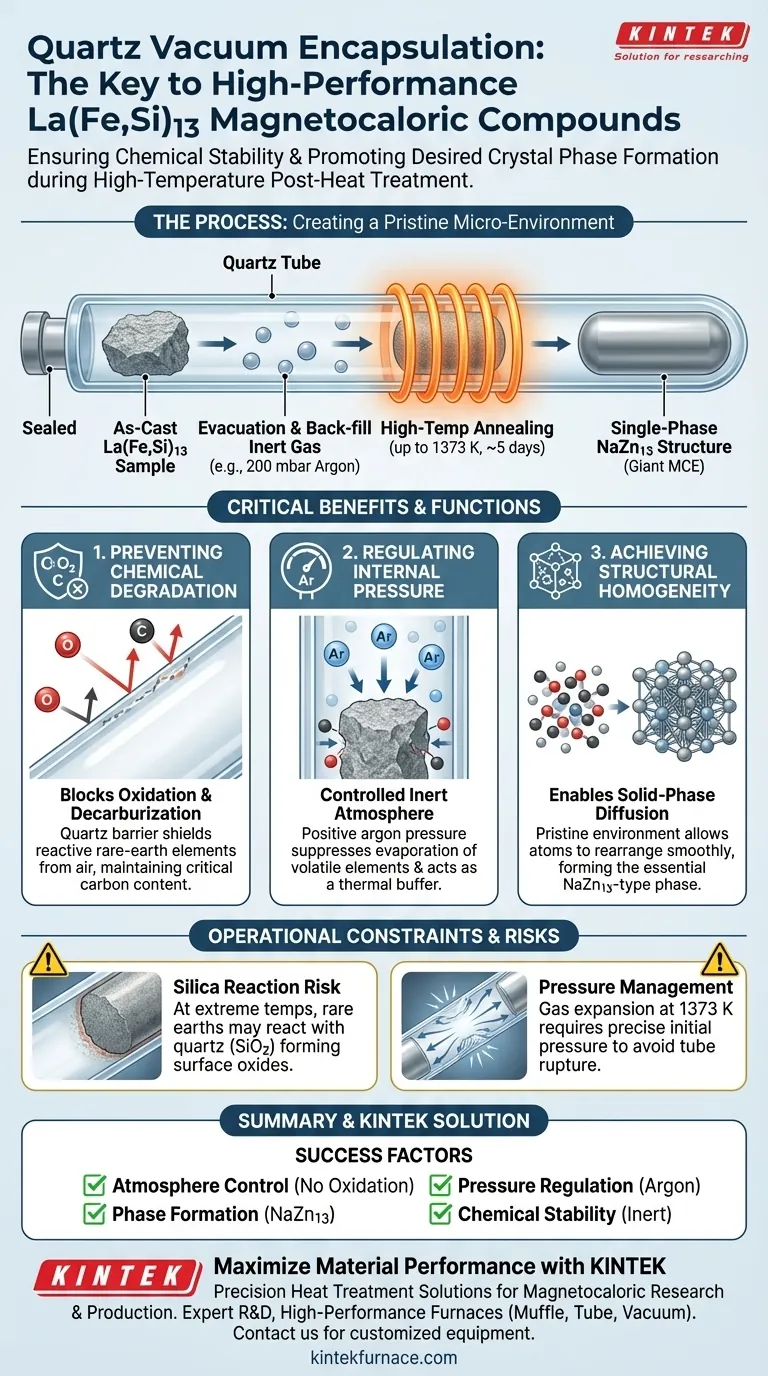
Quartz vacuum encapsulation is the primary method for isolating La(Fe,Si)13-based alloys to ensure chemical stability during high-temperature synthesis. Specifically, sealing as-cast samples under a controlled atmosphere—typically 200 mbar of Argon—shields the material from oxidation and decarburization while it undergoes the extensive annealing required to form the correct crystal phase.
The core function of this technique is to create a pristine, inert environment that facilitates smooth solid-phase diffusion. Without this isolation, the reactive rare-earth elements would degrade before the material could achieve the critical single-phase NaZn13-type structure.

The Critical Role of Environmental Isolation
Preventing Chemical Degradation
La(Fe,Si)13 compounds require aggressive heat treatment, often reaching 1373 K for durations as long as five days.
Exposing rare-earth alloys to open air at these temperatures guarantees rapid oxidation.
Quartz encapsulation prevents this by creating a physical barrier. It also specifically prevents decarburization, ensuring that the carbon content—often critical for tuning magnetic properties—remains stable throughout the process.
Regulating Internal Pressure
The encapsulation is not just a vacuum; it is often back-filled with a specific pressure of inert gas, such as 200 mbar of Argon.
This positive pressure serves a dual purpose: it suppresses the evaporation of volatile elements and provides a thermal buffer.
maintaining a consistent protective pressure is vital for the stability of the sample surface during long-duration heating.
Achieving Structural Homogeneity
Enabling Solid-Phase Diffusion
As-cast alloys are rarely usable in their raw state due to compositional segregation.
To correct this, atoms must physically rearrange themselves within the solid material.
Encapsulation allows this solid-phase diffusion reaction to proceed smoothly over several days without the interference of surface contaminants that could inhibit atomic movement.
Forming the NaZn13-Type Phase
The ultimate goal of this post-heat treatment is to obtain a single-phase NaZn13-type crystal structure.
This specific cubic structure is responsible for the giant magnetocaloric effect (MCE) in these compounds.
If the atmosphere is not strictly controlled via encapsulation, impurity phases form, preventing the stabilization of the NaZn13 structure and severely degrading magnetic performance.
Operational Constraints and Trade-offs
The Risk of Silica Reaction
While quartz is thermally stable, it is not perfectly inert in all conditions.
At extremely high temperatures, there is a risk of the rare-earth elements reacting with the silica ($SiO_2$) in the tube, potentially creating a surface oxide layer.
Pressure Management
Sealing samples under partial argon pressure requires careful calculation.
As the tube heats to 1373 K, the gas expands significantly. If the initial pressure is too high, or the tube volume is too small, the capsule may rupture during the annealing process.
Making the Right Choice for Your Synthesis
To maximize the performance of your magnetocaloric materials, consider your specific processing goals:
- If your primary focus is Phase Purity: Ensure your encapsulation includes a partial pressure of Argon (around 200 mbar) rather than a hard vacuum to suppress volatility while preventing oxidation.
- If your primary focus is Structural Integrity: Carefully clean the as-cast samples before encapsulation to ensure no surface contaminants interfere with the solid-state diffusion required for the NaZn13 phase.
Success in synthesizing La(Fe,Si)13 compounds relies not just on the temperature of the furnace, but on the integrity of the micro-environment created inside the quartz tube.
Summary Table:
| Feature | Quartz Vacuum Encapsulation Benefit |
|---|---|
| Atmosphere Control | Prevents oxidation and decarburization of rare-earth elements. |
| Phase Formation | Facilitates solid-phase diffusion to achieve NaZn13-type structure. |
| Pressure Regulation | Argon back-filling (e.g., 200 mbar) suppresses volatile element evaporation. |
| Chemical Stability | Creates an inert environment for high-temperature annealing (up to 1373 K). |
| Performance Impact | Ensures maximum giant magnetocaloric effect (MCE) by eliminating impurities. |
Maximize Your Material Performance with KINTEK
Precision heat treatment is the cornerstone of advanced material synthesis. Backed by expert R&D and manufacturing, KINTEK offers high-performance Muffle, Tube, Rotary, Vacuum, and CVD systems designed to meet the rigorous demands of magnetocaloric research and industrial production.
Whether you require precise atmosphere control for quartz encapsulation or customizable high-temperature furnaces for solid-phase diffusion, our solutions are tailored to your unique needs.
Ready to elevate your lab's efficiency and achieve superior phase purity?
Contact KINTEK today for expert guidance and customized equipment solutions.
Visual Guide
References
- Fengqi Zhang, Yang Ren. Engineering Light‐Element Modified LaFe <sub>11.6</sub> Si <sub>1.4</sub> Compounds Enables Tunable Giant Magnetocaloric Effect. DOI: 10.1002/advs.202416288
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- High Performance Vacuum Bellows for Efficient Connection and Stable Vacuum in Systems
- CF KF Flange Vacuum Electrode Feedthrough Lead Sealing Assembly for Vacuum Systems
- Ultra High Vacuum Observation Window Stainless Steel Flange Sapphire Glass Sight Glass for KF
People Also Ask
- Why are micrometer-scale micro-fiber filter candles used to treat condensed flue gases in pyrolysis systems?
- What role does a closed pressure vessel play during the carbonation of gamma-C2S? Unlock Rapid Mineralization
- Why is an Ultra-High Vacuum (UHV) System Required for In2Se3? Achieving Atomic-Level Ferroelectric Clarity
- How does the pressure environment affect the metallic thermal reduction for titanium? Master Precision Control
- How does a Mass Flow Controller influence 2D superlattices? Precision CVD Control for Sub-10nm Patterning


