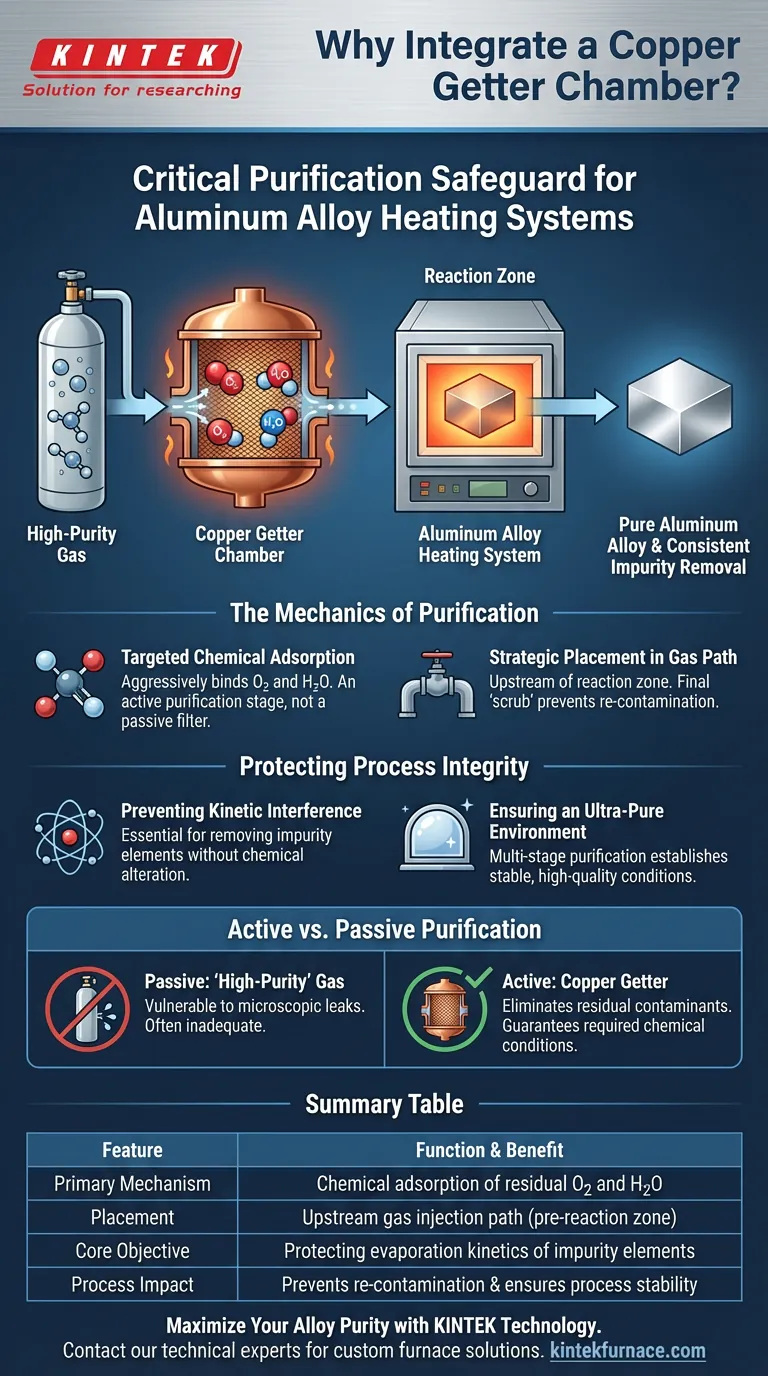
A copper getter chamber acts as a critical purification safeguard integrated directly into the gas injection path of aluminum alloy heating systems. Its specific purpose is to chemically adsorb residual oxygen and moisture from gases before they enter the high-temperature reaction zone. This ensures that the environment remains ultra-pure, preventing contaminants from disrupting the delicate evaporation kinetics necessary for removing impurities from the alloy.
The copper getter chamber serves as a final, active barrier against contamination, eliminating microscopic traces of oxygen and water to maintain the precise chemical conditions required for effective alloy purification.
The Mechanics of Purification
Targeted Chemical Adsorption
The primary mechanism of the copper getter is chemical adsorption. Unlike simple mechanical filters that trap particulate matter, the copper getter chemically reacts with specific impurities.
It aggressively targets and binds with traces of oxygen and moisture found within the gas stream. This transforms the getter into an active purification stage rather than a passive sieve.
Strategic Placement in the Gas Path
The chamber is installed specifically within the gas injection path, upstream of the reaction zone.
By positioning the purification stage here, the system ensures that high-purity gases are "scrubbed" one final time immediately before use. This prevents any re-contamination that might occur in the delivery lines.
Protecting Process Integrity
Preventing Kinetic Interference
The ultimate goal of this integration is to protect the evaporation kinetics of impurity elements.
During the heating process, specific impurities must be evaporated out of the aluminum alloy. If oxygen or moisture is present, they can alter the chemical behavior of these elements, making them difficult or impossible to remove.
Ensuring an Ultra-Pure Environment
The getter enables a "multi-stage purification" strategy.
By removing the final traces of contaminants, the system establishes an ultra-pure environment. This stability is essential for consistent, high-quality results in the heat treatment of aluminum alloys.
Understanding the Operational Requirements
The Necessity of Active Purification
A common pitfall is assuming that sourcing "high-purity" gas cylinders is sufficient for sensitive alloy processing.
The integration of a copper getter acknowledges that passive purity is often inadequate. Without this active chemical adsorption stage, microscopic leaks or residual contamination in the lines could compromise the entire heating process, rendering the theoretical purity of the source gas irrelevant.
Making the Right Choice for Your Goal
To maximize the effectiveness of your aluminum alloy processing, consider how this component aligns with your objectives:
- If your primary focus is Process Stability: Ensure your system includes a copper getter to eliminate oxygen and moisture variables that cause inconsistent impurity removal.
- If your primary focus is Alloy Purity: Rely on the getter's chemical adsorption to prevent environmental contamination from interfering with the evaporation of unwanted elements.
Integrating a copper getter is not just about gas delivery; it is about guaranteeing the chemical conditions required for precise metallurgical results.
Summary Table:
| Feature | Function & Benefit |
|---|---|
| Primary Mechanism | Chemical adsorption of residual $O_2$ and $H_2O$ |
| Placement | Upstream gas injection path (pre-reaction zone) |
| Core Objective | Protecting evaporation kinetics of impurity elements |
| Process Impact | Prevents re-contamination and ensures process stability |
| Material Benefit | Maintains ultra-pure environments for high-quality alloys |
Maximize Your Alloy Purity with KINTEK Technology
Don't let microscopic contaminants compromise your metallurgical results. KINTEK provides industry-leading thermal solutions, including Muffle, Tube, Rotary, and Vacuum systems, all customizable to integrate advanced purification stages like copper getter chambers. Backed by expert R&D and precision manufacturing, we help you maintain the exact chemical conditions required for superior material science.
Ready to elevate your heat treatment precision? Contact our technical experts today to discuss your custom furnace needs.
Visual Guide
References
- Aleksandar M. Mitrašinović, Milinko Radosavljević. Modeling of Impurities Evaporation Reaction Order in Aluminum Alloys by the Parametric Fitting of the Logistic Function. DOI: 10.3390/ma17030728
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- CF KF Flange Vacuum Electrode Feedthrough Lead Sealing Assembly for Vacuum Systems
- Stainless Steel Quick Release Vacuum Chain Three Section Clamp
- Multi Heating Zones CVD Tube Furnace Machine for Chemical Vapor Deposition Equipment
- Magnesium Extraction and Purification Condensing Tube Furnace
- Ultra Vacuum Electrode Feedthrough Connector Flange Power Lead for High Precision Applications
People Also Ask
- Why is a vacuum sealing process necessary for the synthesis of TaAs2 single crystals? Ensuring Purity in CVT Method
- How does a Mass Flow Controller influence 2D superlattices? Precision CVD Control for Sub-10nm Patterning
- Why is a stainless steel autoclave with a Teflon liner necessary for BiVO4? Ensure Purity & High Performance
- What is the function of a Teflon-lined stainless steel autoclave in the hydrothermal synthesis of Bi2O3 precursors?
- Why is a PTFE-lined stainless steel autoclave used for Ni12P5 synthesis? Key Benefits for Nanomaterial Production



















