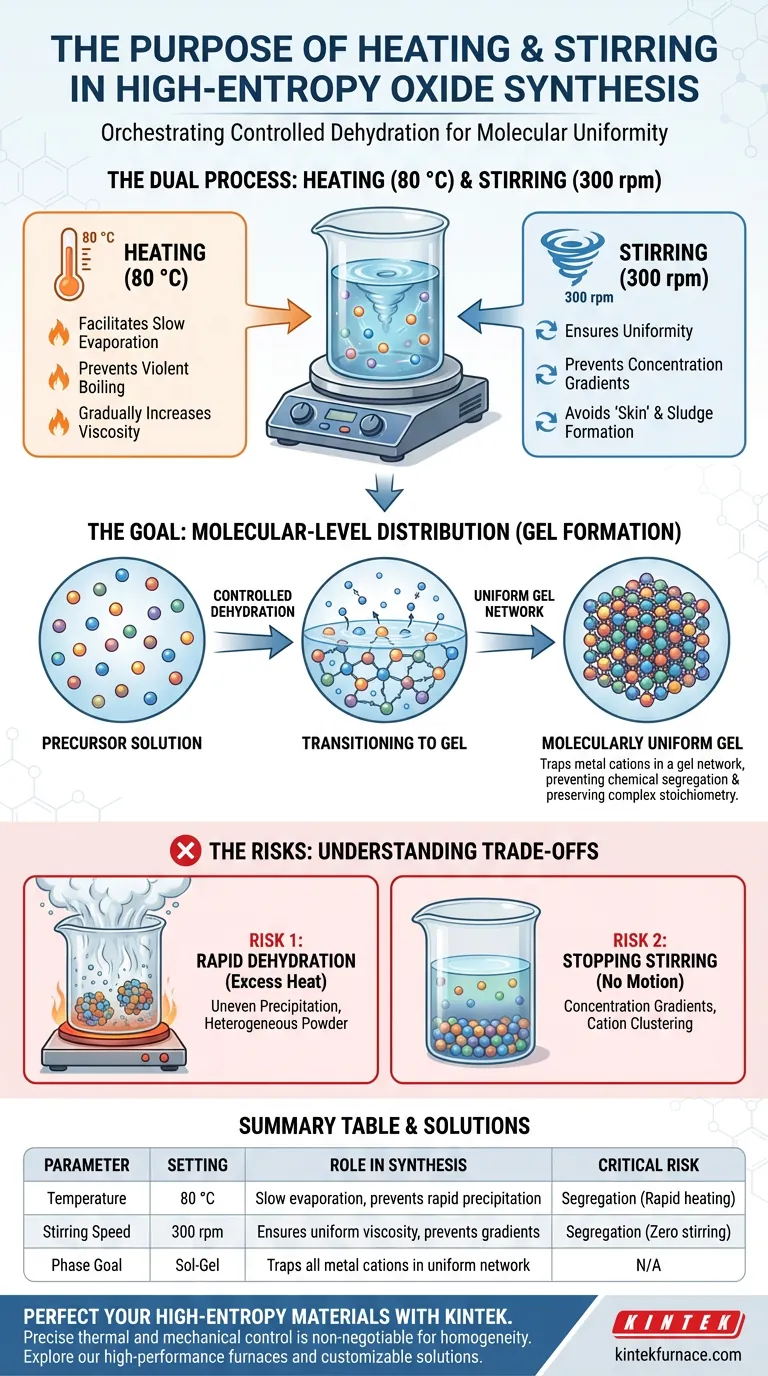
The primary purpose of heating to 80 °C and maintaining 300 rpm stirring is to orchestrate a controlled dehydration process that transforms a precursor solution into a uniform gel. This specific environment facilitates slow solvent evaporation, which gradually increases the solution's viscosity. By managing this transition carefully, you prevent the different metal components from separating or precipitating prematurely.
High-entropy oxides require the precise combination of multiple distinct elements, each with different chemical behaviors. This heating and stirring process ensures that all metal cations are locked into a molecularly uniform network, preventing solubility differences from ruining the material's homogeneity.

The Mechanics of Gel Formation
Facilitating Slow Evaporation
The temperature of 80 °C serves as a driver for evaporation without inducing violent boiling.
This thermal input allows the solvent to leave the system at a steady, manageable rate. It creates a gradual transition rather than a rapid phase change, which is essential for maintaining stability in the solution.
Increasing Solution Viscosity
As the solvent evaporates, the concentration of the solute increases, leading to higher viscosity.
Mechanical stirring at 300 rpm ensures that this viscosity increase happens uniformly throughout the beaker. It prevents the formation of "skin" on the surface or thick sludge at the bottom, ensuring the entire volume thickens as a single unit.
Preventing Chemical Segregation
Addressing Solubility Variations
In high-entropy synthesis, you are dealing with multiple metal cations, each possessing different solubility limits.
Without controlled stirring and heating, elements with lower solubility would precipitate (crash out) of the solution first. This would lead to a chemically segregated product where the elements are clumped together rather than mixed.
Achieving Molecular-Level Distribution
The ultimate goal of this process is to trap the metals in a gel network before they can separate.
By keeping the solution moving and evaporating the solvent slowly, you force the system to freeze into a "molecularly uniform distribution." The gel structure locks the elements in place relative to one another, preserving the complex stoichiometry required for high-entropy oxides.
Understanding the Trade-offs
The Risk of Rapid Dehydration
While it might be tempting to increase the temperature to speed up synthesis, doing so undermines the process.
Excessive heat causes rapid solvent loss, which triggers immediate, uneven precipitation. This bypasses the gelation phase entirely and results in a heterogeneous powder lacking the unique properties of a high-entropy oxide.
The Necessity of Constant Motion
Stopping the stirring to "let the solution settle" is a critical error in this context.
If the mechanical agitation ceases, concentration gradients will form immediately. This allows specific cations to cluster together, destroying the random, uniform distribution that defines high-entropy materials.
Making the Right Choice for Your Goal
To ensure synthesis success, you must prioritize the integrity of the gel network over the speed of production.
- If your primary focus is Material Homogeneity: Stick strictly to the 80 °C/300 rpm regimen to ensure all cations remain mixed at the molecular level during the liquid-to-gel transition.
- If your primary focus is Process Consistency: Monitor the viscosity changes visually; the stirring must maintain a vortex to prevent localized precipitation as the mixture thickens.
The quality of a high-entropy oxide is defined by its disorder and uniformity; this heating and stirring step is the gatekeeper that ensures those properties are locked in.
Summary Table:
| Parameter | Setting | Role in Synthesis |
|---|---|---|
| Temperature | 80 °C | Facilitates slow solvent evaporation without boiling; prevents rapid precipitation. |
| Stirring Speed | 300 rpm | Ensures uniform viscosity; prevents concentration gradients and surface skinning. |
| Phase Goal | Sol-Gel | Transforms solution into a molecularly uniform network trapping all metal cations. |
| Critical Risk | Segregation | Rapid heating or zero stirring leads to elements 'crashing out' based on solubility. |
Perfect Your High-Entropy Materials with KINTEK
Precise thermal and mechanical control is the foundation of successful advanced material synthesis. At KINTEK, we understand that maintaining exact temperatures and stirring rates is non-negotiable for high-entropy oxide homogeneity.
Backed by expert R&D and world-class manufacturing, we provide high-performance Muffle, Tube, Rotary, Vacuum, and CVD systems, along with customizable lab high-temp furnaces tailored to your unique research needs. Ensure process consistency and molecular-level distribution in every batch.
Ready to elevate your laboratory capabilities? Contact KINTEK today for a consultation on our customizable heating solutions!
Visual Guide
References
- Milad Zehtab Salmasi, Hua Song. Tuning High-Entropy Oxides for Oxygen Evolution Reaction Through Electrocatalytic Water Splitting: Effects of (MnFeNiCoX)3O4 (X = Cr, Cu, Zn, and Cd) on Electrocatalytic Performance. DOI: 10.3390/catal15090827
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 1400℃ Muffle Oven Furnace for Laboratory
- Laboratory Muffle Oven Furnace with Bottom Lifting
- 1800℃ High Temperature Muffle Oven Furnace for Laboratory
- 1700℃ High Temperature Muffle Oven Furnace for Laboratory
- High Temperature Muffle Oven Furnace for Laboratory Debinding and Pre Sintering
People Also Ask
- How do repeat sintering processes and specialized sintering molds address the technical challenges of manufacturing oversized flywheel rotor components? Expand Scale and Integrity
- Why are synthesized CdS nanorods dried in a laboratory vacuum oven? Preserving Nanostructure and Chemical Integrity
- What is the function of injecting water in wood thermal modification? Unlock Superior Stability and Hydrophobicity
- Why is multiple remelting necessary for Bi-Sb alloys? Achieve Perfect Compositional Uniformity Today
- How does the choice of ceramic molds affect the results when preparing steel sample ingots? Ensure Maximum Sample Purity



















