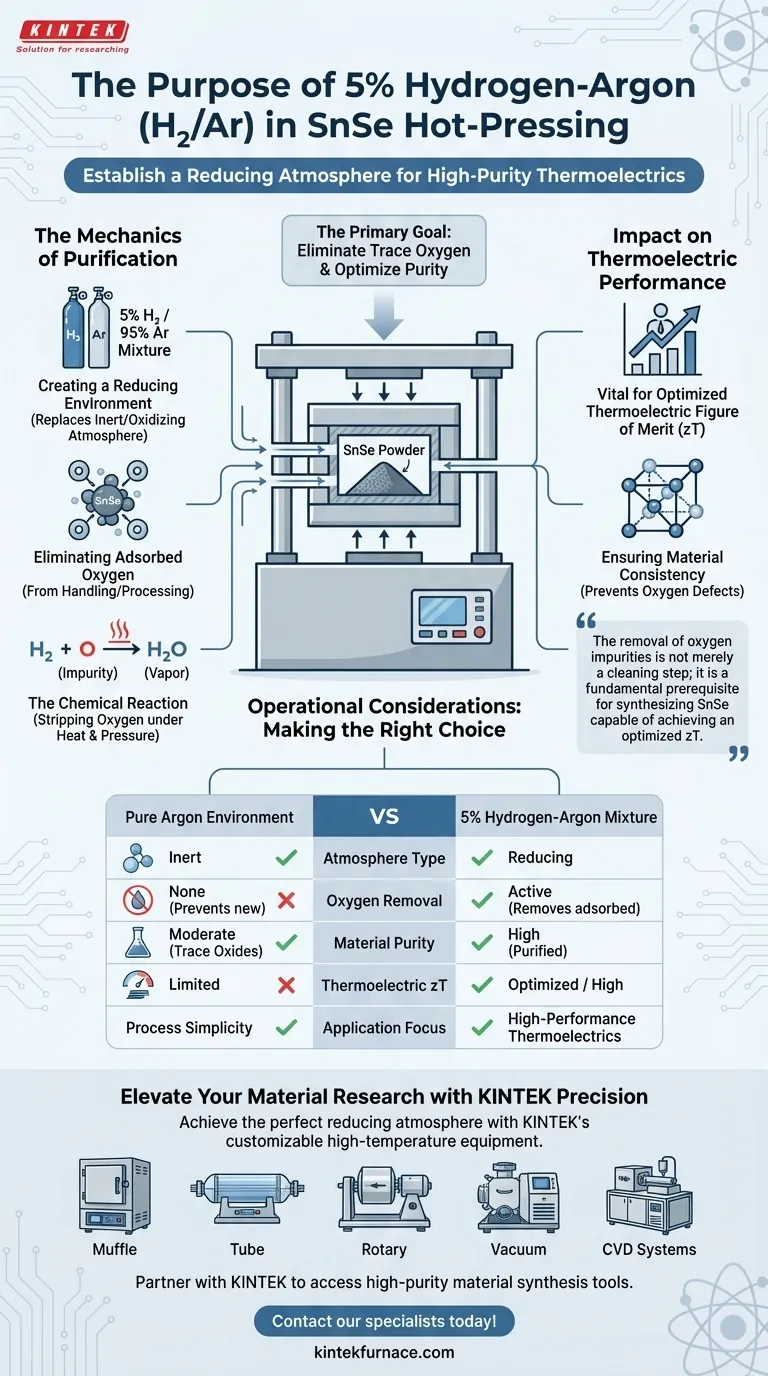
The primary purpose of using a 5% hydrogen-argon mixture is to establish a reducing atmosphere during the hot-pressing process. Specifically, the hydrogen component reacts with and eliminates trace oxygen contamination that the tin selenide (SnSe) may have adsorbed during earlier processing stages. This ensures the final material maintains the high purity levels necessary for performance.
The removal of oxygen impurities is not merely a cleaning step; it is a fundamental prerequisite for synthesizing SnSe capable of achieving an optimized thermoelectric figure of merit (zT).

The Mechanics of Purification
Creating a Reducing Environment
Standard processing environments often introduce contaminants. By introducing a hydrogen-argon mixture, you replace an inert or oxidizing environment with a reducing atmosphere.
Eliminating Adsorbed Oxygen
Tin selenide can adsorb oxygen on its surface during handling or previous mechanical processing. The hydrogen in the mixture actively targets these impurities.
The Chemical Reaction
Under the heat of the press, the hydrogen reacts chemically with the trace oxygen. This reaction effectively strips the oxygen from the material, reversing partial oxidation that may have occurred.
Impact on Thermoelectric Performance
The Link to Figure of Merit (zT)
The primary reference explicitly states that this purification step is vital. Without removing oxygen, the material cannot reach its optimized thermoelectric figure of merit (zT).
Ensuring Material Consistency
Oxygen contamination can act as a defect, altering the intrinsic properties of the semiconductor. The hydrogen treatment ensures the lattice remains close to its intended stoichiometric and chemical state.
Operational Considerations
The Cost of Omission
Skipping the inclusion of hydrogen is a common pitfall in pursuit of simpler processing. However, failing to use a reducing atmosphere leaves adsorbed oxygen within the compacted sample.
Balancing Purity and Complexity
While argon provides an inert background to prevent further reactions, it cannot remove existing oxides. The addition of hydrogen adds a layer of process complexity but is the only way to actively reverse prior contamination.
Making the Right Choice for Your Goal
To maximize the efficacy of your hot-pressing setup, consider your specific performance targets:
- If your primary focus is maximizing zT: You must utilize a reducing agent like hydrogen to eliminate performance-degrading oxygen impurities.
- If your primary focus is process simplicity: You might use pure argon, but you must accept that trace oxidation will likely limit the final performance of the SnSe.
High-performance thermoelectrics require not just precise synthesis, but active purification during the consolidation phase.
Summary Table:
| Feature | Pure Argon Environment | 5% Hydrogen-Argon Mixture |
|---|---|---|
| Atmosphere Type | Inert | Reducing |
| Oxygen Removal | None (Prevents new oxidation) | Active (Removes adsorbed oxygen) |
| Material Purity | Moderate (Contains trace oxides) | High (Purified during pressing) |
| Thermoelectric zT | Limited | Optimized / High |
| Application Focus | Process Simplicity | High-Performance Thermoelectrics |
Elevate Your Material Research with KINTEK Precision
Achieving the perfect reducing atmosphere for SnSe hot-pressing requires reliable, high-temperature equipment. Backed by expert R&D and world-class manufacturing, KINTEK offers a comprehensive range of Muffle, Tube, Rotary, Vacuum, and CVD systems—all fully customizable to meet your specific gas-mixture and thermal processing requirements.
Don't let oxygen impurities compromise your thermoelectric figure of merit (zT). Partner with KINTEK to access the tools needed for high-purity material synthesis.
Contact our laboratory specialists today to find your custom solution!
Visual Guide
References
- Andrew Golabek, Holger Kleinke. Large Improvements in the Thermoelectric Properties of SnSe by Fast Cooling. DOI: 10.3390/ma18020358
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 9MPa Air Pressure Vacuum Heat Treat and Sintering Furnace
- Vacuum Heat Treat Sintering Furnace with Pressure for Vacuum Sintering
- 2200 ℃ Tungsten Vacuum Heat Treat and Sintering Furnace
- Vacuum Heat Treat Furnace with Ceramic Fiber Liner
- 600T Vacuum Induction Hot Press Vacuum Heat Treat and Sintering Furnace
People Also Ask
- What is the function of vacuum & heating in aluminum degassing? Enhancing Composite Integrity & Density
- What is the core advantage of using a Hot Pressing Sintering (HPS) furnace? Enhance SiC/YAG Ceramic Density & Strength
- What role does a vacuum hot pressing furnace play in TiBw/TA15 synthesis? Enhance In-Situ Composite Performance
- What are the mold configuration requirements for sintering non-conductive powders in FAST? Expert Setup Guide
- How does a vacuum system integrated with mechanical and diffusion pumps enhance lead carbonate decomposition? Guide



















