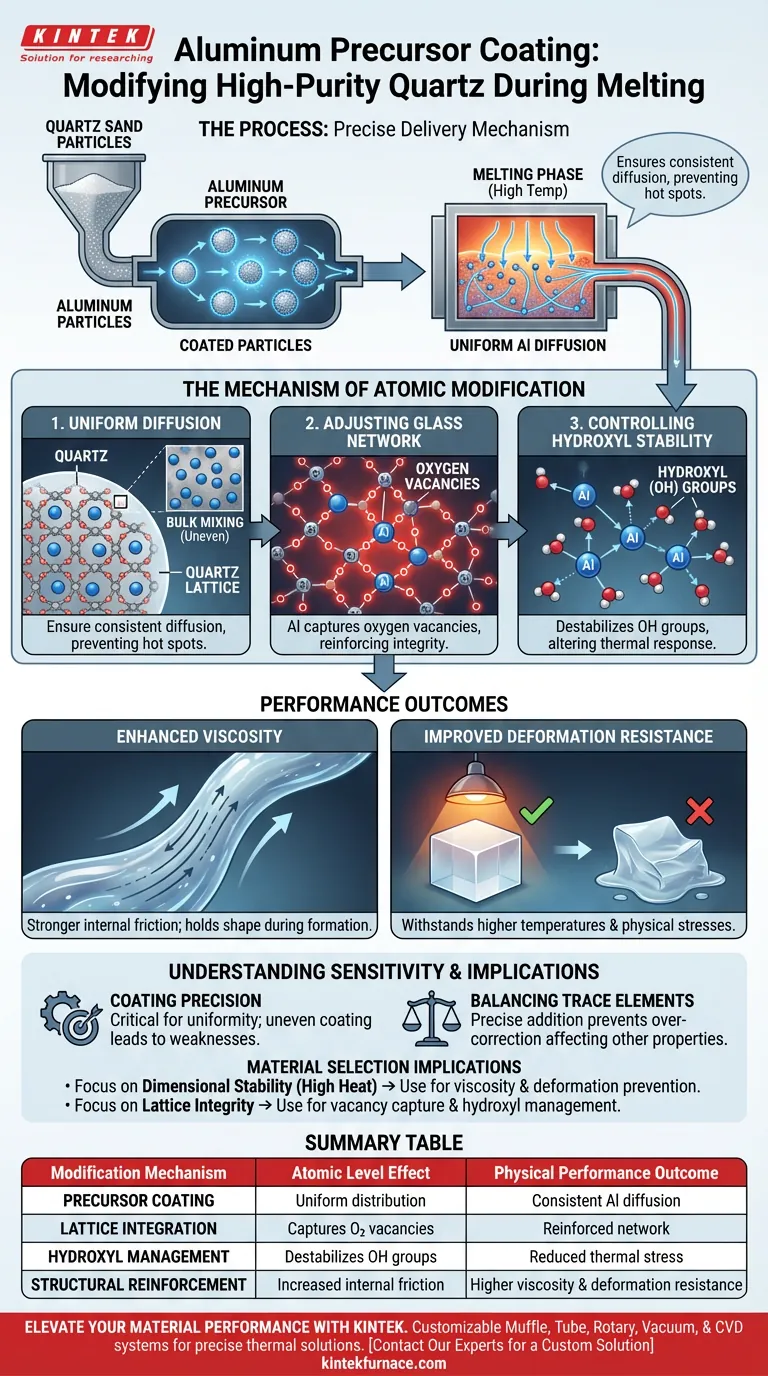
The aluminum precursor coating process acts as a precise delivery mechanism for modifying high-purity quartz. Instead of traditional bulk mixing, this method applies a uniform layer of an aluminum source directly onto individual quartz sand particles. This ensures that when the material enters the melting phase, aluminum diffuses evenly throughout the quartz glass lattice, fundamentally altering its internal structure to improve performance.
By integrating aluminum at the particle level, this process modifies the glass network to capture oxygen vacancies and reduce hydroxyl stability. The critical outcome is a quartz product with significantly enhanced viscosity and resistance to high-temperature deformation.

The Mechanism of Atomic Modification
The effectiveness of this process lies in how it changes the quartz at the microscopic and atomic levels.
Achieving Uniform Diffusion
The primary challenge in modifying quartz is ensuring additives are spread evenly. By coating the precursor on the sand particles prior to melting, the process guarantees a uniform distribution of aluminum.
As the high-temperature melting occurs, this uniform spacing allows the aluminum to diffuse consistent into the quartz glass lattice. This prevents "hot spots" or areas of low concentration that can occur with less precise doping methods.
Adjusting the Glass Network
Once integrated into the lattice, the aluminum atoms actively restructure the glass network.
Specifically, the aluminum acts to capture oxygen vacancies. These vacancies are essentially defects in the atomic structure; by filling or managing them, the aluminum reinforces the integrity of the glass network.
Controlling Hydroxyl Stability
The process also targets the chemical behavior of the glass. It works to reduce the stability of hydroxyl (OH) groups within the structure.
Hydroxyl content is a key variable in quartz performance. By destabilizing these groups, the aluminum doping alters how the glass reacts to thermal stress and dehydration.
Performance Outcomes
The structural changes detailed above translate directly into measurable physical properties in the final product.
Enhanced Viscosity
The tightening of the glass network results in a measurable increase in the material's viscosity.
Higher viscosity indicates a stronger internal friction within the molten or semi-molten glass. This is a desirable trait for manufacturing processes that require the material to hold its shape during formation.
Improved Deformation Resistance
Consequently, the modified quartz exhibits superior resistance to deformation.
Because the lattice defects (oxygen vacancies) are managed and the network is reinforced, the final glass product can withstand higher temperatures and physical stresses without warping or losing its dimensional accuracy.
Understanding Process Sensitivity
While this coating process offers significant advantages, it introduces specific variables that must be managed to ensure quality.
The Necessity of Coating Precision
The success of this modification relies entirely on the uniformity of the initial coating. If the aluminum precursor layer is uneven, the subsequent diffusion during melting will be inconsistent, potentially leading to localized structural weaknesses.
Balancing Trace Elements
This process allows for the "precise addition" of trace elements, but this precision cuts both ways. Deviations in the amount of aluminum applied can over-correct the lattice structure, potentially affecting other material properties beyond viscosity.
Implications for Material Selection
To determine if this modification process aligns with your project requirements, consider the following performance goals:
- If your primary focus is dimensional stability at high heat: Rely on aluminum precursor coating to increase viscosity and prevent deformation under thermal load.
- If your primary focus is lattice integrity: Use this method to actively capture oxygen vacancies and manage the stability of hydroxyl groups within the glass structure.
Ultimately, this coating technique transforms standard quartz sand into a robust, engineered material capable of meeting rigorous thermal and structural standards.
Summary Table:
| Modification Mechanism | Atomic Level Effect | Physical Performance Outcome |
|---|---|---|
| Precursor Coating | Uniform distribution on sand particles | Consistent Al diffusion; no structural hot spots |
| Lattice Integration | Captures oxygen vacancies | Reinforced glass network integrity |
| Hydroxyl Management | Destabilizes OH groups | Reduced thermal stress and dehydration issues |
| Structural Reinforcement | Increased internal friction | Higher viscosity and superior deformation resistance |
Elevate Your Material Performance with KINTEK
Are you looking to optimize your high-purity quartz processing or high-temperature material modification? Backed by expert R&D and manufacturing, KINTEK offers specialized Muffle, Tube, Rotary, Vacuum, and CVD systems, along with other lab high-temp furnaces—all fully customizable to meet your unique research and production needs.
Our advanced thermal solutions provide the precise temperature control and uniform heating environments required for sensitive coating and melting processes. Experience the KINTEK advantage in material science today.
Contact Our Experts for a Custom Solution
Visual Guide
References
- Bartłomiej Adam Gaweł, Marisa Di Sabatino. Influence of aluminium doping on high purity quartz glass properties. DOI: 10.1039/d4ra01716a
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Vacuum Induction Melting Furnace
- High Pressure Laboratory Vacuum Tube Furnace Quartz Tubular Furnace
- Vertical Laboratory Quartz Tube Furnace Tubular Furnace
- 1200℃ Split Tube Furnace Laboratory Quartz Tube Furnace with Quartz Tube
- Magnesium Extraction and Purification Condensing Tube Furnace
People Also Ask
- What role does a Vacuum Induction Melting furnace play in the production of high-aluminum nickel-based superalloys?
- What are the technical advantages of using a Vacuum Induction Melting furnace in the development of advanced packaging steel?
- What is the role of a Vacuum Induction Melting furnace in Fe3Al/Cr3C2 preparation? Purity & Precision for Cladding
- What is the critical role of a Vacuum Induction Melting furnace in FeAl alloy prep? Achieve Ultra-High Purity Alloys
- What is the role of VIM and directional solidification in aero-engine blade substrates? Engineering Extreme Durability



















