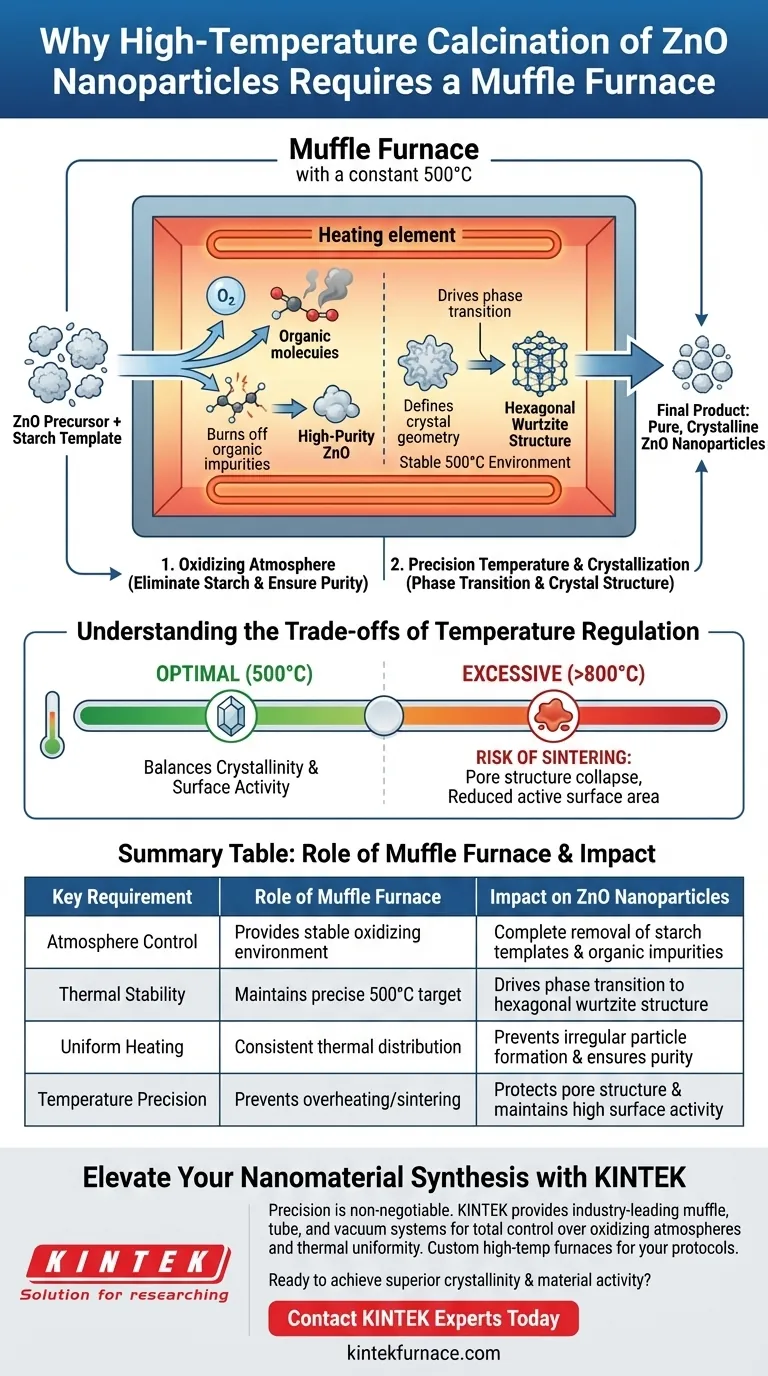
High-temperature calcination in a muffle furnace is strictly required to ensure the simultaneous removal of organic templates and the precise crystallization of the final product. Specifically, it provides a stable, oxidizing environment around 500°C that enables zinc species to undergo phase transition while chemically decomposing starch templates to leave behind pure Zinc Oxide (ZnO).
The muffle furnace acts as a dual-function reactor: it burns off organic impurities through controlled oxidation while maintaining the rigorous thermal stability needed to grow the specific hexagonal wurtzite crystal structure characteristic of high-quality ZnO.

The Critical Role of the Oxidizing Atmosphere
Eliminating the Starch Template
The primary challenge in synthesizing ZnO nanoparticles involves removing the organic framework used during formation. The muffle furnace provides the necessary oxidizing atmosphere to systematically decompose the starch template.
Ensuring Chemical Purity
Without a continuous supply of oxygen at high temperatures, organic residues would contaminate the final material. The furnace ensures the complete elimination of residual organic matter, resulting in a high-purity crystalline product.
Precision Temperature Control and Crystallization
Facilitating Phase Transition
The transformation of precursor materials into final oxides is a thermodynamic process. The muffle furnace maintains a constant temperature (typically 500°C) which drives the zinc species to complete their phase transition effectively.
Defining the Crystal Structure
Temperature stability is directly linked to crystal geometry. The superior temperature control accuracy of the muffle furnace ensures that ZnO crystals grow according to a strict hexagonal wurtzite structure.
Uniform Thermal Environment
Beyond just reaching temperature, the environment must be uniform. A stable thermal environment ensures that precursors react fully and consistently, preventing the formation of irregular or amorphous particles.
Understanding the Trade-offs of Temperature Regulation
The Risk of Sintering
While high heat is necessary, excessive temperatures can be detrimental. If the furnace temperature is set too high (e.g., approaching 800°C), it can lead to severe sintering, causing the pore structure to collapse and reducing the material's active surface area.
Balancing Crystallinity and Activity
There is a delicate balance between forming strong crystals and maintaining surface activity. Accurate temperature regulation is the primary method for balancing material crystallinity with surface oxygen vacancies and adsorption activity.
Making the Right Choice for Your Goal
To optimize your ZnO synthesis, align your furnace parameters with your specific material requirements:
- If your primary focus is High Purity: Prioritize the oxidizing atmosphere capabilities of the furnace to ensure the starch template is fully decomposed.
- If your primary focus is Crystal Structure: Focus on the temperature control accuracy to ensure the stable growth of the hexagonal wurtzite phase without overheating.
Success in ZnO calcination relies on leveraging the muffle furnace not just as a heater, but as a precision instrument for chemical purification and structural engineering.
Summary Table:
| Key Requirement | Role of Muffle Furnace | Impact on ZnO Nanoparticles |
|---|---|---|
| Atmosphere Control | Provides stable oxidizing environment | Complete removal of starch templates & organic impurities |
| Thermal Stability | Maintains precise 500°C target | Drives phase transition to hexagonal wurtzite structure |
| Uniform Heating | Consistent thermal distribution | Prevents irregular particle formation and ensures purity |
| Temperature Precision | Prevents overheating/sintering | Protects pore structure and maintains high surface activity |
Elevate Your Nanomaterial Synthesis with KINTEK
Precision is non-negotiable when engineering hexagonal wurtzite structures and high-purity ZnO. KINTEK provides industry-leading muffle, tube, and vacuum systems designed to give you total control over oxidizing atmospheres and thermal uniformity. Backed by expert R&D and manufacturing, our high-temp lab furnaces are fully customizable to meet your unique calcination protocols.
Ready to achieve superior crystallinity and material activity?
Visual Guide
References
- Roumaissa Djafarou, Tarek Hidouri. Starch-Assisted Eco-Friendly Synthesis of ZnO Nanoparticles: Enhanced Photocatalytic, Supercapacitive, and UV-Driven Antioxidant Properties with Low Cytotoxic Effects. DOI: 10.3390/ijms26020859
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 1400℃ Muffle Oven Furnace for Laboratory
- 1700℃ High Temperature Muffle Oven Furnace for Laboratory
- 1800℃ High Temperature Muffle Oven Furnace for Laboratory
- 1200℃ Muffle Oven Furnace for Laboratory
- High Temperature Muffle Oven Furnace for Laboratory Debinding and Pre Sintering
People Also Ask
- How is a laboratory high-temperature muffle furnace utilized in g-C3N4 synthesis? Optimize Your Thermal Polycondensation
- What is the significance of using a laboratory high-temperature muffle furnace for metal phosphate catalysts?
- How does a high-temperature laboratory muffle furnace affect material properties? Transform Anodic Oxide Films Fast
- How is a laboratory muffle furnace used in 3D-printed PP-CF cross-linking? Achieve Thermal Stability at 150 °C
- What role does a muffle furnace play in refractory bricks? Enhance Performance and Durability Testing



















