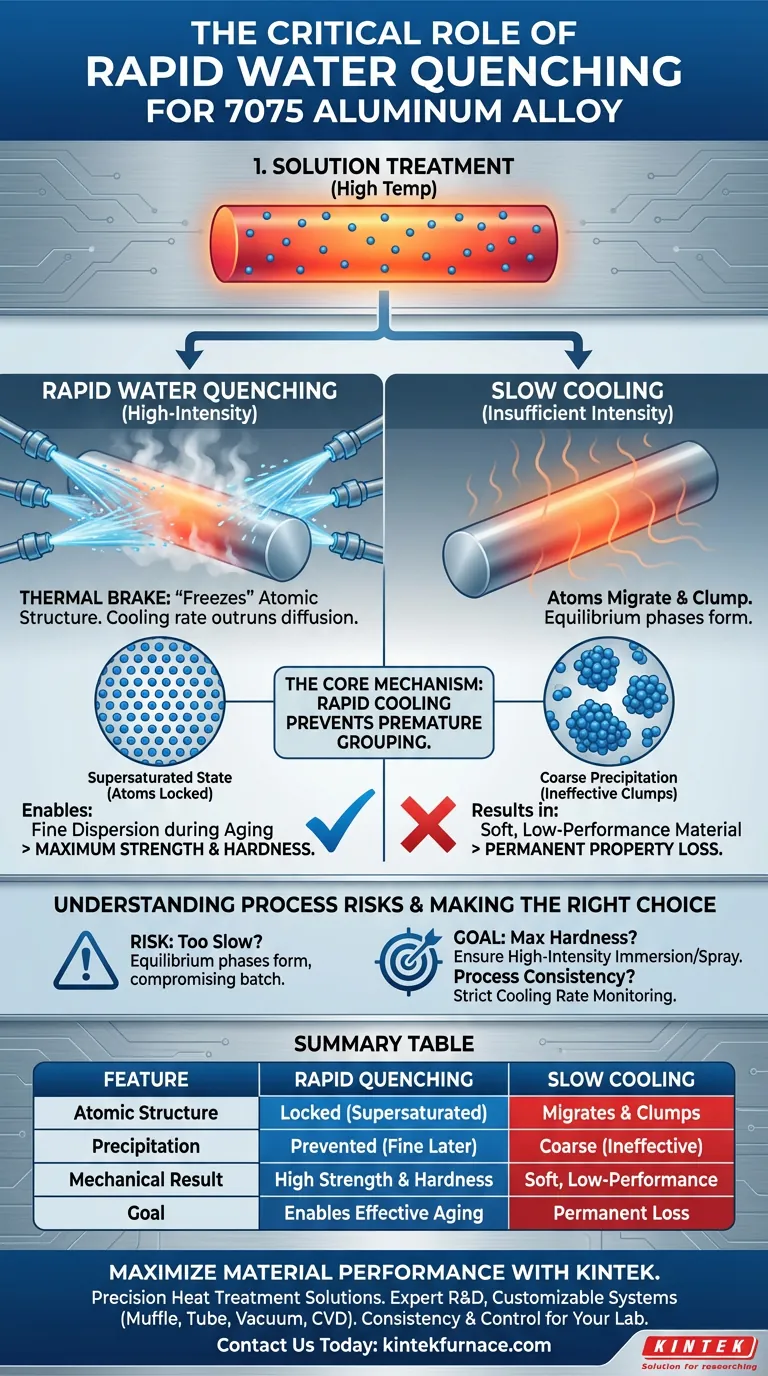
Rapid water quenching is the fundamental prerequisite for achieving high strength in 7075 aluminum. By subjecting the heated billet to high-intensity spraying or immersion, the system forces the material to cool instantaneously from its solution treatment temperature to room temperature. This extreme cooling rate is required to lock solute atoms in place, preventing them from grouping together prematurely.
The Core Mechanism The quench acts as a thermal brake that "freezes" the atomic structure created at high temperatures. Without this rapid cooling, the strengthening elements would precipitate out as coarse particles during the cool-down, rendering the material incapable of achieving its maximum hardness during the final aging process.
The Physical Necessity of Rapid Cooling
Locking the Atomic Structure
At high solution temperatures, the alloying elements (solutes) are dissolved uniformly within the aluminum matrix.
The primary function of the water quench is to preserve this high-temperature arrangement.
By dropping the temperature instantly, the system denies these atoms the time and thermal energy required to move or separate.
Preventing Coarse Precipitation
If 7075 aluminum is allowed to cool slowly, the dissolved elements will naturally migrate out of the solution.
This results in "coarse precipitation," where the strengthening phases form large, ineffective clumps.
Once these coarse phases form during cooling, they cannot contribute to the material's strength, leading to a softer, lower-performance end product.
Enabling the Hardening Process
Creating a Supersaturated State
The quench does not directly harden the metal; rather, it creates a chemically unstable "supersaturated" condition.
This state is the mandatory starting point for effective heat treatment.
It ensures that the alloying elements remain trapped in the solution, waiting to be activated.
Facilitating Fine Dispersion
The ultimate goal of 7075 heat treatment is to produce fine, dispersed strengthening phases.
Because the quench successfully prevented coarse clumping, subsequent natural or artificial aging processes can control the precipitation precisely.
This controlled release leads to the fine microstructural dispersion necessary to achieve the alloy's specified hardness.
Understanding the Process Risks
The Consequence of Insufficient Intensity
The primary reference highlights that the quenching system must use high-intensity spraying or immersion.
A lack of intensity results in a cooling rate that is too slow to "outrun" the natural diffusion of atoms.
If the system fails to achieve this critical speed, equilibrium phases will form during the drop to room temperature, permanently compromising the mechanical properties of the batch.
Making the Right Choice for Your Goal
To maximize the performance of 7075 aluminum, the quenching phase must be viewed as the critical bridge between heating and aging.
- If your primary focus is Maximum Hardness: Ensure your system provides high-intensity immersion or spray to completely suppress coarse precipitation.
- If your primary focus is Process Consistency: Monitor the cooling rate strictly to guarantee the atomic structure is frozen uniformly from the solution temperature.
The entire potential of the alloy relies on the speed of the quench to enable effective aging.
Summary Table:
| Feature | Rapid Quenching | Slow Cooling |
|---|---|---|
| Atomic Structure | Locked in supersaturated state | Elements migrate and clump |
| Precipitation | Prevented (Fine dispersion later) | Coarse, ineffective particles form |
| Mechanical Result | High strength and hardness | Soft, low-performance material |
| Goal | Enables effective aging | Permanent loss of properties |
Maximize Your Material Performance with KINTEK
Precision in heat treatment is the difference between high-performance alloys and material failure. At KINTEK, we understand the critical nature of cooling rates and thermal stability.
Backed by expert R&D and manufacturing, KINTEK offers Muffle, Tube, Rotary, Vacuum, and CVD systems, along with specialized lab high-temp furnaces—all fully customizable to meet your unique metallurgical requirements. Whether you are treating 7075 aluminum or developing advanced ceramics, our equipment ensures the consistency and control your lab demands.
Ready to elevate your thermal processing? Contact our expert team today to find the perfect customizable solution for your research and production needs.
Visual Guide
References
- Hao Wu, Richard Turner. Microstructure Characterisation and Modelling of Pre-Forging Solution Treatment of 7075 Aluminium Alloy Using Novel Heating Methods. DOI: 10.3390/jmmp9010002
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Custom Made Versatile CVD Tube Furnace Chemical Vapor Deposition CVD Equipment Machine
- High Performance Vacuum Bellows for Efficient Connection and Stable Vacuum in Systems
- HFCVD Machine System Equipment for Drawing Die Nano Diamond Coating
- Multi Heating Zones CVD Tube Furnace Machine for Chemical Vapor Deposition Equipment
- Laboratory Quartz Tube Furnace RTP Heating Tubular Furnace
People Also Ask
- What future trends are expected in the development of CVD tube furnaces? Discover Smarter, More Versatile Systems
- What are the key features of CVD tube furnaces for 2D material processing? Unlock Precision Synthesis for Superior Materials
- What is the working principle of a CVD tube furnace? Achieve Precise Thin Film Deposition for Your Lab
- Which industries and research fields benefit from CVD tube furnace sintering systems for 2D materials? Unlock Next-Gen Tech Innovations
- Why are CVD tube furnace sintering systems indispensable for 2D material research and production? Unlock Atomic-Scale Precision








