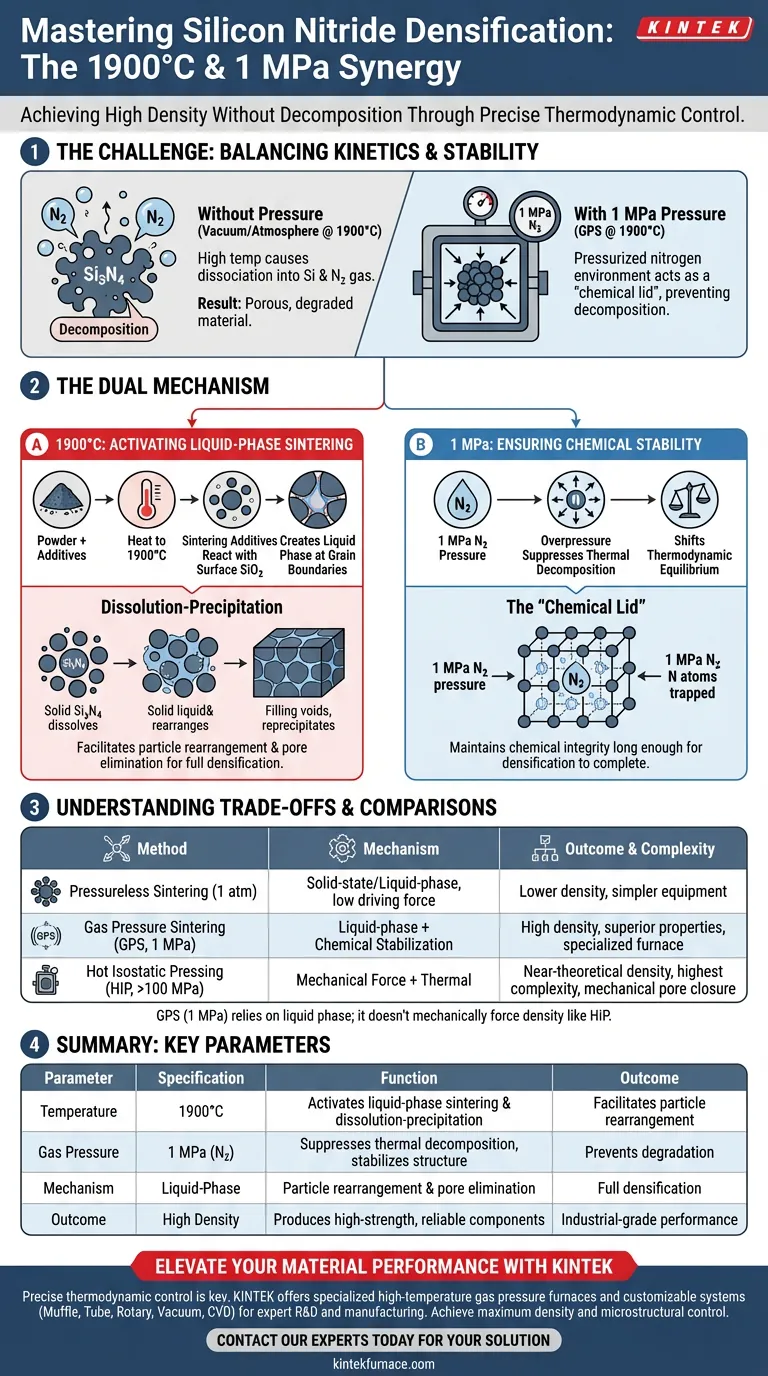
The application of 1900°C heat combined with 1 MPa gas pressure creates the specific thermodynamic conditions required to densify Silicon Nitride without chemically degrading it. While the extreme temperature drives the liquid-phase mechanism needed to eliminate porosity, the pressurized nitrogen environment is the critical stabilizer that prevents the material from decomposing during the process.
Achieving high density in Silicon Nitride requires balancing kinetic energy with chemical stability. The 1900°C temperature facilitates the necessary particle rearrangement, but this is only viable because the 1 MPa nitrogen pressure suppresses thermal decomposition, allowing the material to survive the sintering cycle.
The Mechanism of Densification at 1900°C
To understand why this specific temperature is non-negotiable, you must look at how Silicon Nitride particles fuse. Unlike metals that might sinter via solid-state diffusion, Silicon Nitride relies on a liquid phase.
Activating Sintering Additives
Pure Silicon Nitride is notoriously difficult to densify because of its strong covalent bonds and low self-diffusion rates.
At 1900°C, sintering additives (such as oxides mixed into the powder) react with the silicon dioxide naturally present on the surface of the Silicon Nitride particles.
The Dissolution-Precipitation Process
This reaction creates a liquid phase at the grain boundaries.
Through a process called dissolution-precipitation, the solid Silicon Nitride dissolves into this liquid, rearranges, and reprecipitates. This fills the voids between particles, resulting in full densification.
The Critical Role of 1 MPa Pressure
If you were to heat Silicon Nitride to 1900°C in a standard vacuum or at atmospheric pressure, the material would fail to densify properly because it would begin to dissociate.
Suppressing Thermal Decomposition
At temperatures approaching 1900°C, Silicon Nitride becomes thermodynamically unstable. It tends to decompose into silicon liquid and nitrogen gas.
By introducing 1 MPa of nitrogen gas pressure, you effectively shift the thermodynamic equilibrium.
Maintaining Chemical Stability
This overpressure acts as a "chemical lid," countering the internal vapor pressure of the nitrogen trying to escape the lattice.
This ensures the material remains chemically stable long enough for the densification process to complete, resulting in a finished component with high integrity rather than a porous or degraded one.
Understanding the Trade-offs
While Gas Pressure Sintering (GPS) at these parameters offers superior material properties, it is important to recognize the operational complexities involved.
Equipment Complexity vs. Material Quality
Achieving 1 MPa is significantly more complex than standard pressureless sintering (1 atm). It requires specialized furnace chambers capable of handling both high thermal loads and pressurized gases safely.
However, this added complexity is the "cost of entry" for achieving densities that pressureless sintering cannot match.
Limitation of Low Pressure
While 1 MPa is sufficient to stop decomposition at 1900°C, it is primarily a chemical stabilizer.
Unlike Hot Isostatic Pressing (HIP) which uses much higher pressures (often >100 MPa) to mechanically force pore closure, the 1 MPa used here relies entirely on the liquid phase for densification. If the powder chemistry is incorrect, 1 MPa will not mechanically force the material to full density.
Making the Right Choice for Your Goal
The decision to utilize a gas pressure sintering furnace involves balancing your need for material performance against processing costs.
- If your primary focus is Maximum Density: Ensure your additives are optimized for 1900°C, as the pressure alone cannot densify the material without the liquid phase.
- If your primary focus is Microstructural Control: Monitor the 1 MPa pressure carefully, as drops in pressure at peak temperature will lead to immediate surface degradation.
Ultimately, using 1 MPa at 1900°C is the industry-standard method for producing high-performance Silicon Nitride components that require both high strength and reliability.
Summary Table:
| Parameter | Specification | Function in Silicon Nitride Sintering |
|---|---|---|
| Temperature | 1900°C | Activates liquid-phase sintering and dissolution-precipitation. |
| Gas Pressure | 1 MPa (N2) | Suppresses thermal decomposition and stabilizes chemical structure. |
| Mechanism | Liquid-Phase | Facilitates particle rearrangement and pore elimination. |
| Outcome | High Density | Produces high-strength, reliable components for industrial use. |
Elevate Your Material Performance with KINTEK
Precise control over thermodynamics is the key to mastering Silicon Nitride densification. At KINTEK, we provide the specialized equipment necessary to achieve these rigorous standards.
Backed by expert R&D and world-class manufacturing, KINTEK offers Muffle, Tube, Rotary, Vacuum, and CVD systems, alongside our advanced high-temperature gas pressure furnaces. All our systems are fully customizable to meet your unique laboratory or industrial needs, ensuring you achieve maximum density and superior microstructural control every time.
Ready to optimize your sintering process? Contact our technical experts today to find the perfect high-temperature solution for your application.
Visual Guide
References
- You Zhou, Manabu Fukushima. Effects of rare‐earth oxides on microstructure, thermal conductivity, and mechanical properties of silicon nitride. DOI: 10.1111/jace.70028
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 9MPa Air Pressure Vacuum Heat Treat and Sintering Furnace
- Vacuum Heat Treat Sintering Furnace with Pressure for Vacuum Sintering
- 600T Vacuum Induction Hot Press Vacuum Heat Treat and Sintering Furnace
- Vacuum Heat Treat Sintering and Brazing Furnace
- 1700℃ High Temperature Laboratory Tube Furnace with Alumina Tube
People Also Ask
- How does a vacuum system integrated with mechanical and diffusion pumps enhance lead carbonate decomposition? Guide
- What is the function of vacuum & heating in aluminum degassing? Enhancing Composite Integrity & Density
- Why is a high-vacuum furnace required for CP-Ti annealing? Protect Purity and Prevent Embrittlement
- Why control nitrogen partial pressure in martensitic steel treatment? Prevent Denitrogenation & Protect Hardness
- How does temperature control at 950°C affect SiC/Cu-Al2O3 composites? Optimize Sintering for High Strength



















