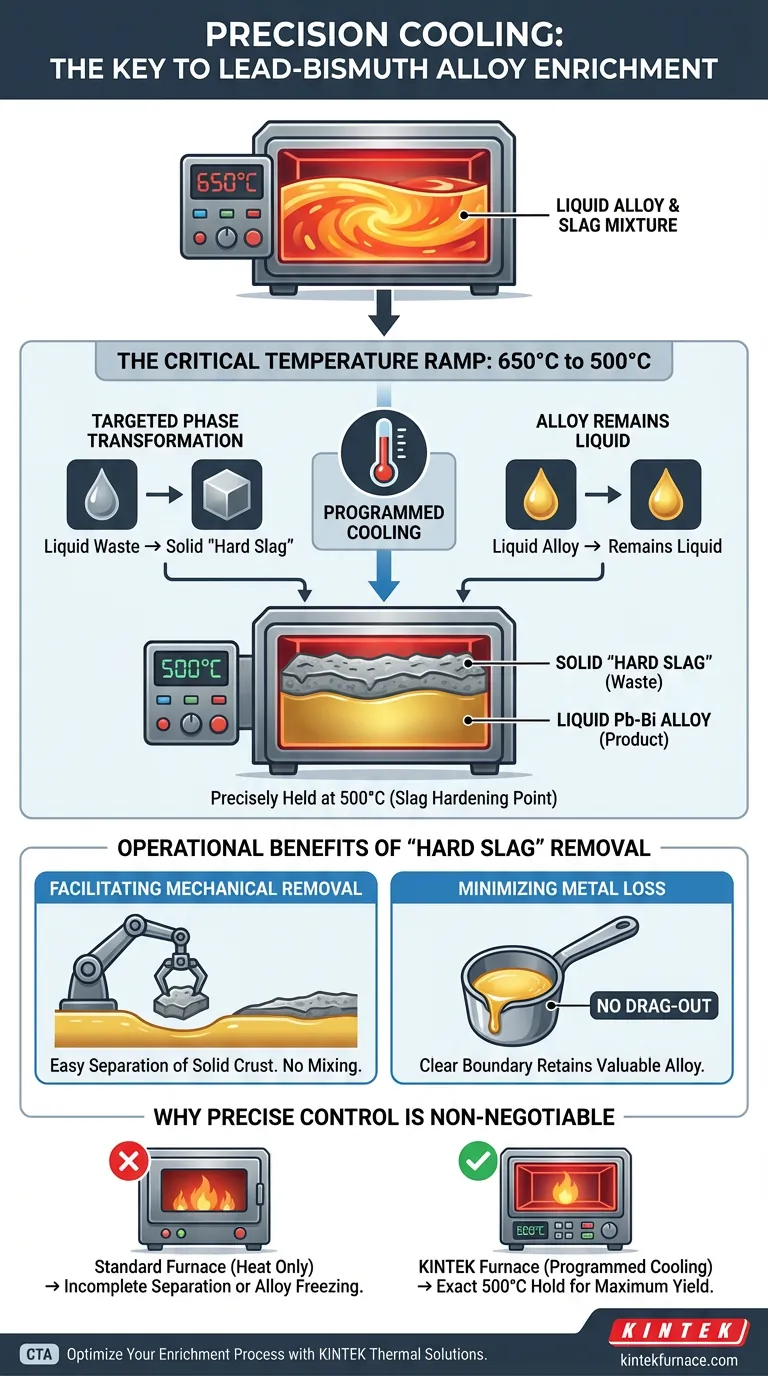
Precise thermal management is the key to efficient impurity separation. An industrial electric furnace is necessary in the final enrichment stage to execute a specific, programmed temperature drop from 650°C to 500°C. This controlled cooling solidifies the waste slag while keeping the lead-bismuth alloy liquid, enabling clean physical separation.
By utilizing programmed cooling to induce a selective phase change, the furnace transforms liquid contaminants into a solid crust that can be easily removed without sacrificing the valuable liquid metal alloy.

The Mechanics of Selective Solidification
Targeted Phase Transformation
The primary function of the furnace in this stage is not heating, but programmed cooling.
The goal is to force a physical state change in the lead metaborate slag. By lowering the temperature, you cause this specific waste material to transition from a liquid state into a solid "hard slag."
The Critical Temperature Window
Success depends on a precise descent from 650°C down to 500°C.
The furnace must control this ramp and hold the temperature at 500°C. This is the "slag hardening temperature," the specific point where the waste solidifies but the lead-bismuth alloy remains in a liquid state.
Operational Benefits of Hard Slag Removal
Facilitating Mechanical Removal
When slag is in a liquid state, it is difficult to distinguish and separate from the liquid metal.
Once the furnace cools the mixture to 500°C, the slag forms a solid crust. This allows for straightforward mechanical removal of the impurities from the surface of the liquid metal.
Minimizing Metal Loss
Attempting to clean liquid slag often results in "drag-out," where valuable alloy is accidentally removed along with the waste.
Solidifying the slag creates a clear boundary between waste and product. This significantly reduces metal loss, ensuring that the maximum amount of enriched lead-bismuth is retained during the cleaning process.
Understanding the Trade-offs
The Necessity of Advanced Control
A standard furnace that only provides heat is insufficient for this process.
The equipment must be capable of active or programmed cooling. This requirement likely increases equipment complexity or cost compared to simple heating units, as it requires precise modulation of temperature decreases.
Precision is Non-Negotiable
There is very little margin for error in this thermal window.
If the furnace fails to cool to exactly 500°C, the slag will remain liquid, preventing separation. Conversely, overcooling could risk solidifying the alloy itself, halting the process entirely.
Optimizing High-Purity Enrichment
To ensure the highest efficiency in your lead-bismuth enrichment process, consider the following:
- If your primary focus is Material Yield: Ensure your furnace controls are calibrated to hold exactly at 500°C to create a distinct solid slag crust, preventing the accidental removal of liquid alloy.
- If your primary focus is Process Speed: Utilize equipment with automated cooling profiles to transition from 650°C to 500°C as rapidly as the material properties allow.
By leveraging precise temperature control to induce phase changes, you transform a complex purification challenge into a straightforward mechanical task.
Summary Table:
| Process Feature | Temperature/Action | Benefit to Enrichment |
|---|---|---|
| Starting Point | 650°C (Liquid State) | Complete melting of alloy and slag mixture |
| Cooling Target | 500°C (Hold Point) | Solidifies 'hard slag' while metal remains liquid |
| Separation Method | Mechanical Removal | Easy extraction of solid crust from liquid metal surface |
| Yield Optimization | Phase Control | Minimizes metal 'drag-out' and reduces material loss |
Optimize Your Enrichment Process with KINTEK
Precision is non-negotiable when separating high-value alloys from waste. KINTEK provides industry-leading thermal solutions, including Muffle, Tube, Rotary, Vacuum, and CVD systems, engineered for the rigorous demands of lead-bismuth enrichment.
Backed by expert R&D and manufacturing, our furnaces offer the advanced programmed cooling and thermal stability required to hit the critical 500°C slag-hardening window every time. Whether you need a standard setup or a fully customizable high-temp furnace tailored to your unique metallurgy, KINTEK ensures maximum material yield and process efficiency.
Ready to minimize metal loss and streamline your purification?
Contact KINTEK experts today for a custom consultation
Visual Guide
References
- Investigation of the Process of Increasing Bismuth Content in Lead Alloys Using the Oxygen Oxidation Method. DOI: 10.3390/pr13051276
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 2200 ℃ Tungsten Vacuum Heat Treat and Sintering Furnace
- 1700℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- 1400℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- 1400℃ Muffle Oven Furnace for Laboratory
- Vacuum Heat Treat Furnace with Ceramic Fiber Liner
People Also Ask
- What is the function of vacuum & heating in aluminum degassing? Enhancing Composite Integrity & Density
- Why are synthesized CdS nanorods dried in a laboratory vacuum oven? Preserving Nanostructure and Chemical Integrity
- What is the function of injecting water in wood thermal modification? Unlock Superior Stability and Hydrophobicity
- What is the purpose of bottom-entry argon injection? Enhance Lithium-ion Battery Safety & Purge Efficiency
- What functions does glucose perform in lithium-ion sieve synthesis? Enhance Carbothermal Reduction for LiMnO2 Purity



















