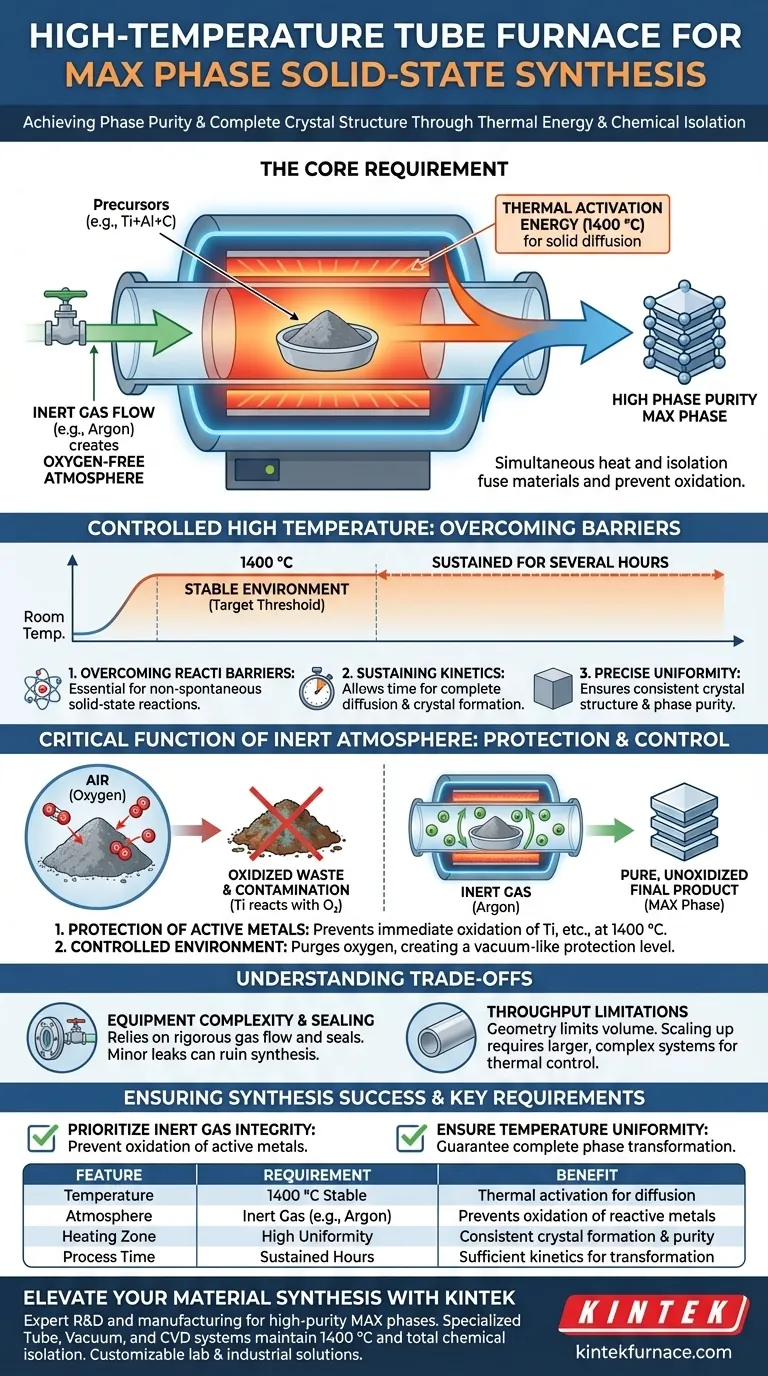
To successfully synthesize MAX phases via solid-state reaction, a high-temperature tube furnace with inert gas protection is mandatory because it simultaneously provides extreme thermal energy and total chemical isolation. Specifically, the furnace maintains a stable 1400 °C environment to drive the reaction kinetics, while a continuous flow of inert gas (such as argon) prevents the oxidation of highly reactive metal powders like titanium.
The Core Requirement
Solid-state synthesis is a battle against both thermodynamic barriers and chemical contamination. The tube furnace system solves this by delivering the thermal activation energy needed to fuse solid materials while maintaining an artificial, oxygen-free atmosphere to ensure the final product attains high phase purity.

The Role of Controlled High Temperature
Overcoming Reaction Barriers
Solid-state reactions do not occur spontaneously at room temperature; the atoms are locked in place. To facilitate the reaction between precursors, the system must provide significant thermal activation energy. A tube furnace is essential because it can reach and sustain the specific 1400 °C threshold required for MAX phase formation.
Sustaining Reaction Kinetics
Reaching the target temperature is only half the battle; the environment must be held stable for the reaction to complete. The furnace ensures the reactants are exposed to this heat for several hours, allowing sufficient time for diffusion and crystal lattice formation. Without this prolonged exposure, the phase transformation would remain incomplete.
Ensuring Phase Purity through Uniformity
In solid-state chemistry, temperature gradients can lead to inconsistent products. The high-temperature tube furnace offers precise temperature uniformity within its heating zone. This ensures that the entire sample undergoes the same thermal history, resulting in a complete crystal structure and high phase purity.
The Critical Function of Inert Atmosphere
Protection of Active Metals
MAX phase precursors often include active metals, such as titanium, which are highly susceptible to reacting with oxygen. If exposed to air at 1400 °C, these metals would oxidize immediately rather than reacting with the other precursors to form the MAX phase. This oxidation would irreversibly contaminate the sample and degrade the material properties.
Creating a Controlled Environment
The tube design allows for the precise management of the internal atmosphere. By introducing a continuous flow of inert gas (typically argon), the system purges atmospheric oxygen and creates a vacuum-like protection level. This isolation is the single most critical factor in achieving a pure, unoxidized final product.
Understanding the Trade-offs
Equipment Complexity and Sealing
While a tube furnace offers superior atmosphere control compared to a box furnace, it requires a rigorous setup. The system relies entirely on the integrity of the gas flow and seals; even a minor leak can introduce enough oxygen to ruin the synthesis of sensitive MAX phases. Operators must constantly verify the tightness of flanges and the purity of the gas source.
Throughput Limitations
The geometry of a tube furnace inherently limits the volume of material that can be processed at once. While it provides the temperature uniformity necessary for high-quality research samples, scaling up production often requires significantly larger, more complex tube systems to maintain that same degree of thermal control.
Ensuring Synthesis Success
The choice of equipment is not just about heating; it is about controlling the thermodynamic and chemical environment.
- If your primary focus is Phase Purity: Prioritize the integrity of your inert gas system (Argon flow) to prevent the oxidation of active metals like titanium.
- If your primary focus is Crystal Structure: Ensure your furnace is calibrated for maximum temperature uniformity to guarantee complete phase transformation across the entire sample.
Success in MAX phase synthesis is defined by the ability to maintain 1400 °C heat without admitting a single molecule of oxygen.
Summary Table:
| Feature | Requirement for MAX Phase Synthesis | Benefit |
|---|---|---|
| Temperature | 1400 °C Stable Environment | Provides thermal activation energy for solid-state diffusion. |
| Atmosphere | Inert Gas (e.g., Argon) | Prevents oxidation of reactive metals like Titanium at high heat. |
| Heating Zone | High Temperature Uniformity | Ensures consistent crystal lattice formation and phase purity. |
| Process Time | Sustained for several hours | Allows sufficient kinetics for complete phase transformation. |
Elevate Your Material Synthesis with KINTEK
Precise control over temperature and atmosphere is the difference between high-purity MAX phases and oxidized waste. Backed by expert R&D and manufacturing, KINTEK offers specialized Tube, Vacuum, and CVD systems designed to maintain a perfect 1400 °C environment with total chemical isolation.
Whether you need customizable lab high-temp furnaces or industrial-scale solutions, we provide the equipment necessary to protect your active metals and ensure complete crystal transformation.
Ready to optimize your solid-state synthesis? Contact our technical experts today to find the perfect furnace for your research.
Visual Guide
References
- Savannah E. Pas, Micah J. Green. Rapid Electrothermal Heating and Molten Salt Etching to Produce Ti <sub>3</sub> C <sub>2</sub> MXenes. DOI: 10.1002/admi.202500355
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 1700℃ Controlled Inert Nitrogen Atmosphere Furnace
- 1400℃ Controlled Inert Nitrogen Atmosphere Furnace
- 1200℃ Controlled Inert Nitrogen Atmosphere Furnace
- 1400℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- 1700℃ High Temperature Laboratory Tube Furnace with Alumina Tube
People Also Ask
- What challenges are associated with inert atmosphere furnaces? Overcome High Costs and Complexity
- What does inert mean in furnace atmospheres? Protect materials from oxidation with inert gases.
- How is atmosphere control managed during furnace operation? Master Precise Gas Environments for Superior Results
- What makes inert atmosphere furnaces different from standard tube furnaces? Key Benefits for Material Protection
- What is the purpose of a chemically inert atmosphere in a furnace? Protect Materials from Oxidation & Contamination



















