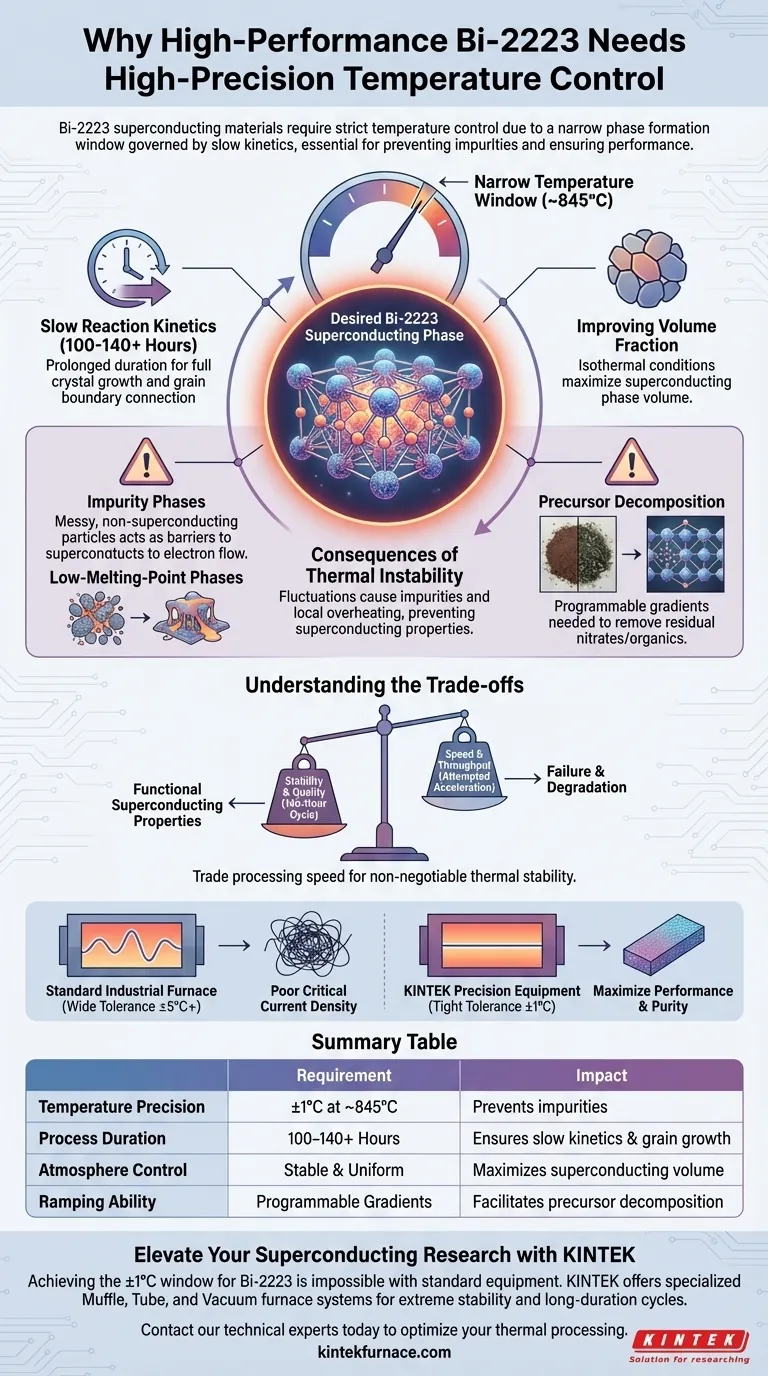
High-performance Bi-2223 superconducting materials demand high-precision temperature control because their specific phase formation occurs within an exceptionally narrow temperature window governed by slow reaction kinetics. Without rigorous thermal stability, typically maintained around 845°C for extended periods, the material suffers from incomplete phase transformation and the proliferation of non-superconducting impurities.
The critical temperature and current density of Bi-2223 depend entirely on maintaining thermodynamic equilibrium over hundreds of hours; even minor temperature fluctuations disrupt this balance, leading to permanent performance degradation.

The Challenge of Phase Formation
The Narrow Temperature Window
The formation of the desired Bi-2223 superconducting phase is thermodynamically fragile. It requires a highly specific temperature, often centered around 845°C, to initiate and sustain the reaction.
Deviating from this precise setpoint disrupts the phase equilibrium. This precision is required to ensure the solid-phase reactions occur exactly as intended without triggering side reactions.
Slow Reaction Kinetics
Unlike materials that react instantly, Bi-2223 features extremely slow reaction kinetics. The atomic rearrangement required to form the superconducting crystal structure takes significant time.
Consequently, heat treatments must often extend up to 140 hours. This prolonged duration allows for the full growth of superconducting grains and the necessary improvement of grain boundary connections.
Improving Volume Fraction
The goal of this long, stable heat treatment is to maximize the volume fraction of the superconducting phase. A higher volume fraction directly correlates to better electrical performance.
By maintaining isothermal conditions, the process ensures that the internal chemical reactions are driven to completion.
Consequences of Thermal Instability
Preventing Impurity Phases
If the temperature fluctuates during the long sintering process, the material will form impurity phases rather than the desired Bi-2223 structure. These non-superconducting residues act as barriers to electron flow.
High-precision equipment eliminates these fluctuations, minimizing residues and ensuring a pure final product.
Managing Low-Melting-Point Phases
Inaccurate temperature control runs the risk of local overheating. This can cause low-melting-point phases within the mixture to melt prematurely.
Controlled heating prevents this melting, preserving the structural integrity required for the subsequent formation of the high-purity superconducting phase.
Precursor Decomposition
Before the main superconducting phase forms, residual nitrates and organic components in the precursor powder must be removed. A programmable furnace allows for specific gradients to discharge these components fully.
This establishes a clean material foundation, ensuring that impurities do not become trapped within the final crystal lattice.
Understanding the Trade-offs
The Cost of Stability vs. Speed
There is no shortcut to high performance with Bi-2223; attempting to accelerate the process by raising temperatures or shortening soak times inevitably leads to failure.
Operators must trade processing speed for thermal stability. The energy and time costs of a 140-hour cycle are high, but they are the non-negotiable price for functional superconducting properties.
Equipment Sensitivity
Standard industrial furnaces often lack the precision required for this specific application. Using equipment with wide tolerance bands (e.g., ±5°C or more) will likely result in a batch with poor critical current density, regardless of the quality of the raw materials.
Making the Right Choice for Your Goal
To maximize the performance of Bi-2223 materials, your equipment strategy must prioritize stability over throughput.
- If your primary focus is critical current density (Jc): Ensure your furnace can maintain ±1°C stability at 845°C for over 100 hours to maximize grain boundary connectivity.
- If your primary focus is purity: Utilize programmable ramping to fully decompose organic precursors before reaching the sintering phase.
Precision in thermal processing is not merely a variable in Bi-2223 production; it is the defining factor that separates a superconductor from a simple ceramic.
Summary Table:
| Factor | Requirement for Bi-2223 | Impact on Material Performance |
|---|---|---|
| Temperature Precision | ±1°C at ~845°C | Prevents non-superconducting impurity phases |
| Process Duration | 100 to 140+ Hours | Ensures slow reaction kinetics & grain growth completion |
| Atmosphere Control | Stable & Uniform | Maximizes volume fraction of superconducting phase |
| Ramping Ability | Programmable Gradients | Facilitates precursor decomposition & impurity removal |
Elevate Your Superconducting Research with KINTEK
Achieving the narrow ±1°C thermal window required for high-performance Bi-2223 is impossible with standard industrial equipment. At KINTEK, we understand that in superconducting material science, precision is the difference between success and failure.
Backed by expert R&D and manufacturing, KINTEK offers specialized Muffle, Tube, and Vacuum furnace systems engineered for extreme stability and programmable long-duration cycles. Our equipment is fully customizable to meet the unique demands of Bi-2223 sintering and other advanced material processes, ensuring you achieve maximum critical current density and phase purity every time.
Ready to optimize your thermal processing? Contact our technical experts today to find the perfect precision furnace for your laboratory.
Visual Guide
References
- Kun Yang, Junwei Liu. Thermal Deformation Behavior and Microstructural Evolution of Multicomponent Mg-Li-Zn-Al-Y Alloys under Hot Compression. DOI: 10.3390/ma17020489
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 2200 ℃ Graphite Vacuum Heat Treat Furnace
- 2200 ℃ Tungsten Vacuum Heat Treat and Sintering Furnace
- Vacuum Heat Treat Sintering Furnace Molybdenum Wire Vacuum Sintering Furnace
- 9MPa Air Pressure Vacuum Heat Treat and Sintering Furnace
- High Temperature Muffle Oven Furnace for Laboratory Debinding and Pre Sintering
People Also Ask
- What is the primary application of vacuum heat treating furnaces in aerospace? Enhance Component Performance with Precision
- How does a vacuum heat treating furnace improve the condition of metal alloys? Achieve Superior Metal Performance
- How does a vacuum heat treat furnace work? Achieve Pristine, High-Performance Results
- How does vacuum heat treating work in terms of temperature and time control? Master Precise Material Transformations
- What is the role of high-precision furnaces in Inconel 718 heat treatment? Master Microstructural Engineering



















