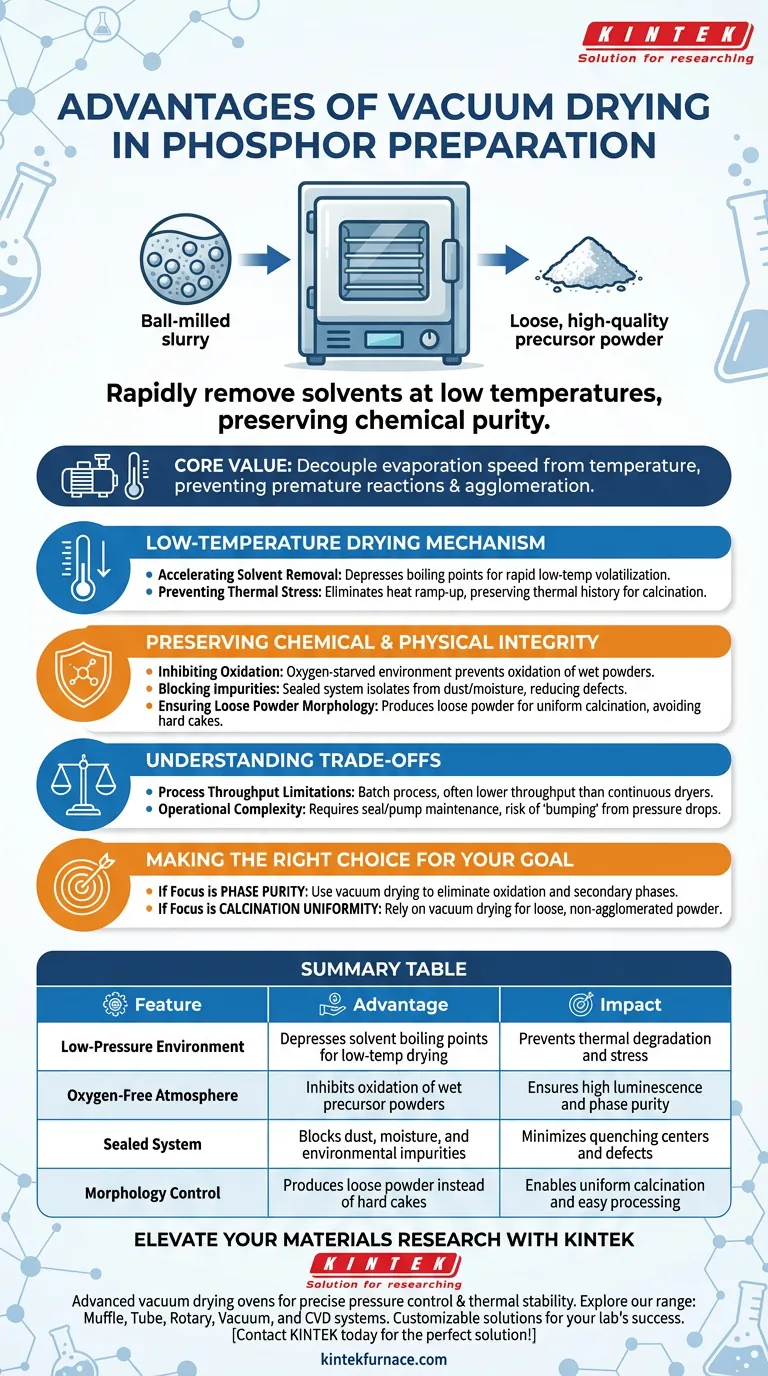
The primary advantage of using a vacuum drying oven in phosphor preparation is the ability to rapidly remove organic solvents from ball-milled slurries at low temperatures while preserving chemical purity. By operating under negative pressure, the oven accelerates evaporation without subjecting the material to the thermal stress that typically causes oxidation or environmental contamination. This process yields a loose, high-quality precursor powder that is chemically stable and physically optimized for the subsequent calcination phase.
The core value of vacuum drying lies in decoupling evaporation speed from temperature. It allows you to dry phosphor precursors efficiently without triggering the premature chemical reactions or agglomeration that occur in standard thermal ovens.
The Mechanism of Low-Temperature Drying
Accelerating Solvent Removal
In phosphor preparation, precursors are often processed as ball-milled slurries containing organic solvents like ethanol. A vacuum drying oven lowers the internal pressure, which significantly depresses the boiling point of these solvents.
This allows the liquid to volatilize rapidly at relatively low temperatures. You achieve high drying efficiency without needing to ramp up the heat, which is often detrimental to sensitive precursor materials.
Preventing Thermal Stress
Standard drying methods rely on heat to drive off moisture, which can inadvertently activate the material or degrade its structure. By utilizing a vacuum, you remove the thermal variable from the drying equation.
This ensures that the energy input is used strictly for phase change (evaporation) rather than heating the powder mass, preserving the thermal history of the sample for the actual calcination step.
Preserving Chemical and Physical Integrity
Inhibiting Oxidation
One of the most critical risks during the drying of wet powders is oxidation. The primary reference highlights that the vacuum environment effectively starves the process of oxygen.
This prevents the wet powder from oxidizing during the transition from slurry to solid. Maintaining the correct oxidation state of the precursor is essential for ensuring the final phosphor achieves the desired luminescence properties.
blocking Environmental Impurities
Drying in an open or standard convection oven exposes the sample to circulating air, which may carry dust or moisture. A vacuum oven operates as a sealed system.
This isolation prevents the introduction of environmental impurities that could act as quenching centers or defects in the final phosphor crystal lattice.
Ensuring Loose Powder Morphology
Perhaps the most practical advantage for processing is the physical state of the dried product. Vacuum drying results in a loose precursor powder rather than a hard, caked mass.
This "loose" morphology is ideal for calcination. It ensures uniform heat distribution during the firing process and reduces the need for aggressive post-drying grinding, which could introduce contamination.
Understanding the Trade-offs
Process Throughput Limitations
While vacuum drying offers superior quality, it is generally a batch process. It is often less continuous and has a lower throughput compared to conveyor belt dryers or spray dryers used in industrial mass production.
Operational Complexity
Vacuum systems require maintenance of seals and pumps to ensure consistent pressure. If the pressure drops too suddenly, there is a risk of "bumping" or splattering, where the solvent boils violently, potentially displacing the powder or coating the oven interior.
Making the Right Choice for Your Goal
To maximize the quality of your phosphor synthesis, consider how these variables align with your specific objectives.
- If your primary focus is Phase Purity: Use vacuum drying to eliminate oxidation risks and prevent the formation of secondary phases caused by environmental impurities.
- If your primary focus is Calcination Uniformity: Rely on vacuum drying to produce a loose, non-agglomerated precursor powder that reacts evenly during high-temperature firing.
Ultimately, vacuum drying is the superior choice when the chemical fidelity and physical morphology of the precursor are prioritized over raw processing speed.
Summary Table:
| Feature | Advantage in Phosphor Preparation | Impact on Final Product |
|---|---|---|
| Low-Pressure Environment | Depresses solvent boiling points for low-temp drying | Prevents thermal degradation and stress |
| Oxygen-Free Atmosphere | Inhibits oxidation of wet precursor powders | Ensures high luminescence and phase purity |
| Sealed System | Blocks dust, moisture, and environmental impurities | Minimizes quenching centers and defects |
| Morphology Control | Produces loose powder instead of hard cakes | Enables uniform calcination and easy processing |
Elevate Your Materials Research with KINTEK
Don't let oxidation or contamination compromise your phosphor synthesis. KINTEK’s advanced vacuum drying ovens provide the precise pressure control and thermal stability required to produce high-purity, loose precursor powders.
Backed by expert R&D and world-class manufacturing, we offer a comprehensive range of lab equipment including Muffle, Tube, Rotary, Vacuum, and CVD systems. Whether you need a standard setup or a fully customizable high-temperature furnace for unique research needs, our team is ready to support your laboratory's success.
Contact KINTEK today to find the perfect solution for your lab!
Visual Guide
References
- YU Xin-hong, Wei Feng. Anti-thermal-quenching and colour-tuneable Tb3+/Ce3+-doped phosphor from natural wollastonite. DOI: 10.2298/pac2404395y
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Vacuum Heat Treat Sintering and Brazing Furnace
- Vacuum Heat Treat Sintering Furnace Molybdenum Wire Vacuum Sintering Furnace
- Dental Porcelain Zirconia Sintering Ceramic Vacuum Press Furnace
- Molybdenum Vacuum Heat Treat Furnace
- Vacuum Hot Press Furnace Machine Heated Vacuum Press
People Also Ask
- What is one of the most important uses of vacuum heat treating furnaces in aerospace? Achieve Superior Strength in Aircraft Alloys
- What are the steps involved in a typical vacuum brazing treatment? Master the Process for Strong, Clean Joints
- How does vacuum heat treating affect the grain structure of metal alloys? Achieve Precise Microstructure Control
- What is the role of high-precision furnaces in Inconel 718 heat treatment? Master Microstructural Engineering
- What are the advantages of using a vacuum heat treatment furnace? Achieve Superior Material Quality and Control












