The primary purpose of using a controlled anaerobic environment is to facilitate the process of peat pyrolysis. By heating peat within sealed, oxygen-deprived spaces, the process drives off volatile matter to create a concentrated, high-energy fuel known as peat char.
Core Takeaway By restricting oxygen during heating, raw peat is transformed into peat char (or "smiddy coal"). This transformation significantly increases the fuel's calorific value, making it a viable alternative to charcoal for demanding industrial applications like iron smelting.

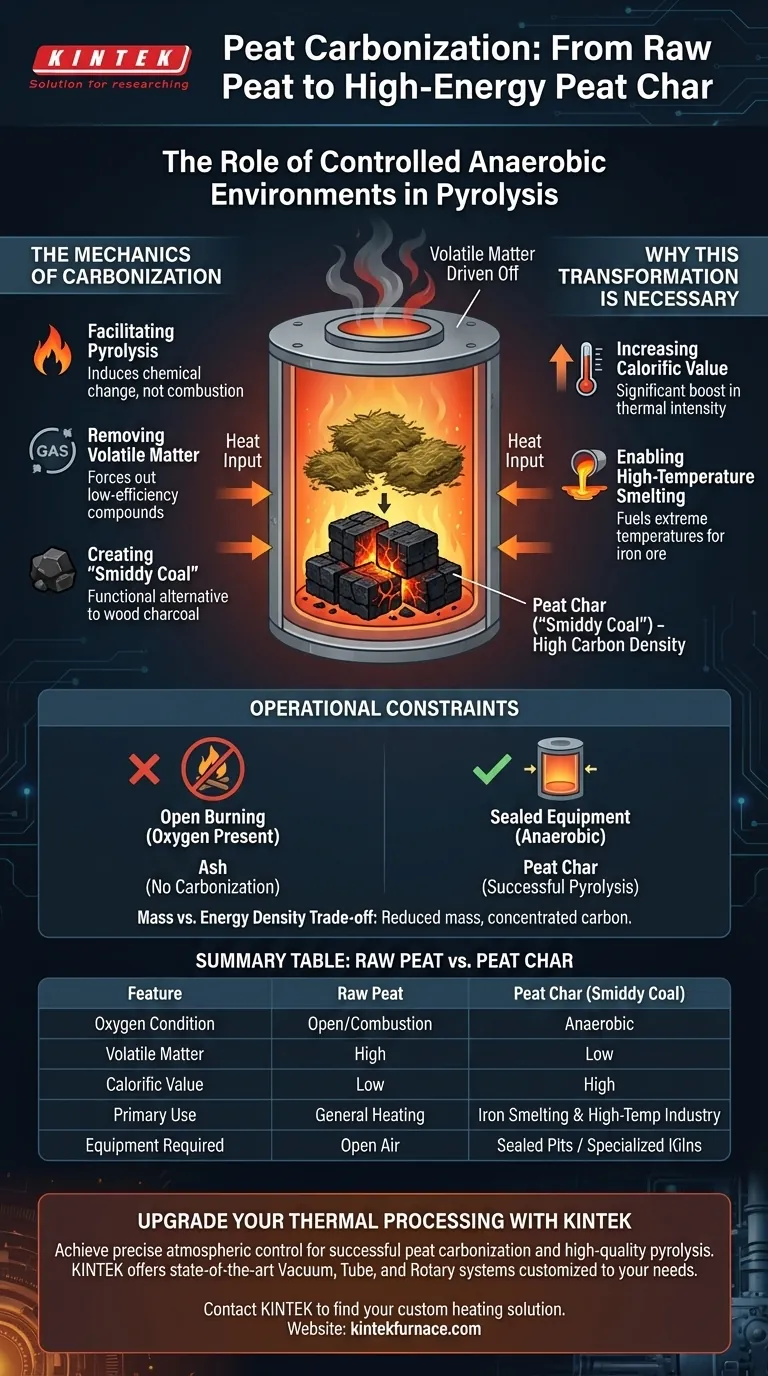
The Mechanics of Carbonization
Facilitating Pyrolysis
To convert peat into a higher-grade fuel, you must induce pyrolysis rather than simple combustion.
This requires a controlled anaerobic (oxygen-free) environment. This is typically achieved using sealed pit-based equipment that isolates the peat from the surrounding air while subjecting it to heat.
Removing Volatile Matter
Raw peat contains a high percentage of volatile components that limit its burning efficiency.
The anaerobic heating process forces these volatile compounds out of the material. What remains is a carbon-dense residue that behaves differently than the raw stock.
Creating "Smiddy Coal"
The end product of this carbonization is peat char, also historically known as smiddy coal.
This material acts as a direct functional alternative to wood charcoal, specifically engineered for high-performance needs.
Why This Transformation is Necessary
Increasing Calorific Value
The most critical outcome of carbonization is a dramatic boost in calorific value.
Raw peat does not naturally possess the energy density required for intense industrial heat. Peat char, having lost its volatile impurities, burns with significantly greater thermal intensity.
Enabling High-Temperature Smelting
The specific goal of producing peat char is to fuel iron smelting processes.
Smelting requires sustained, extreme temperatures that raw peat cannot generate. The controlled anaerobic process upgrades the peat into a fuel capable of melting iron ore effectively.
Understanding the Operational Constraints
The Necessity of Sealed Equipment
You cannot achieve the production of smiddy coal through open burning.
If oxygen is present, the peat will simply burn away into ash. The use of sealed pits is not optional; it is the fundamental requirement to maintain the anaerobic conditions needed for chemical transformation.
Mass vs. Energy Density
The process involves a trade-off between volume and intensity.
By driving off volatile matter, you reduce the overall mass of the material. However, this loss is necessary to concentrate the carbon content required for high-temperature applications.
Making the Right Choice for Your Goal
To determine if this process aligns with your requirements, consider your end-use application:
- If your primary focus is iron smelting: You must use peat char (smiddy coal), as raw peat lacks the necessary calorific value to reach melting temperatures.
- If your primary focus is process setup: You must invest in sealed pit-based equipment to ensure a strictly anaerobic environment for successful pyrolysis.
Mastering the anaerobic environment is the key to unlocking the industrial potential of peat.
Summary Table:
| Feature | Raw Peat | Peat Char (Smiddy Coal) |
|---|---|---|
| Oxygen Condition | Open/Combustion | Anaerobic (Oxygen-Free) |
| Volatile Matter | High | Low (Driven off) |
| Calorific Value | Low | High |
| Primary Use | General Heating | Iron Smelting & High-Temp Industry |
| Equipment Required | Open Air | Sealed Pits / Specialized Kilns |
Upgrade Your Thermal Processing with KINTEK
To achieve successful peat carbonization and high-quality pyrolysis, precise atmospheric control is non-negotiable. KINTEK provides state-of-the-art Vacuum, Tube, and Rotary systems designed to maintain the strictly anaerobic environments necessary for producing high-grade peat char.
Backed by expert R&D and world-class manufacturing, our lab and industrial high-temperature furnaces are fully customizable to your unique research or production needs. Ensure the highest calorific value for your materials today.
Contact KINTEK to find your custom heating solution
Visual Guide
References
- Paul M. Jack. Feeling the Peat: Investigating peat charcoal as an iron smelting fuel for the Scottish Iron Age. DOI: 10.54841/hm.682
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Electric Rotary Kiln Small Rotary Furnace for Activated Carbon Regeneration
- Vacuum Heat Treat Sintering Furnace with Pressure for Vacuum Sintering
- High Temperature Muffle Oven Furnace for Laboratory Debinding and Pre Sintering
- Small Vacuum Heat Treat and Tungsten Wire Sintering Furnace
- 1200℃ Controlled Inert Nitrogen Atmosphere Furnace
People Also Ask
- What are the primary applications of electric rotary kilns? Precision Thermal Processing for High-Value Materials
- What are the main applications of a rotary kiln electric furnace? Achieve Uniform Heat Treatment for Powders
- What are the primary functions of electric rotary kilns? Achieve Precise High-Temperature Processing
- What materials can be processed in an electric rotary kiln? Versatile Solutions for Advanced Materials
- What is an electric rotary kiln and how does it function? Discover Precision Heating for Sensitive Materials











