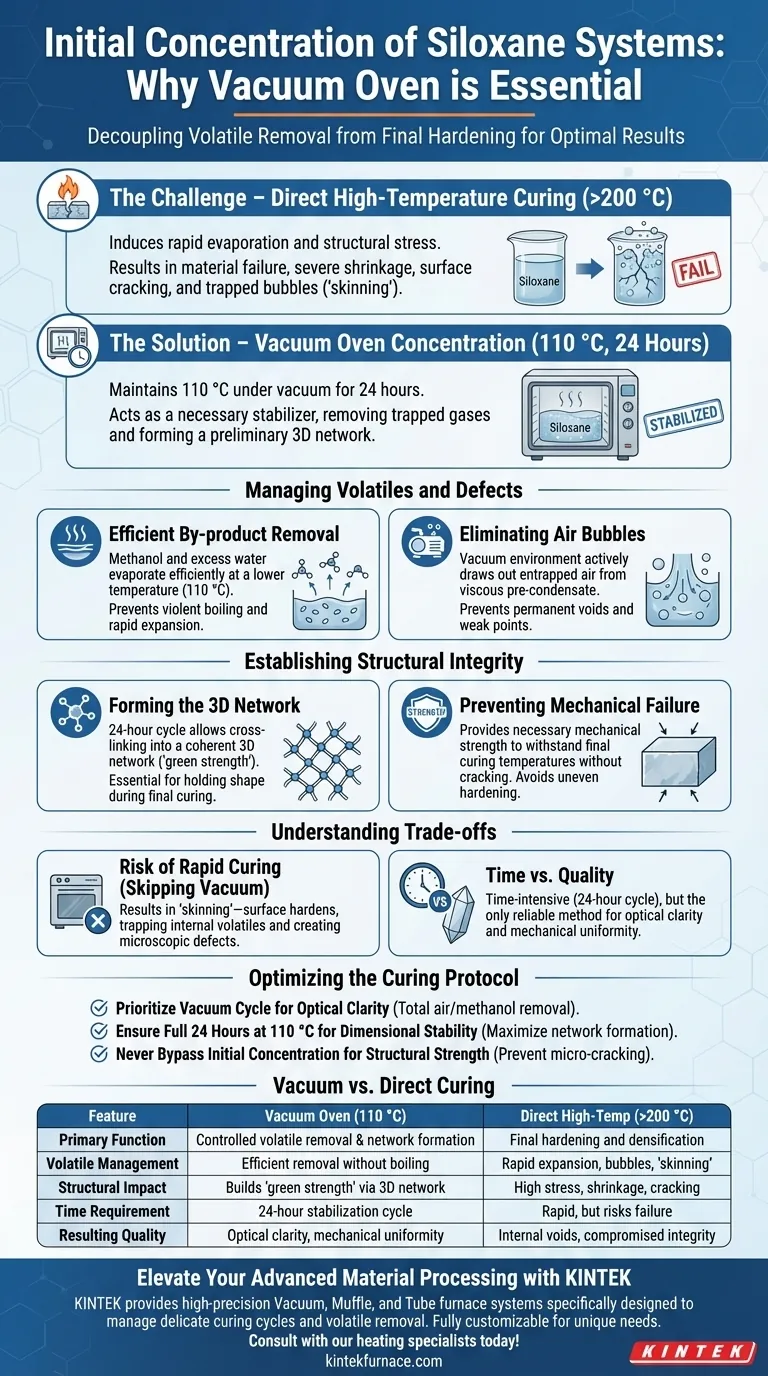
The initial concentration of siloxane systems requires a vacuum oven to decouple volatile removal from final hardening. By maintaining a temperature of 110 °C under vacuum for 24 hours, this process removes reaction by-products like methanol and water without triggering the rapid, destructive shrinkage caused by direct high-temperature thermal curing.
Direct high-temperature curing induces rapid evaporation and structural stress, leading to material failure. The vacuum oven stage acts as a necessary stabilizer, removing trapped gases and forming a preliminary three-dimensional network to ensure the material has the mechanical strength to withstand final curing without cracking.

Managing Volatiles and Defects
Efficient By-product Removal
Siloxane synthesis reactions generate volatile by-products, specifically methanol and excess water.
Using a vacuum oven at 110 °C allows these substances to evaporate efficiently at a lower thermal threshold. This prevents the violent boiling or rapid expansion that would occur if the material were immediately exposed to high heat.
Eliminating Air Bubbles
As the siloxane system condenses, the pre-condensate becomes increasingly viscous.
This viscosity makes it difficult for air bubbles to escape naturally. The vacuum environment actively draws out entrapped air, preventing the formation of permanent voids or weak points within the material structure.
Establishing Structural Integrity
Forming the Three-Dimensional Network
The primary goal of the initial concentration phase is to establish a stable chemical backbone.
The 24-hour cycle allows the siloxane to cross-link into a coherent three-dimensional network. This "green strength" is essential for the material to hold its shape during subsequent processing steps.
Preventing Mechanical Failure
Directly exposing the uncured system to final curing temperatures (typically around 200 °C) causes aggressive volume changes.
Without the initial vacuum stage, the material lacks the mechanical strength to resist this stress. This leads to severe shrinkage and surface cracking as the material hardens unevenly.
Understanding the Trade-offs
The Risk of Rapid Curing
Attempting to accelerate production by skipping the vacuum stage often results in "skinning."
The surface hardens before the internal volatiles escape, trapping gas inside. This results in a compromised internal structure filled with microscopic defects.
Time vs. Quality
The vacuum process is time-intensive, requiring a full 24-hour cycle at 110 °C.
While this creates a bottleneck compared to rapid thermal curing, it is the only reliable method to ensure optical clarity and mechanical uniformity in siloxane systems.
Optimizing the Curing Protocol
To achieve a defect-free siloxane finish, align your process with the following priorities:
- If your primary focus is Optical Clarity: Prioritize the vacuum cycle to ensure total removal of air bubbles and methanol, which causes hazing.
- If your primary focus is Dimensional Stability: Ensure the 110 °C phase runs the full 24 hours to maximize network formation before introducing high heat.
- If your primary focus is Structural Strength: Never bypass the initial concentration step, as it prevents the micro-cracking that weakens the final product.
Properly staging the cure ensures the material is robust enough to survive the final transformation into a high-performance solid.
Summary Table:
| Feature | Vacuum Oven Concentration (110 °C) | Direct High-Temperature Curing (>200 °C) |
|---|---|---|
| Primary Function | Controlled volatile removal & network formation | Final hardening and densification |
| Volatile Management | Efficient removal of methanol/water without boiling | Rapid expansion leading to bubbles and 'skinning' |
| Structural Impact | Builds 'green strength' via 3D network | High stress; causes shrinkage and cracking if unstaged |
| Time Requirement | 24-hour stabilization cycle | Rapid, but risks catastrophic material failure |
| Resulting Quality | Optical clarity and mechanical uniformity | Internal voids and compromised structural integrity |
Elevate Your Advanced Material Processing with KINTEK
Don't let structural defects compromise your siloxane systems. KINTEK provides high-precision Vacuum, Muffle, and Tube furnace systems specifically designed to manage delicate curing cycles and volatile removal.
Backed by expert R&D and manufacturing, our systems are fully customizable to meet your unique laboratory or production needs—ensuring dimensional stability and optical clarity every time. Consult with our heating specialists today to find your perfect thermal solution!
Visual Guide
References
- Max Briesenick, Guido Kickelbick. Thermal Post-Cross-Linking of Siloxane/Silsesquioxane Hybrids with Polycyclic Aromatic Units for Tailored Softening Behavior in High-Temperature Applications. DOI: 10.3390/molecules30173532
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Vacuum Heat Treat Sintering and Brazing Furnace
- Vacuum Heat Treat Sintering Furnace Molybdenum Wire Vacuum Sintering Furnace
- Dental Porcelain Zirconia Sintering Ceramic Vacuum Press Furnace
- Vacuum Hot Press Furnace Machine for Lamination and Heating
- Vacuum Hot Press Furnace Machine Heated Vacuum Press
People Also Ask
- What are some applications of vacuum brazing? Achieve Strong, Clean Joints in Aerospace and More
- What are the advantages of using a vacuum heat treatment furnace? Achieve Superior Material Quality and Control
- What is the significance of vacuum brazing in modern manufacturing? Achieve Strong, Pure Joints for Critical Applications
- What is the role of high-precision furnaces in Inconel 718 heat treatment? Master Microstructural Engineering
- What is the temperature of a vacuum furnace brazing? Optimize Your Joint Strength and Cleanliness

















