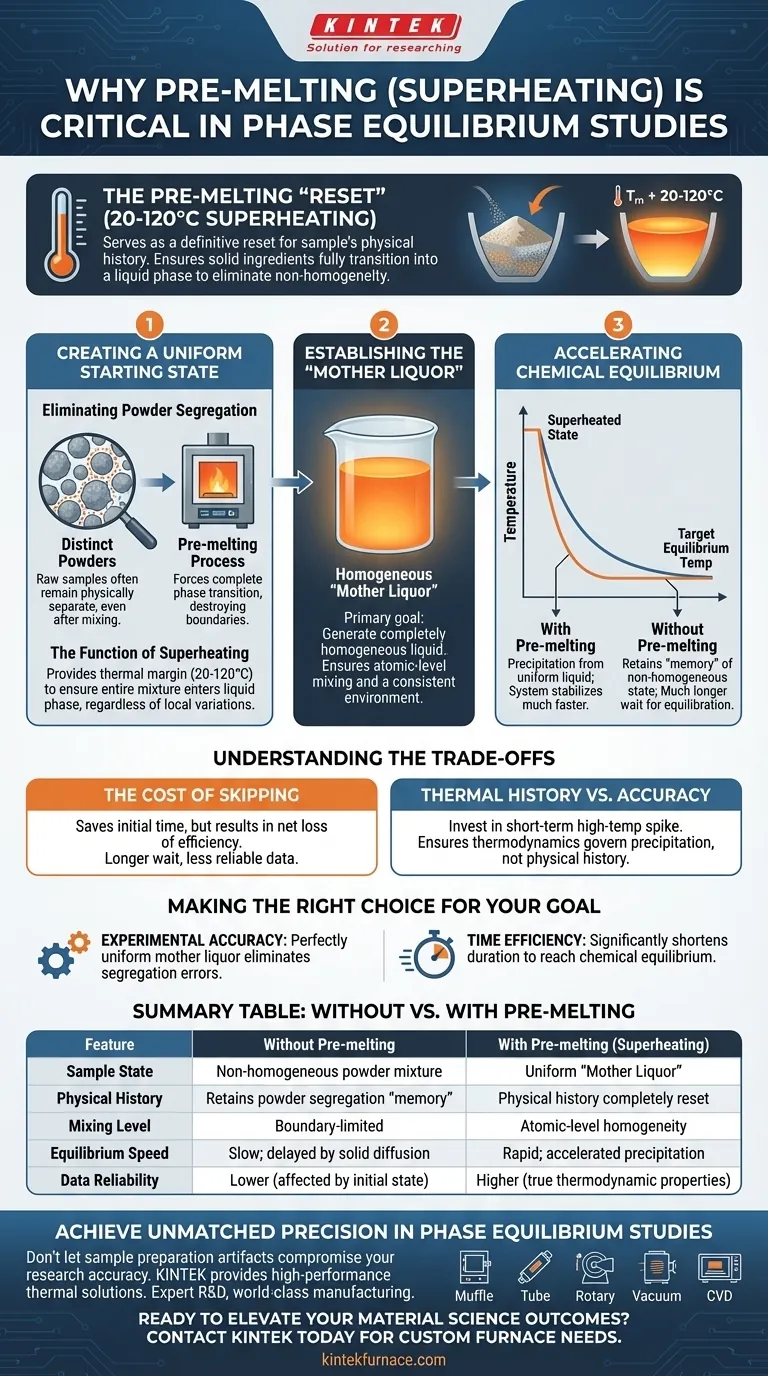
The pre-melting process serves as a definitive "reset" for your sample's physical history. By subjecting the mixture to temperatures 20 to 120 degrees Celsius above the melting point, you ensure the solid ingredients fully transition into a liquid phase. This is necessary to thoroughly mix the components and eliminate the non-homogeneity inherent in the original individual powders.
Superheating creates a uniform "mother liquor," which is essential for accurate phase precipitation. This process removes inconsistencies in the starting material, thereby accelerating the time required to reach true chemical equilibrium at the target temperature.

Creating a Uniform Starting State
Eliminating Powder Segregation
Raw samples often begin as mixtures of distinct powders. Even with mechanical mixing, these solids remain physically separate particles with boundaries.
Directly heating these powders to the target equilibrium temperature often preserves this segregation. The pre-melting process destroys these boundaries by forcing a complete phase transition.
The Function of Superheating
Heating the sample 20 to 120 degrees Celsius beyond the melting point provides a critical thermal margin.
This "superheating" ensures that the entire mixture enters the liquid phase, regardless of local variations in composition or melting points. It guarantees that no residual solids remain to skew the results.
The Impact on Equilibrium Kinetics
Establishing the "Mother Liquor"
The primary goal of this high-temperature step is to generate a completely homogeneous liquid, referred to as the mother liquor.
This liquid serves as the uniform baseline for the experiment. By ensuring the elements are mixed at an atomic level, you create a consistent environment for the next phase of the study.
Accelerating Chemical Equilibrium
Once the homogeneous mother liquor is established, the temperature is lowered to the specific target for phase equilibrium study.
Because the precipitation of phases occurs from a uniform liquid rather than a chaotic mixture of powders, the system stabilizes much faster. This significantly accelerates the achievement of chemical equilibrium.
Understanding the Trade-offs
The Cost of Skipping Pre-melting
Omitting the superheating step saves a small amount of initial heating time, but usually results in a net loss of efficiency.
Without pre-melting, the sample retains the "memory" of its original non-homogeneous state. This forces you to wait much longer for the system to equilibrate at the target temperature, often yielding less reliable data.
Thermal History vs. Equilibrium Accuracy
The trade-off here is between immediate target heating and preparation quality.
You invest energy in a short-term high-temperature spike to ensure that the subsequent phase precipitation is governed by thermodynamics rather than the physical history of the powder mixture.
Making the Right Choice for Your Goal
To maximize the reliability of your phase equilibrium studies, apply the pre-melting strategy based on your specific needs:
- If your primary focus is Experimental Accuracy: Use pre-melting to ensure that phase precipitation is derived from a perfectly uniform mother liquor, eliminating errors caused by powder segregation.
- If your primary focus is Time Efficiency: Implement pre-melting to significantly shorten the duration required for the system to settle into chemical equilibrium at the target temperature.
By standardizing your liquid phase first, you ensure your results reflect true material properties rather than sample preparation artifacts.
Summary Table:
| Feature | Without Pre-melting | With Pre-melting (Superheating) |
|---|---|---|
| Sample State | Non-homogeneous powder mixture | Uniform "Mother Liquor" |
| Physical History | Retains powder segregation "memory" | Physical history completely reset |
| Mixing Level | Boundary-limited | Atomic-level homogeneity |
| Equilibrium Speed | Slow; delayed by solid diffusion | Rapid; accelerated precipitation |
| Data Reliability | Lower (affected by initial state) | Higher (true thermodynamic properties) |
Achieve Unmatched Precision in Phase Equilibrium Studies
Don't let sample preparation artifacts compromise your research accuracy. KINTEK provides the high-performance thermal solutions you need to master complex phase transitions. Backed by expert R&D and world-class manufacturing, we offer a comprehensive range of Muffle, Tube, Rotary, Vacuum, and CVD systems, as well as other lab high-temperature furnaces—all fully customizable to your unique experimental parameters.
Ready to elevate your material science outcomes? Contact KINTEK today to discuss your custom furnace needs
Visual Guide
References
- Hamed Abdeyazdan, Evgueni Jak. Phase equilibria in the CuO <sub>0.5</sub> –SbO <sub>1.5</sub> –SiO <sub>2</sub> system. DOI: 10.1111/jace.70123
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Vacuum Heat Treat Sintering Furnace Molybdenum Wire Vacuum Sintering Furnace
- 1700℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- 1400℃ Controlled Inert Nitrogen Atmosphere Furnace
- Vacuum Heat Treat Sintering Furnace with Pressure for Vacuum Sintering
- Vacuum Heat Treat Sintering and Brazing Furnace
People Also Ask
- What is the core function of high-strength fan blades in a vacuum tempering furnace? Ensure Thermal Uniformity.
- Why is a high-vacuum furnace required for CP-Ti annealing? Protect Purity and Prevent Embrittlement
- Why is high-purity argon protection required during the casting of alloy steel samples? Preserve Sample Integrity
- What is the purpose of bottom-entry argon injection? Enhance Lithium-ion Battery Safety & Purge Efficiency
- Why is strict control of vacuum pressure essential during EB-PBF of Ti–6Al–4V? Ensuring Purity and Beam Precision



















