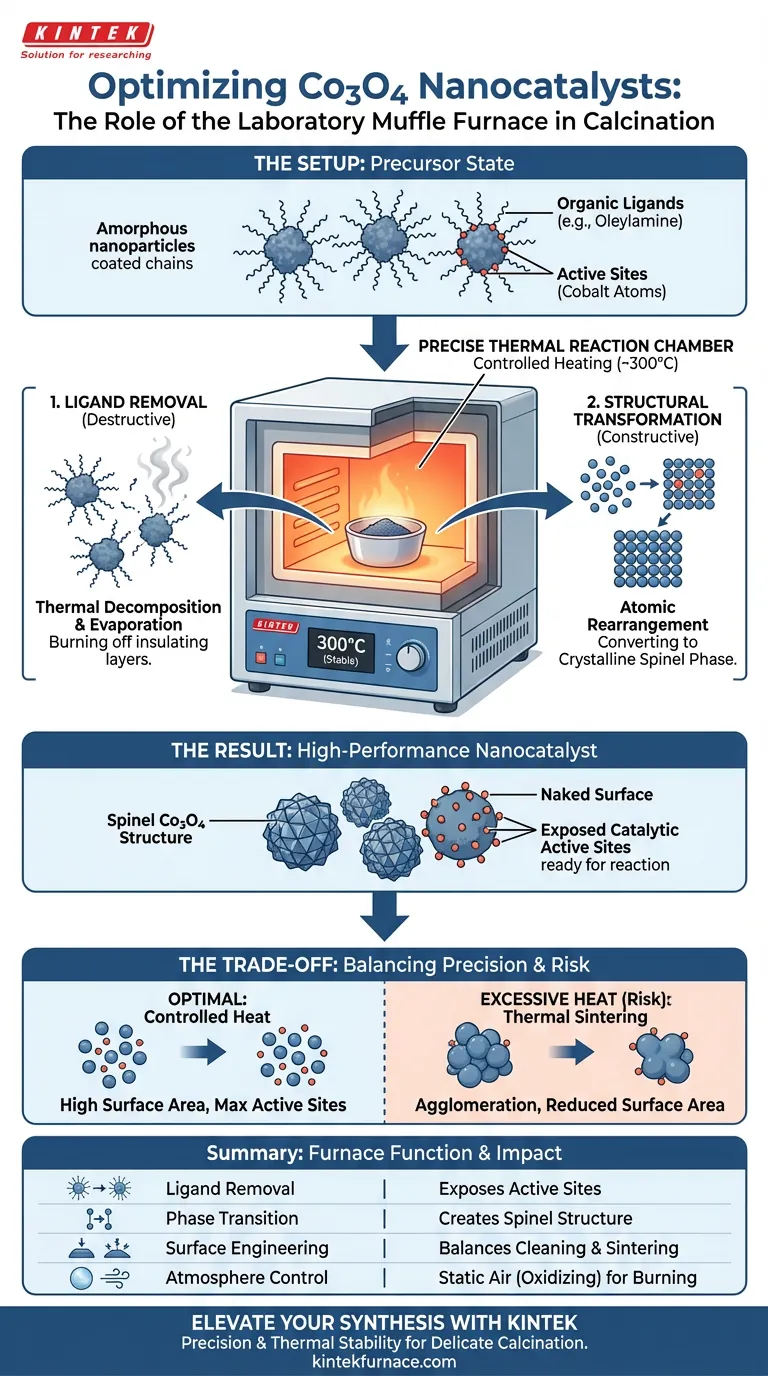
A laboratory muffle furnace acts as the precise thermal reaction chamber required to synthesize high-performance Co3O4 nanocatalysts. By maintaining a stable high-temperature environment, typically centered around 300°C, it facilitates the thermal decomposition necessary to strip away long-chain organic ligands like oleylamine. This controlled heating is the defining step that converts amorphous precursors into a highly crystalline spinel phase, directly exposing the active sites required for catalysis.
The muffle furnace does more than simply dry the material; it drives a critical phase transition that burns off insulating organic layers to reveal the catalytically active spinel structure underneath.
The Process of Ligand Removal
The primary function of the muffle furnace in this specific context is purification through heat. Before calcination, the nanoparticles are often coated in organic stabilizers used during the initial synthesis.
Thermal Decomposition of Organics
The precursors typically contain long-chain organic ligands, such as oleylamine. These molecules stabilize the particles during synthesis but act as contaminants that block chemical reactions during actual use. The muffle furnace provides the thermal energy required to break the chemical bonds of these ligands, causing them to decompose and evaporate.
Exposing Active Sites
If these organic ligands remain on the surface, they physically obstruct the reactant molecules from reaching the cobalt atoms. By ensuring the complete removal of these "capping agents," the furnace creates a "naked" surface. This exposure is essential for maximizing the density of catalytic active sites available for reaction.
Driving Structural Transformation
Beyond purification, the muffle furnace acts as a crystallizer. The heat treatment is not merely destructive (removing ligands) but constructive (building structure).
Conversion from Amorphous Phases
Initially, the cobalt precursors may exist in an amorphous (disordered) or intermediate state. The thermal energy provided by the furnace allows the atoms to mobilize and rearrange themselves. This reorganization transforms the disordered material into a chemically stable structure.
Formation of Spinel Co3O4
The specific goal of this thermal treatment is to achieve the spinel phase of Co3O4. This specific crystalline arrangement is associated with high catalytic activity. The muffle furnace ensures the temperature remains stable enough to promote this crystallization without causing structural collapse.
Understanding the Trade-offs
While the muffle furnace is essential, the parameters must be managed carefully to avoid degrading the catalyst.
The Risk of Thermal Sintering
While high heat improves crystallinity, excessive heat or uncontrolled spikes can cause the nanoparticles to fuse together (sintering). This agglomeration drastically reduces the specific surface area, meaning there is less surface available for catalytic reactions, effectively neutralizing the benefits of the clean surface.
Atmosphere Limitations
Standard muffle furnaces typically operate in a static air environment (an oxidizing atmosphere). This is excellent for burning off organic ligands like oleylamine. However, if your specific synthesis requires a reducing atmosphere (to prevent oxidation) or an inert gas flow, a standard box resistance furnace may require specific modifications or a different furnace type.
Making the Right Choice for Your Goal
To maximize the efficiency of your Co3O4 nanocatalysts, you must tailor the calcination protocol to your specific objectives.
- If your primary focus is Maximizing Catalytic Activity: Ensure the temperature is held strictly at the optimal point (e.g., 300°C) to fully remove oleylamine without inducing particle agglomeration.
- If your primary focus is Structural Stability: Prioritize a furnace with high thermal uniformity to ensure the entire batch transforms into the spinel phase evenly, avoiding pockets of amorphous material.
Success in calcination relies not just on reaching high temperatures, but on maintaining the precise thermal stability required to balance ligand removal with crystal growth.
Summary Table:
| Process Stage | Function of Muffle Furnace | Impact on Co3O4 Nanocatalyst |
|---|---|---|
| Ligand Removal | Thermal decomposition of organic stabilizers (e.g., oleylamine) | Exposes active sites by removing insulating surface layers |
| Phase Transition | Facilitates atomic rearrangement at controlled temperatures | Converts amorphous precursors into a highly active spinel phase |
| Surface Engineering | Provides stable, uniform heating environments | Maximizes specific surface area by balancing cleaning and sintering |
| Atmosphere Control | Static air (oxidizing) environment | Effectively burns off carbonaceous residues from precursors |
Elevate Your Nanomaterial Synthesis with KINTEK
Precision is the difference between a contaminated precursor and a high-performance nanocatalyst. KINTEK provides industry-leading muffle, tube, and vacuum furnaces designed to deliver the thermal stability required for delicate calcination processes. Whether you are removing organic ligands or driving complex phase transitions, our equipment ensures uniform heating without the risk of thermal sintering.
Why choose KINTEK?
- Expert R&D: Systems engineered for precise temperature control (up to 300°C and beyond).
- Versatile Solutions: Choose from Muffle, Rotary, or CVD systems tailored for laboratory and industrial scales.
- Customizable Designs: We adapt our technology to meet your unique atmosphere and thermal requirements.
Ready to optimize your catalytic activity? Contact our experts today to find the perfect furnace for your research.
Visual Guide
References
- Felix T. Haase, Beatriz Roldán Cuenya. Role of Fe decoration on the oxygen evolving state of Co<sub>3</sub>O<sub>4</sub> nanocatalysts. DOI: 10.1039/d3ee02809g
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 1200℃ Muffle Oven Furnace for Laboratory
- 1400℃ Muffle Oven Furnace for Laboratory
- 1800℃ High Temperature Muffle Oven Furnace for Laboratory
- High Temperature Muffle Oven Furnace for Laboratory Debinding and Pre Sintering
- Laboratory Muffle Oven Furnace with Bottom Lifting
People Also Ask
- What is the critical role of a laboratory high-temperature muffle furnace in TiO2/LDH? Unlock Superior Crystallization
- How does a high-temperature laboratory muffle furnace affect material properties? Transform Anodic Oxide Films Fast
- What role does a muffle furnace play in refractory bricks? Enhance Performance and Durability Testing
- What is the role of a laboratory high-temperature muffle furnace in the carbonization of sunflower seed husks?
- Why is a laboratory high-temperature muffle furnace used for BaTiO3? Achieve Optimal Tetragonal Crystalline Phases



















