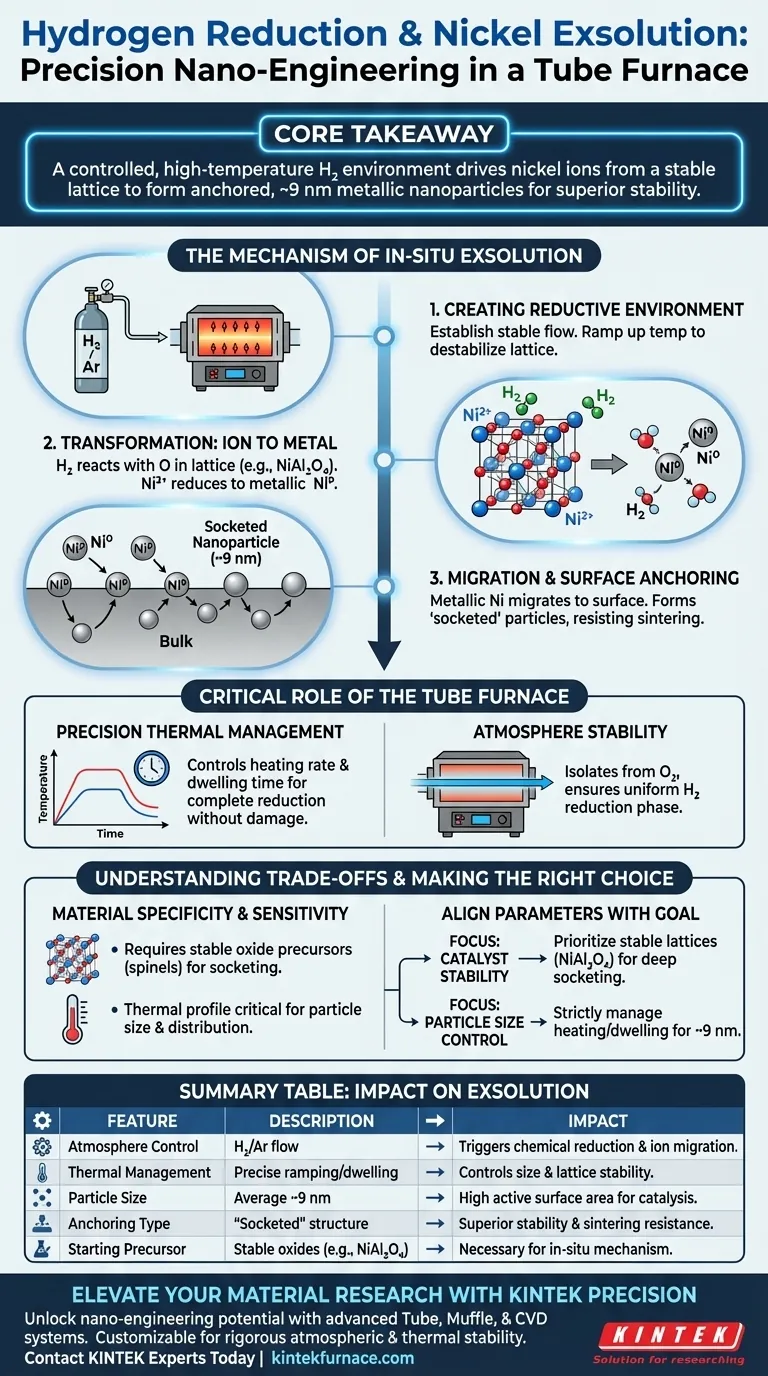
The hydrogen reduction process functions as a precision mechanism for structural transformation. It utilizes the controlled, high-temperature environment of a laboratory tube furnace to chemically extract nickel ions from within a stable crystal lattice and convert them into active metallic nanoparticles anchored to the surface.
Core Takeaway The tube furnace facilitates exsolution by maintaining a stable reducing atmosphere that forces nickel ions to migrate out of bulk oxide structures. This process generates "socketed" metallic nanoparticles (approximately 9 nm) that possess superior stability due to a strong, intrinsic interaction with the support material.

The Mechanism of In-Situ Exsolution
Creating the Reductive Environment
The process begins by establishing a stable reductive atmosphere within the tube furnace, often utilizing a mixture of hydrogen and argon.
The furnace maintains this gas flow while ramping up to high temperatures, creating the thermodynamic conditions necessary to destabilize the nickel within the oxide lattice.
Transformation from Ion to Metal
Inside the furnace, the hydrogen gas reacts with the oxygen bound to the nickel ions residing in stable lattices, such as $\mathbf{NiAl_2O_4}$ or Ni-Mg solid solutions.
This chemical reduction transforms the nickel species from an ionic state into metallic nickel.
Migration and Surface Anchoring
As the nickel reduces, it is forced to migrate from the bulk interior of the material to the outer surface.
Rather than simply sitting on top, these emerging particles become "socketed" into the surface of the support.
This results in metallic nanoparticles with an average size of approximately 9 nm, characterized by a strong metal-support interaction that resists sintering (clumping).
The Critical Role of the Tube Furnace
Precision Thermal Management
The tube furnace allows for the precise management of the heating rate and dwelling time.
Controlling these variables is essential to ensure the metal precursors are fully reduced into active nanoparticles without damaging the underlying support structure.
Atmosphere Stability
Successful exsolution requires a consistent, uninterrupted flow of the reducing gas.
The tube furnace isolates the sample from ambient oxygen, ensuring that the hydrogen reduction phase proceeds efficiently and uniformly across the material.
Understanding the Trade-offs
Material Specificity
This process is not universal; it relies on starting with specific stable oxide lattices (like spinels) that contain the nickel ions.
If the precursor material does not have the correct crystal structure, the "socketing" effect—which provides the stability—will not occur.
Process Sensitivity
The quality of the resulting microstructure is highly sensitive to the thermal profile.
Inadequate heating may result in incomplete reduction, while excessive dwelling times could potentially alter the desired particle size distribution despite the anchoring effect.
Making the Right Choice for Your Goal
To maximize the efficacy of hydrogen reduction for exsolution, align your parameters with your desired outcome:
- If your primary focus is catalyst stability: Prioritize the use of stable oxide lattices like $\mathbf{NiAl_2O_4}$ to ensure the resulting nanoparticles are deeply socketed and resistant to movement.
- If your primary focus is particle size control: Strictly manage the heating rate and dwelling time in the furnace to maintain the average particle size near the optimal 9 nm benchmark.
By precisely controlling the thermal and chemical environment, you transform the tube furnace from a simple heater into a tool for nano-engineering.
Summary Table:
| Feature | Description | Impact on Nickel Exsolution |
|---|---|---|
| Atmosphere Control | Hydrogen/Argon flow | Triggers chemical reduction and ion migration. |
| Thermal Management | Precise ramping/dwelling | Controls nanoparticle size and lattice stability. |
| Particle Size | Average ~9 nm | Ensures high active surface area for catalysis. |
| Anchoring Type | "Socketed" structure | Provides superior stability and sintering resistance. |
| Starting Precursor | Stable oxides (e.g., NiAl2O4) | Necessary for the in-situ exsolution mechanism. |
Elevate Your Material Research with KINTEK Precision
Unlock the full potential of nano-engineering with KINTEK’s advanced thermal solutions. Backed by expert R&D and manufacturing, we provide high-performance Tube, Muffle, Rotary, Vacuum, and CVD systems specifically designed to maintain the rigorous atmospheric and thermal stability required for nickel exsolution and hydrogen reduction processes.
Whether you need to control nanoparticle size or ensure deep-surface anchoring, our systems are fully customizable to meet your unique lab requirements. Achieve superior catalyst stability and consistent research results today.
Ready to optimize your high-temperature processes?
Contact KINTEK Experts Now
Visual Guide
References
- Kyung Hee Oh, Ji Chan Park. Scalable Exsolution‐Derived E‐Ni/m‐MgAlO <sub>x</sub> Catalysts with Anti‐Sintering Stability for Methane Dry Reforming. DOI: 10.1002/smll.202508028
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 1400℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- Laboratory Quartz Tube Furnace RTP Heating Tubular Furnace
- Laboratory Vacuum Tilt Rotary Tube Furnace Rotating Tube Furnace
- 1700℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- Vertical Laboratory Quartz Tube Furnace Tubular Furnace
People Also Ask
- What factors should be considered when selecting a high temperature tube furnace? Ensure Precision and Reliability for Your Lab
- In what scenarios are laboratory high-temperature tube furnaces or muffle furnaces utilized? Study MgTiO3-CaTiO3 Ceramics
- How does a laboratory high-temperature tube furnace contribute to the conversion of electrospun fibers? Expert Insights
- How do high-temperature laboratory tube furnaces ensure environmental stability? Precision Thermal Reduction Tips
- What role do high-performance box or tube furnaces play in LATP sintering? Master Densification & Ionic Conductivity



















