washing stage | Creates a high-surface-area, dense pore network |
Maximize Your Material Performance with KINTEK
Precise chemical activation requires reliable, high-performance equipment. At KINTEK, we support your research and production with expert R&D and world-class manufacturing. Whether you are developing activated carbon or advanced composites, our Muffle, Tube, Rotary, Vacuum, and CVD systems are fully customizable to meet your unique temperature and atmospheric requirements.
Ready to elevate your laboratory's efficiency? Contact us today to discuss your specific needs and discover how KINTEK’s high-temperature furnaces can provide the thermal precision your project deserves.
Visual Guide
References
- Xiaoyang Guo, Steven T. Boles. Holistic Processing of Sawdust to Enable Sustainable Hybrid Li-Ion Capacitors. DOI: 10.1007/s11837-024-06542-1
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
People Also Ask
- What is an electric rotary kiln and how does it function? Discover Precision Heating for Sensitive Materials
- What is the significance of the refractory lining in a rotary kiln electric furnace? Unlock Efficiency and Longevity
- What advantages do electric rotary kilns offer over fuel-based kilns? Boost Efficiency and Purity in Your Process
- What are the advantages of electric rotary kilns compared to fuel-fired ones? Boost Precision and Efficiency in Your Process
- What are the main applications of a rotary kiln electric furnace? Achieve Uniform Heat Treatment for Powders
